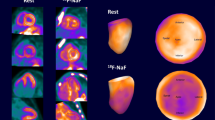
Abstract
Background
18F-florbetapir PET is emerging as an excellent quantitative tool to quantify cardiac light chain (AL) amyloidosis burden. The primary aim of this study was to determine interobserver reproducibility and intraobserver repeatability, defined per the recommendations of the Quantitative Imaging Biomarker Alliance technical performance group, of PET 18F-florbetapir retention index (RI) in patients with cardiac AL amyloidosis.
Methods
The study cohort comprised 37 subjects with systemic AL amyloidosis enrolled in the prospective study: Molecular Imaging of Primary Amyloid Cardiomyopathy (clinical trials.gov NCT: 02641145). Using 10 mCi of 18F-florbetapir, a 60-minute dynamic cardiac scan was acquired. Global and segmental left ventricular estimates of retention index (RI) of 18F-florbetapir were calculated (Carimas 2.9 software, Turku, Finland). RI was analyzed twice, at least 24 hours apart, by two independent observers. Intraobserver repeatability and interobserver reproducibility were evaluated using Bland–Altman plots and scatter plots with fitted linear regression curves.
Results
All reproducibility (interobserver, r = 0.98) and repeatability (intraobserver, R=0.99 for each observer) measures of 18F-florbetapir RI are excellent. On the Bland–Altman plots, the agreement limits for global 18F-florbetapir RI were high and ranged for reproducibility (interobserver) from − 9.3 to + 9.4% (Fig. 1), and for repeatability (observer 1 from − 10.8 to + 10.7% and from − 9.2 to + 11.4%, for observer 2).
Conclusions
The present study showed excellent interobserver reproducibility and intraobserver repeatability of 18F-florbetapir PET retention index in patients with cardiac AL amyloidosis.





Similar content being viewed by others
Abbreviations
- AL:
-
Light chain
- AL-CMP:
-
Light chain amyloidosis with cardiomyopathy
- PET:
-
Positron imaging tomography
- RI:
-
Retention index
- LV:
-
Left ventricle
- NT-ProBNP:
-
N-terminal pro b-type natriuretic peptide
- CTnT:
-
Cardiac troponin T
- eGFR:
-
Estimated glomerular filtration rate
- MICA:
-
Molecular Imaging of Primary Amyloid Cardiomyopathy
References
Falk RH, Alexander KM, Liao R, Dorbala S. AL (light-chain) cardiac amyloidosis: A review of diagnosis and therapy. J Am Coll Cardiol. 2016;68:1323–41.
Ruberg FL, Berk JL. Transthyretin (TTR) cardiac amyloidosis. Circulation. 2012;126:1286–300.
Richards DB, Cookson LM, Berges AC, Barton SV, Lane T, Ritter JM, et al. Therapeutic clearance of amyloid by antibodies to serum amyloid P component. N Engl J Med. 2015;373:1106–14.
Gertz MA, Landau H, Comenzo RL, Seldin D, Weiss B, Zonder J, et al. First-in-human phase I/II study of NEOD001 in patients with light chain amyloidosis and persistent organ dysfunction. J Clin Oncol. 2016;34:1097–103.
Dorbala S, Kijewski MF, Park MA. Quantitative molecular imaging of cardiac amyloidosis: The journey has begun. J Nucl Cardiol. 2016;23:751–3.
Park MA, Padera RF, Belanger A, Dubey S, Hwang DH, Veeranna V, et al. 18F-florbetapir binds specifically to myocardial light chain and transthyretin amyloid deposits: Autoradiography study. Circ Cardiovasc Imaging. 2015;8:e002954.
Dorbala S. Imaging cardiac amyloidosis: An opportunity for nuclear cardiology. J Nucl Cardiol. 2014;21:1043–4.
Raunig DL, Mcshane LM, Pennello G, Gatsonis C, Carson PL, Voyvodic JT, et al. Quantitative imaging biomarkers: A review of statistical methods for technical performance assessment. Stat Methods Med Res. 2014;24:27–67.
Dorbala S, Vangala D, Semer J, Strader C, Bruyere JR Jr, Di Carli MF, et al. Imaging cardiac amyloidosis: A pilot study using (18)F-florbetapir positron emission tomography. Eur J Nucl Med Mol Imaging. 2014;41:1652–62.
Nesterov SV, Turta O, Han C, Maki M, Lisinen I, Tuunanen H, et al. C-11 acetate has excellent reproducibility for quantification of myocardial oxidative metabolism. Eur Heart J Cardiovasc Imaging. 2015;16:500–6.
Kero T, Lindsjo L, Sorensen J, Lubberink M. Accurate analysis and visualization of cardiac (11)C-PIB uptake in amyloidosis with semiautomatic software. J Nucl Cardiol. 2016;23:741–50.
Disclosure
RHF has received consulting fees from Ionis Pharmaceuticals and Alnylam Pharmaceuticals and research funding from GlaxoSmithKline. SD serves as a consultant for Proclara, Pfizer, AAA, and GEHC and has received research funds from Pfizer. The other authors do not have any conflicts to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarizes the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
The authors have also provided an audio summary of the article, which is available to download as ESM, or to listen to via the JNC/ASNC Podcast.
Funding
SD and RF are supported by NIH RO1 Grant (RO1 HL 130563); SD is supported by American Heart Association Grant (AHA 16 CSA 2888 0004).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nodoushani, A., El-Sady, M.S., Park, MA. et al. Reproducibility and Repeatability of Assessment of Myocardial Light Chain Amyloidosis Burden Using 18F-Florbetapir PET/CT. J. Nucl. Cardiol. 28, 2004–2010 (2021). https://doi.org/10.1007/s12350-019-01961-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-019-01961-z