Abstract
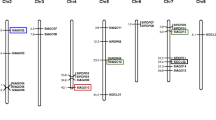
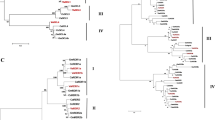
Key components of the RNA interference (RNAi) pathway include the Dicer-like (DCL), Argonaute (AGO), and RNA-dependent RNA polymerase (RDR) gene families. While these components have been studied in various plant species, their functional validation in wheat remains unexplored particularly under heat stress. In this study, a comprehensive genome-wide analysis to identify, and characterize DCL, AGO, and RDR genes in wheat and their expression patterns was carried out. Using phylogenetic analysis with orthologous genes from Arabidopsis and rice, we identified a total of 82 AGO, 31 DCL, and 31 RDR genes distributed across the 21 chromosomes of wheat. To understand the regulatory network, a network analysis of miRNAs that target RNA-silencing genes was performed. Our analysis revealed that 13 miRNAs target AGO genes, 8 miRNAs target DCL genes, and 10 miRNAs target RDR genes at different sites, respectively. Additionally, promoter analysis of the RNA-silencing genes was done and identified the presence of 132 cis-elements responsive to stress and phytohormones. To examine their expression patterns, we performed RNA-seq analysis in the flag leaf samples of wheat exposed to both normal and heat stress conditions. To understand the regulation of RNA silencing, we experimentally analysed the transcriptional changes in response to gradient heat stress treatments. Our results showed constitutive expression of the AGO1, AGO9, and DCL2 gene families, indicating their importance in the overall biological processes of wheat. Notably, RDR1, known to be involved in small interfering RNA (siRNA) biogenesis, exhibited higher expression levels in wheat leaf tissues. These findings suggest that these genes may play a role in responses to stress in wheat, highlighting their significance in adapting to environmental challenges. Overall, our study provides additional knowledge to understand the mechanisms underlying heat stress responses and emphasizes the essential roles of these gene families in wheat.







Similar content being viewed by others
References
Ahmed FF, Hossen MI, Sarkar MAR, et al (2021) Genome-wide identification of DCL, AGO and RDR gene families and their associated functional regulatory elements analyses in banana (Musa acuminata)
Akond Z, Rahman H, Ahsan MA et al (2022) Comprehensive in silico analysis of RNA silencing-related genes and their regulatory elements in wheat (Triticum aestivum L.). Biomed Res Int. https://doi.org/10.1155/2022/4955209
Alleman M, Sidorenko L, McGinnis K et al (2006) An RNA-dependent RNA polymerase is required for paramutation in maize. Nature 442:295–298. https://doi.org/10.1038/nature04884
Bai M, Yang GS, Chen WT et al (2012) Genome-wide identification of Dicer-like, Argonaute and RNA-dependent RNA polymerase gene families and their expression analyses in response to viral infection and abiotic stresses in Solanum lycopersicum. Gene 501:52–62. https://doi.org/10.1016/j.gene.2012.02.009
Bailey TL, Johnson J, Grant CE, Noble WS (2015) The MEME Suite. Nucleic Acids Res 43:W39–W49. https://doi.org/10.1093/nar/gkv416
Bologna NG, Voinnet O (2014) The diversity, biogenesis, and activities of endogenous silencing small RNAs in Arabidopsis. Annu Rev Plant Biol 65:473–503. https://doi.org/10.1146/annurev-arplant-050213-035728
Bordoloi KS, Agarwala N (2021) MicroRNAs in plant insect interaction and insect pest control. Plant Gene 26:100271. https://doi.org/10.1016/j.plgene.2021.100271
Branscheid A, Marchais A, Schott G et al (2015) SKI2 mediates degradation of RISC 5′-cleavage fragments and prevents secondary siRNA production from miRNA targets in Arabidopsis. Nucleic Acids Res 43:10975–10988. https://doi.org/10.1093/nar/gkv1014
Cannon SB, Mitra A, Baumgarten A et al (2004) The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. BMC Plant Biol 4:1–21. https://doi.org/10.1186/1471-2229-4-10
Cao JY, Xu YP, Li W et al (2016) Genome-wide identification of dicer-like, argonaute, and RNA-dependent RNA polymerase gene families in brassica species and functional analyses of their arabidopsis homologs in resistance to sclerotinia sclerotiorum. Front Plant Sci 7:1–17. https://doi.org/10.3389/fpls.2016.01614
Carbonell A, Carrington JC (2015) Antiviral roles of plant ARGONAUTES. Curr Opin Plant Biol 27:111–117. https://doi.org/10.1016/j.pbi.2015.06.013
Chávez-Hernández EC, Alejandri-Ramírez ND, Juárez-González VT, Dinkova TD (2015) Maize miRNA and target regulation in response to hormone depletion and light exposure during somatic embryogenesis. Front Plant Sci 6:1–14. https://doi.org/10.3389/fpls.2015.00555
Chen C, Chen H, Zhang Y et al (2020) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13:1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Chung BYW, Valli A, Deery MJ et al (2019) Distinct roles of Argonaute in the green alga Chlamydomonas reveal evolutionary conserved mode of miRNA-mediated gene expression. Sci Rep 9:11091. https://doi.org/10.1038/s41598-019-47415-x
