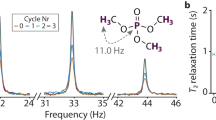
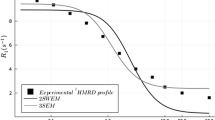
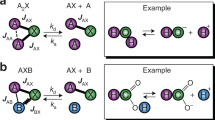
Abstract
Nuclear magnetic resonance (NMR) magnetization-transfer (MT) experiments provide a convenient tool for studying rapid sub-second membrane-transport processes in situ in metabolically active cells. These experiments are used with membrane-permeable substances when separate (resolved) NMR signals are observed from their populations inside and outside the cells. Here, we provide a description of the theory and practice of the most common NMR MT experiments that have been used to study membrane-transport processes in human erythrocytes (red blood cells; RBCs). The procedures, involved in the analysis of the experimental data for defining mechanisms of transport, and for estimating values of kinetic parameters in the corresponding mathematical models, are given special attention.







Similar content being viewed by others
Abbreviations
- B-M:
-
Bloch–McConnell
- DST:
-
Differential saturation transfer
- EXSY:
-
Exchange spectroscopy
- FID:
-
Free induction decay
- Ht :
-
Haematocrit
- IT:
-
Inversion transfer
- MT:
-
Magnetization transfer
- NMR:
-
Nuclear magnetic resonance
- NOE:
-
Nuclear Overhauser effect
- RBC:
-
Red blood cell
- RF:
-
Radio-frequency
- ST:
-
Saturation transfer
References
Abel EW, Coston TPJ, Orrell KG, Šik V, Stephenson D (1986) Two-dimensional NMR exchange spectroscopy. Quantitative treatment of multisite exchanging systems. J Magn Reson 70:34–53
Alger JR, Prestegard JH (1977) Investigation of peptide bond isomerization by magnetization transfer NMR. J Magn Reson 27:137–141
Ardenkjær-Larsen JH, Fridlund B, Gram A, Hansson G, Hansson L, Lerche MH, Servin R, Thaning M, Golman K (2003) Increase in signal-to-noise ratio of >10,000 times in liquid-state NMR. Proc Natl Acad Sci USA 100:10158–10163
Bain AD, Duns GJ (1997) Chemical exchange measurements in NMR. In: Batta G, Kövér KE, Szántay C (eds) Methods for structure elucidation by high-resolution NMR. Elsevier, Amsterdam, pp 227–263
Bellon SF, Chen D, Johnston ER (1987) Quantitative 1D exchange spectroscopy. J Magn Reson 73:168–173
Bloch F (1946) Nuclear induction. Phys Rev 70:460–474
Bloch R (1973) Inhibition of glucose transport in the human erythrocyte by cytochalasin B. Biochemistry 12:4799–4801
Boulanger Y, Vinay P, Desroches M (1985) Measurement of a wide range of intracellular sodium concentrations in erythrocytes by 23Na nuclear magnetic resonance. Biophys J 47:553–561
Bremer J, Mendz GL, Moore WJ (1984) Skewed exchange spectroscopy. Two-dimensional method for the measurement of cross relaxation in proton NMR spectroscopy. J Am Chem Soc 106:4691–4696
Bresciani S, Lebl T, Slawin AMZ, O’Hagan D (2010) Fluorosugars: synthesis of the 2,3,4-trideoxy-2,3,4-trifluoro hexose analogues of D-glucose and D-altrose and assessment of their erythrocyte transmembrane transport. Chem Commun 46:5434–5436
Brown TR (1980) Saturation transfer in living systems. Philos Trans R Soc Lond B 289:441–444
Bubb WA, Kirk K, Kuchel PW (1988) Ethylene glycol as a thermometer for X-nucleus spectroscopy in biological samples. J Magn Reson 77:363–368
Bulliman BT, Kuchel PW, Chapman BE (1989) “Overdetermined” one-dimensional NMR exchange analysis. A 1D counterpart of the 2D EXSY experiment. J Magn Reson 82:131–138
Cabantchik ZI, Rothstein A (1974) Membrane proteins related to anion permeability of human red blood cells. J Membrane Biol 15:207–226
