Abstract
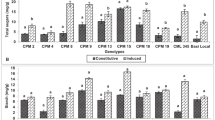
There is a complex interplay of biochemical interactions which determine the host plant reaction to herbivory. We studied the Chilo partellus-induced biochemical plant defense system in sorghum. Present studies found that the total protein, starch and total sugar contents were significantly lower, while antioxidant enzymes like ascorbate oxidase (AO), catalase (CAT), ascorbate peroxidase (APX), phenyl ammonia lyase (PAL), tyrosine ammonia lyase (TAL), and nonenzymatic antioxidants such as total phenols, total antioxidants and ferric ion reducing antioxidant power (FRAP) higher in the seedlings of all the test sorghum genotypes as compared to susceptible genotype, Swarna both under healthy and C. partellus damaged conditions. Further, the C. partellus damage resulted in significant increase in amounts of all the test constitutional, enzymatic and nonenzymatic biochemicals in the test genotypes, however their percent increase was highly variable across genotypes. These studies demonstrate that both constitutive and C. partellus damage induced enzymatic and nonenzymatic antioxidants like APX, AO, PAL, TAL, CAT, total phenols and FRAP were significantly greater in IS 2205, IS 18,551, ICSV 1, ICSV 700 and ICSV 93,046 than in susceptible check (Swarna) indicating their role in plant defense. Furthermore, these genotypes can be used in sorghum breeding for resistance against C. partellus.




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Aebi, H. (1984). Catalase in vitro Methods in Enzymology, 105, 121–126.
Ali, M. B., Hahn, E. J., & Paek, K. Y. (2005). Effects of light intensities on antioxidant enzymes and malondildehyde content during short-term acclimatization on micro propagated Phalaenopsis plantlet Environmental and Experimental Botany, 54, 109–120.
Asada, K. (1992). Ascorbate peroxidase–a hydrogen peroxide scavenging enzyme in plants. Physiologia Plantarum, 85, 235–241.
Asada, K. (2006). Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiology, 141(2), 391–396.
Benzie, I. F., & Strain, J. J. (1999). Ferric reducing /antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods in Enzymology, 299, 15–27.
Bhoi, T. K., Trivedi, N., Kumar, H., Tanwar, A. K., & Dhillon, M. K. (2021). Biochemical defense in maize against Chilo partellus (Swinhoe) through activation of enzymatic and nonenzymatic antioxidants. Indian Journal of Experimental Biology, 59, 54–63.
Bi, J. L., & Felton, G. W. (1995). Foliar oxidative stress and insect herbivory: primary compounds, secondary metabolites and reactive oxygen species as components of induced resistance. Journal of Chemical Ecology, 21, 1511–1530.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Chen, Y., Ni, X., & Buntin, G. D. (2009). Physiological, nutritional, and biochemical bases of corn resistance to foliage-feeding fall armyworm. Journal of Chemical Ecology, 35, 297–306.
Clegg, K. M. (1956). The application of the anthrone reagent to the estimation of starch in cereals. Journal of the Science of Food and Agriculture, 7, 40–44.
Cornelissen, T. G., & Fernandes, G. W. (2001). Patterns of attack by herbivores on the tropical shrub Bauhinia brevipes (Leguminosae): vigour or chance? European Journal of Entomology, 98, 37–40.
Dhillon, M. K., & Chaudhary, D. P. (2015). Biochemical interactions for antibiosis mechanism of resistance to Chilo partellus (Swinhoe) in different maize types. Arthropod-Plant Interactions, 9(4), 373–382.
Dhillon, M. K., & Chaudhary, D. P. (2018). Physicochemical mechanisms of resistance in sorghum to Chilo partellus (Swinhoe). Indian Journal of Experimental Biology, 59, 29–38.
Dhillon, M. K., & Kumar, S. (2017). Amino acid profiling of Sorghum bicolor vis-à-vis Chilo partellus (Swinhoe) for biochemical interactions and plant resistance. Arthropod-Plant Interactions, 11(4), 537–550.
Diallinas, G., Pateraki, I., Sanmartin, M., Scossa, A., & Stilianou, E. (1997). Melon ascorbate oxidase: cloning of a multigene family, induction during fruit development and repression by wounding. Plant Molecular Biology, 34, 759–770.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28, 350–356.
Felton, G. W., Donato, K. K., Broadway, R. M., & Duffey, S. S. (1992). Impact of oxidized plant phenolics on the nutritional quality of dietary protein to a noctuid herbivore. Journal of Insect Physiology, 38, 277–285.
Fritz, R. R., Hodcins, D. S., & Abell, C. W. (1976). Phenylalanine ammonia lyase induction and purification from yeast and clearance in mammals. Journal of Biological Chemistry, 251, 4646–4650.
Gill, S. S., & Tuteja, N. (2010). Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiology and Biochemistry, 48, 909–930.
He, J., Chen, F., Chen, S., Lv, G., Deng, Y., Fang, Z., Guan, Z., & He, C. (2011). Chrysanthemum leaf epidermal infestation. Journal of Plant Physiology, 168(7), 687–693.
Hildebrand, D. F., Rodriquez, J. G., Brown, G. C., Luu, K. T., & Volden, C. S. (1986). Peroxidative responses of leaves in two soybean genotypes injured by two spotted spider mite (Acari: Tetranychidae). Applied Entomology and Zoology, 20, 348–349.
Howe, G. A., & Jander, G. (2008). Plant immunity to insect herbivores. Annual Review of Plant Biology, 59, 41–66.
Howe, G. A., & Schilmiller, A. L. (2002). Oxylipin metabolism in response to stress. Current Opinion in Plant Biology, 5, 230–236.