Chapman BE, Kuchel PW (1990) Fluoride transmembrane exchange in human erythrocytes measured with 19F NMR magnetization transfer. Eur Biophys J 19:41–45
Chu SC, Pike MM, Fossel ET, Smith TW, Balschi JA, Springer CS (1984) Aqueous shift reagents for high-resolution cationic nuclear magnetic resonance. III. Dy(TTHA)3−, Tm(TTHA)3−, and Tm(PPP)2 7−. J Magn Reson 56:33–47
Claridge TD (2009) High-resolution NMR techniques in organic chemistry, 2nd edn. Elsevier, Amsterdam
Dacie JV, Lewis SM (1975) Practical Haematology, 5th edn. Churchill Livingstone, Edinburgh
Dahlquist FW, Longmuir KJ, Du Vernet RB (1975) Direct observation of chemical exchange by a selective pulse NMR technique. J Magn Reson 17:406–410
Durrant CJ, Hertzberg MP, Kuchel PW (2003) Magnetic susceptibility: further insights into macroscopic and microscopic fields and the sphere of Lorentz. Concept Magn Reson A 18A:72–95
Eliav U, Naumann C, Navon G, Kuchel PW (2009) Double quantum transition as the origin of the central dip in the z-spectrum of HDO in variably stretched gel. J Magn Reson 198:197–203
Engler RE, Johnston ER, Wade CG (1988) Dynamic parameters from nonselectively generated 1D exchange spectra. J Magn Reson 77:377–381
Eytan GD, Kuchel PW (1999) Mechanism of action of P-glycoprotein in relation to passive membrane permeation. Int Rev Cytol 190:175–250
Forsén S, Hoffman RA (1963) Study of moderately rapid chemical exchange reactions by means of nuclear magnetic double resonance. J Chem Phys 39:2892–2901
Gabel SA, O’Connell TM, Murphy E, London RE (1997) Inhibition of glucose transport in human red blood cells by adenosine antagonists. Am J Physiol Cell Physiol 272:C1415–C1419
Gabr RE, Weiss RG, Bottomley PA (2008) Correcting reaction rates measured by saturation-transfer magnetic resonance spectroscopy. J Magn Reson 191:248–258
Griffin JF, Rampal AL, Jung CY (1982) Inhibition of glucose transport in human erythrocytes by cytochalasins: a model based on diffraction studies. Proc Natl Acad Sci USA 79:3759–3763
Gupta RK, Gupta P (1982) Direct observation of resolved resonances from intra- and extracellular sodium-23 ions in NMR studies of intact cells and tissues using dysprosium(III)tripolyphosphate as paramagnetic shift reagent. J Magn Reson 47:344–350
Hills BP, Belton PS (1989) NMR studies of membrane transport. Ann R NMR S 21:99–159
Jeener J, Meier BH, Bachmann P, Ernst RR (1979) Investigation of exchange processes by two‐dimensional NMR spectroscopy. J Chem Phys 71:4546–4553
Johnston ER, Dellwo MJ, Hendrix J (1986) Quantitative 2D exchange spectroscopy using time-proportional phase incrementation. J Magn Reson 66:399–409
Kell DB, Oliver SG (2014) How drugs get into cells: tested and testable predictions to help discriminate between transporter-mediated uptake and lipoidal bilayer diffusion. Front Pharmacol. doi:10.3389/fphar.2014.00231
Kim HW, Rossi P, Shoemaker RK, DiMagno SG (1998) Structure and transport properties of a novel, heavily fluorinated carbohydrate analogue. J Am Chem Soc 120:9082–9083
Kirk K (1990) NMR methods for measuring membrane transport rates. NMR Biomed 3:1–16
Kirk K, Kuchel PW (1985) Red cell volume changes monitored using a new 31P NMR procedure. J Magn Reson 62:568–572
Kirk K, Kuchel PW (1986) Equilibrium exchange of dimethyl methylphosphonate across the human red cell membrane measured using NMR spin transfer. J Magn Reson 68:311–318
Kirk K, Kuchel PW (1988a) The contribution of magnetic susceptibility effects to transmembrane chemical shift differences in the 31P NMR spectra of oxygenated erythrocyte suspensions. J Biol Chem 263:130–134
Kirk K, Kuchel PW (1988b) Physical basis of the effect of hemoglobin on the 31P NMR chemical shifts of various phosphoryl compounds. Biochemistry 27:8803–8810
Kirk K, Kuchel PW, Labotka RJ (1988) Hypophosphite ion as a 31P nuclear magnetic resonance probe of membrane potential in erythrocyte suspensions. Biophys J 54:241–247
Kuchel PW (1987) Steady-state parameters of an enzyme from NMR spin transfer with thermal variation. Biochem J 244:247–248
Kuchel PW (1990) Spin-exchange NMR spectroscopy in studies of the kinetics of enzymes and membrane transport. NMR Biomed 3:102–119
Kuchel PW (2015) Nuclear magnetic resonance spectroscopy. In: Clarke RJ, Khalid MAA (eds) Pumps, channels, and transporters: methods of functional analysis. Wiley, Hoboken, pp 211–243
Kuchel PW, Chapman BE (1983) NMR spin exchange kinetics at equilibrium in membrane transport and enzyme systems. J Theor Biol 105:569–589
Kuchel PW, Naumann C (2008) 2H2O quadrupolar splitting used to measure water exchange in erythrocytes. J Magn Reson 192:48–59
Kuchel PW, Bulliman BT, Chapman BE, Kirk K (1987a) The use of transmembrane differences in saturation transfer for measuring fast membrane transport; application to H13CO3 − exchange across the human erythrocyte. J Magn Reson 74:1–11
Kuchel PW, Chapman BE, Potts JR (1987b) Glucose transport in human erythrocytes measured using 13C NMR spin transfer. FEBS Lett 219:5–10
Kuchel PW, Bulliman BT, Chapman BE, Mendz GL (1988) Variances of rate constants estimated from 2D NMR exchange spectra. J Magn Reson 76:136–142
Kuchel PW, Chapman BE, Xu ASL (1992) Rates of anion transfer across erythrocyte membranes measured with NMR spectroscopy. In: Bamberg E, Passow H (eds) The band 3 proteins: anion transporters, binding proteins and senescent antigens. Elsevier, Amsterdam, pp 105–119
Kuchel PW, Kirk K, King GF (1994) NMR methods for measuring membrane transport. In: Hilderson HJ, Ralston GB (eds) Physicochemical methods in the study of biomembranes. Plenum, New York, pp 247–327
Kuchel PW, Chapman BE, Bubb WA, Hansen PE, Durrant CJ, Hertzberg MP (2003) Magnetic susceptibility: solutions, emulsions, and cells. Concept Magn Reson A 18A:56–71
Kuchel PW, Shishmarev D, Puckeridge M, Levitt MH, Naumann C, Chapman BE (2015) NMR of 133Cs+ in stretched hydrogels: one-dimensional, z- and NOESY spectra, and probing the ion’s environment in erythrocytes. J Magn Reson 261:110–120
Kumar A, Wagner G, Ernst RR, Wüthrich K (1981) Buildup rates of the nuclear Overhauser effect measured by two-dimensional proton magnetic resonance spectroscopy: implications for studies of protein conformation. J Am Chem Soc 103:3654–3658
Labotka RJ (1984) Measurement of intracellular pH and deoxyhemoglobin concentration in deoxygenated erythrocytes by phosphorus-31 nuclear magnetic resonance. Biochemistry 23:5549–5555
Labotka RJ, Omachi A (1987) Erythrocyte anion transport of phosphate analogs. J Biol Chem 262:305–311
Labotka RJ, Lundberg P, Kuchel PW (1995) Ammonia permeability of erythrocyte membrane studied by 14N and 15N saturation transfer NMR spectroscopy. Am J Physiol Cell Physiol 268:C686–C699
Led JJ, Gesmar H (1982) The applicability of the magnetization-transfer NMR technique to determine chemical exchange rates in extreme cases. The importance of complementary experiments. J Magn Reson 49:444–463