Huang, H., Ullah, F., Zhou, D. X., Yi, M., & Zhao, Y. (2019). Mechanisms of ROS regulation of plant development and stress responses. Frontiers in Plant Science, 10, 800.
Huang, Y., Sharma, H. C., & Dhillon, M. K. (2013). Bridging conventional and molecular genetics of sorghum insect resistance. In A. K. Paterson (Ed.), Genomics of the Saccharinae, Plant Genetics and Genomics: crops and models (Vol. 11, pp. 367–389). Springer.
Kabre, G. B., & Ghorpade, S. A. (1999). Susceptibility to maize stem borer, Chilo partellus (Swinhoe) in relation to sugars, proteins and free amino acids content of maize germplasm and F1 hybrids. Journal of Insect Science, 12, 37–40.
Kumar, H. (2018). Behavioral, biological and biochemical studies in sorghum for resistance to Chilo partellus (Swinhoe). M.Sc. Thesis. Division of Entomology, ICAR-Indian Agricultural Research Institute, New Delhi, India, 56 pp.
Libik-Konieczny, M., Surówka, E., Kuźniak, E., Nosek, M., & Miszalski, Z. (2011). Effects of Botrytis cinerea and Pseudomonas syringae infection on the antioxidant profile of Mesembryanthemum crystallinum C3/CAM intermediate plant. Journal of Plant Physiology, 168(10), 1052–1059.
Maffei, M. E., Mithöfer, A., & Boland, W. (2007). Before gene expression: early events in plant–insect interaction. Trends in Plant Science, 12(7), 310–316.
Prieto, P., Pineda, M., & Aguilar, M. (1999). Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Analytical Biochemistry, 269, 337–341.
Rani, P. U., & Jyothsna, Y. (2010). Biochemical and enzymatic changes in rice plants as a mechanism of defense. Acta Physioliae Plantarum, 32, 695.
Roitsch, T. (1999). Source-sink regulation by sugar and stress. Current Opinion in Plant Biology, 2, 198–206.
Sau, A. K., Dhillon, M. K., & Trivedi, N. (2022). Activation of antioxidant defense in maize in response to attack by Sesamia inferens (Walker). Phytoparasitica. https://doi.org/10.1007/s12600-022-00996-2
Sharma, H. C. (1993). Host plant resistance to insects in sorghum and its role in integrated pest management. Crop Protection, 12, 11–34.
Sharma, H. C., Dhillon, M. K., Pampapathy, G., & Reddy, B. V. S. (2007). Inheritance of resistance to spotted stem borer, Chilo partellus in sorghum, Sorghum bicolor Euphytica, 156, 117–128.
Sharma, H. C., Leuschner, K., Nwanze, K. F., & Taneja, S. L. (1992). Techniques to screen sorghum for resistance to insect pests. Information Bulletin No. 32. International Crops Research Institute for the Semi-Arid Tropics, Patancheru 502 324, Andhra Pradesh, India, 48 pp.
Sharma, H. C., Taneja, S. L., Kameswara Rao, N., & Prasada Rao, K. E. (2003). Evaluation of sorghum germplasm for resistance to insect pests. Information Bulletin No. 63. International Crops Research Institute for the Semi-Arid Tropics, Patancheru 502 324, Andhra Pradesh, India, 177 pp.
Sharma, H. C., War, A. R., Pathania, M., Sharma, S. P., Akbar, S. M., & Munghate, R. S. (2016). Elevated CO2 influences host plant defense response in chickpea against Helicoverpa armigera Arthropod-Plant Interactions, 10(2), 171–181.
Singleton, V. L., & Rossi, J. A. (1965). Colorimetry of total phenolics with phosphomolybdic- phosphotungestic acid reagents. American Journal of Enology and Viticulture, 16, 144–158.
Smith, C. M., & Clement, S. L. (2012). Molecular bases of plant resistance to arthropods. Annual Review of Entomology, 57, 309–328.
Thorpe, T. A., & Beaudoin-Eagan, L. D. (1985). Tyrosine and phenylalanine ammonia lyase activities during shoot initiation in tobacco callus cultures. Plant Physiology, 78, 438–441.
Tománková, K., Luhová, L., Petřivalský, M., Peč, P., & Lebeda, A. (2006). Biochemical aspects of reactive oxygen species formation in the interaction between Lycopersicon spp. and Oidium neolycopersici Physiological and Molecular Plant Pathology, 68(1–3), 22–32.
Vashisth, S., Jaba, J., Sharma, S. P., & Sharma, H. C. (2022). Biochemical mechanisms of induced resistance to Chilo partellus in sorghum. International Journal of Pest Management. https://doi.org/10.1080/09670874.2022.2036863
Wu, J., & Baldwin, I. T. (2010). New insights into plant responses to attack from insect herbivores. Annual Review of Genetics, 44, 1–24.
Zhao, L. Y., Chen, J. L., Cheng, D. F., Sun, J. R., Liu, Y., & Tian, Z. (2009). Biochemical and molecular characterizations of Sitobion avenae–induced wheat defense responses. Crop Protection, 28, 435–442.
Acknowledgements
The necessary facilities and senior research fellowship to the first author by ICAR-Indian Agricultural Research Institute, New Delhi are gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
Mukesh K. Dhillon and Hemant Kumar conceptualized and designed the study. Experimental set up, data collection and analysis were performed by Hemant Kumar and Tanmaya K. Bhoi. The manuscript was written by Mukesh K. Dhillon and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent for publication
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, H., Dhillon, M.K. & Bhoi, T.K. Stress-induced defense in sorghum in response to attack by the spotted stem borer, Chilo partellus (Swinhoe). Phytoparasitica 51, 49–61 (2023). https://doi.org/10.1007/s12600-022-01037-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-022-01037-8