London RE (1994) In vivo NMR studies utilizing fluorinated probes. In: Gillies RJ (ed) NMR in physiology and biomedicine. Academic, San Diego, pp 263–277
London RE, Gabel SA (1989) Determination of membrane potential and cell volume by 19F NMR using trifluoroacetate and trifluoroacetamide probes. Biochemistry 28:2378–2382
Maciel GE, Natterstad JJ (1965) Carbon-13 chemical shifts of the carbonyl group. III. Solvent effects. J Chem Phys 42:2752–2759
Macura S, Ernst RR (1980) Elucidation of cross relaxation in liquids by two-dimensional NMR spectroscopy. Mol Phys 41:95–117
Macura S, Huang Y, Suter D, Ernst RR (1981) Two-dimensional chemical exchange and cross-relaxation spectroscopy of coupled nuclear spins. J Magn Reson 43:259–281
Mariappan SVS, Rabenstein DL (1992) Analysis of inversion-magnetization-transfer experiments. J Magn Reson 100:183–188
Marion D, Wüthrich K (1983) Application of phase sensitive two-dimensional correlated spectroscopy (COSY) for measurements of 1H-1H spin-spin coupling constants in proteins. Biochem Bioph Res Co 113:967–974
MathWorks (2005) MATLAB: the language of technical computing. Desktop tools and development environment. MathWorks, Natick
McConnell HM (1958) Reaction rates by nuclear magnetic resonance. J Chem Phys 28:430–431
Morales MF, Horovitz M, Botts J (1962) The distribution of tracer substrate in an enzyme-substrate system at equilibrium. Arch Biochem Biophys 99:258–264
Moré JJ (1978) The Levenberg-Marquardt algorithm: implementation and theory. In: Watson GA (ed) Numerical analysis. Springer, Berlin, pp 105–116
Morris GA, Freeman R (1978) Selective excitation in Fourier transform nuclear magnetic resonance. J Magn Reson 29:433–462
Mulquiney PJ, Kuchel PW (2003) Modelling metabolism with Mathematica: detailed examples including erythrocyte metabolism. CRC, Boca Raton
O’Connell TM, Gabel SA, London RE (1994) Anomeric dependence of fluorodeoxyglucose transport in human erythrocytes. Biochemistry 33:10985–10992
Ohuchi M, Hosono M, Furihata K, Seto H (1987) A method for pure absorption 2D spectroscopy using complex fourier transformation in both dimensions. J Magn Reson 72:279–297
Pagès G, Kuchel PW (2013) Mathematical modeling and data analysis of NMR experiments using hyperpolarized 13C metabolites. Magn Reson Insights 6:13–21
Perrin CL (1989) Optimum mixing time for chemical kinetics by 2D NMR. J Magn Reson 82:619–621
Perrin CL, Dwyer TJ (1990) Application of two-dimensional NMR to kinetics of chemical exchange. Chem Rev 90:935–967
Perrin CL, Engler RE (1990) Weighted linear-least-squares analysis of EXSY data from multiple 1D selective inversion experiments. J Magn Reson 90:363–369
Perrin CL, Gipe RK (1984) Multisite kinetics by quantitative two-dimensional NMR. J Am Chem Soc 106:4036–4038
Potts JR, Kuchel PW (1992) Anomeric preference of fluoroglucose exchange across human red-cell membranes: 19F-NMR studies. Biochem J 281:753–759
Potts JR, Kirk K, Kuchel PW (1989) Characterization of the transport of the nonelectrolyte dimethyl methylphosphonate across the red cell membrane. NMR Biomed 1:198–204
Potts JR, Hounslow AM, Kuchel PW (1990) Exchange of fluorinated glucose across the red-cell membrane measured by 19F-NMR magnetization transfer. Biochem J 266:925–928
Price WS, Kuchel PW (1990) Hypophosphite transport in human erythrocytes studied by overdetermined one-dimensional NMR exchange analysis. NMR Biomed 3:59–63
Puckeridge M, Chapman BE, Conigrave AD, Kuchel PW (2012) Quantitative model of NMR chemical shifts of 23Na+ induced by TmDOTP: applications in studies of Na+ transport in human erythrocytes. J Inorg Biochem 115:211–219
Raftos JE, Bulliman BT, Kuchel PW (1990) Evaluation of an electrochemical model of erythrocyte pH buffering using 31P nuclear magnetic resonance data. J Gen Physiol 95:1183–1204
Ripoche P, Bertrand O, Gane P, Birkenmeier C, Colin Y, Cartron J-P (2004) Human Rhesus-associated glycoprotein mediates facilitated transport of NH3 into red blood cells. Proc Natl Acad Sci USA 101:17222–17227
Robinson G, Kuchel PW, Chapman BE, Doddrell DM, Irving MG (1985) A simple procedure for selective inversion of NMR resonances for spin transfer enzyme kinetic measurements. J Magn Reson 63:314–319
Rumsey SC, Kwon O, Xu GW, Burant CF, Simpson I, Levine M (1997) Glucose transporter isoforms GLUT1 and GLUT3 transport dehydroascorbic acid. J Biol Chem 272:18982–18989
Savitz D, Sidel VW, Solomon AK (1964) Osmotic properties of human red cells. J Gen Physiol 48:79–94
States DJ, Haberkorn RA, Ruben DJ (1982) A two-dimensional nuclear Overhauser experiment with pure absorption phase in four quadrants. J Magn Reson 48:286–292
Stein WD (1986) Transport and diffusion across cell membranes. Academic, Orlando
Stein WD, Litman T (2015) Channels, carriers, and pumps: an introduction to membrane transport, 2nd edn. Academic, San Diego
Stewart IM, Chapman BE, Kirk K, Kuchel PW, Lovric VA, Raftos JE (1986) Intracellular pH in stored erythrocytes. Refinement and further characterisation of the 31P-NMR methylphosphonate procedure. BBA-Mol Cell Res 885:23–33
Szekely D, Chapman BE, Bubb WA, Kuchel PW (2006) Rapid exchange of fluoroethylamine via the Rhesus complex in human erythrocytes: 19F NMR magnetization transfer analysis showing competition by ammonia and ammonia analogues. Biochemistry 45:9354–9361
Wittenkeller L, Mota de Freitas D, Geraldes CFGC, Tomé AJR (1992) Physical basis for the resolution of intra- and extracellular 133Cs NMR resonances in Cs+-loaded human erythrocyte suspensions in the presence and absence of shift reagents. Inorg Chem 31:1135–1144
Wolfram S (2003) The Mathematica book, 5th edn. Wolfram Research, Champaign
Xu ASL, Potts JR, Kuchel PW (1991) The phenomenon of separate intra- and extracellular resonances of difluorophosphate in 31P and 19F NMR spectra of erythrocytes. Magn Reson Med 18:193–198
Acknowledgments
This work was funded by the Australian Research Council Discovery Project grant DP140102596 to PWK and the Leverhulme Trust grant RPG-2015-211 to PWK and Professor Bruno Linclau, who is thanked for critical reading of the draft of this manuscript. We convey our best wishes to Professor Don Winzor on his 80th birthday in 2016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human erythrocytes were in accordance with the ethical standards of the University of Sydney Human Ethics Committee. Informed consent was obtained from all individual participants included in the study.
Additional information
This article is part of a Special Issue on ‘Analytical Quantitative Relations in Biochemistry’ edited by Damien Hall and Stephen Harding.
Rights and permissions
About this article
Cite this article
Shishmarev, D., Kuchel, P.W. NMR magnetization-transfer analysis of rapid membrane transport in human erythrocytes. Biophys Rev 8, 369–384 (2016). https://doi.org/10.1007/s12551-016-0221-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-016-0221-y