Cui DL, Meng JY, Ren XY et al (2020) Genome-wide identification and characterization of DCL, AGO and RDR gene families in Saccharum spontaneum. Sci Rep 10:1–17. https://doi.org/10.1038/s41598-020-70061-7
Douglas RN, Wiley D, Sarkar A et al (2010) Ragged seedling2 encodes an ARGONAUTE7-like protein required for mediolateral expansion, but not dorsiventrality, of maize leaves. Plant Cell 22:1441–1451. https://doi.org/10.1105/tpc.109.071613
Duan C, Zhang H, Tang K et al (2015) Specific but interdependent functions for Arabidopsis AGO 4 and AGO 6 in RNA-directed DNA methylation. EMBO J 34:581–592. https://doi.org/10.15252/embj.201489453
Durán-Figueroa N, Vielle-Calzada JP (2010) ARGONAUTE9-dependent silencing of transposable elements in pericentromeric regions of Arabidopsis. Plant Signal Behav 5:1476–1479. https://doi.org/10.4161/psb.5.11.13548
Fang X, Qi Y (2015) RNAi in plants: an argonaute-centered view. Plant Cell 28:272–285. https://doi.org/10.1105/tpc.15.00920
FAO (2022) Prospects and Situation: Crop Prospects and Food Situation #2, July 2022
Finn RD, Coggill P, Eberhardt RY et al (2016) The Pfam protein families database: towards a more sustainable future. Nucleic Acids Res 44:D279–D285. https://doi.org/10.1093/nar/gkv1344
Goodstein DM, Shu S, Howson R et al (2012) Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res 40:1178–1186. https://doi.org/10.1093/nar/gkr944
Gualtieri C, Leonetti P, Macovei A (2020) Plant miRNA Cross-kingdom transfer targeting parasitic and mutualistic organisms as a tool to advance modern agriculture. Front Plant Sci 11:1–7. https://doi.org/10.3389/fpls.2020.00930
Gyula P, Baksa I, Tóth T et al (2018) Ambient temperature regulates the expression of a small set of sRNAs influencing plant development through NF-YA2 and YUC2. Plant Cell Environ 41:2404–2417. https://doi.org/10.1111/pce.13355
Haag JR, Ream TS, Marasco M et al (2012) In vitro transcription activities of Pol IV, Pol V, and RDR2 Reveal Coupling of Pol IV and RDR2 for dsRNA Synthesis in Plant RNA Silencing. Mol Cell 48:811–818. https://doi.org/10.1016/j.molcel.2012.09.027
Hamar E, Szaker HM, Kis A et al (2020) Genome-wide identification of RNA silencing-related genes and their expressional analysis in response to heat stress in barley (Hordeum vulgare l.). Biomolecules 10:1–26. https://doi.org/10.3390/biom10060929
Harvey JJW, Lewsey MG, Patel K et al (2011) An antiviral defense role of AGO2 in plants. PLoS ONE 6:1–6. https://doi.org/10.1371/journal.pone.0014639
Havecker ER, Wallbridge LM, Hardcastle TJ et al (2010) The arabidopsis RNA-directed DNA methylation argonautes functionally diverge based on their expression and interaction with target loci. Plant Cell 22:321–334. https://doi.org/10.1105/tpc.109.072199
Holoch D, Moazed D (2015) RNA-mediated epigenetic regulation of gene expression. Nat Rev Genet 16:71–84. https://doi.org/10.1038/nrg3863
Hu B, Jin J, Guo AY et al (2015) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296–1297. https://doi.org/10.1093/bioinformatics/btu817
Iki T (2017) Messages on small RNA duplexes in plants. J Plant Res 130:7–16. https://doi.org/10.1007/s10265-016-0876-2
Graham Ruby J, Jan CH, Bartel DP (2010) Intronic microRNA precursors that bypass Drosha processing. Nature 136:642–655. https://doi.org/10.1016/j.cell.2009.01.035.Origins
Jagtap S, Shivaprasad PV (2014) Diversity, expression and mRNA targeting abilities of Argonaute-targeting miRNAs among selected vascular plants. BMC Genomics 15:1–14. https://doi.org/10.1186/1471-2164-15-1049
Kakar KU, Nawaz Z, Cui Z et al (2018) Evolutionary and expression analysis of CAMTA gene family in Nicotiana tabacum yielded insights into their origin, expansion and stress responses. Sci Rep 8:1–14. https://doi.org/10.1038/s41598-018-28148-9
Kasschau KD, Fahlgren N, Chapman EJ et al (2007) Genome-wide profiling and analysis of Arabidopsis siRNAs. PLoS Biol 5:0479–0493. https://doi.org/10.1371/journal.pbio.0050057
Kochetov AV, Ischenko IV, Vorobiev DG et al (1998) Eukaryotic mRNAs encoding abundant and scarce proteins are statistically dissimilar in many structural features. FEBS Lett 440:351–355. https://doi.org/10.1016/S0014-5793(98)01482-3
Krishnatreya DB, Baruah PM, Dowarah B et al (2021) Genome-wide identification, evolutionary relationship and expression analysis of AGO, DCL and RDR family genes in tea. Sci Rep 11:1–18. https://doi.org/10.1038/s41598-021-87991-5
Kruszka K, Pacak A, Swida-Barteczka A et al (2014) Transcriptionally and post-transcriptionally regulated microRNAs in heat stress response in barley. J Exp Bot 65:6123–6135. https://doi.org/10.1093/jxb/eru353
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Larkin MA, Blackshields G, Brown NP et al (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948. https://doi.org/10.1093/bioinformatics/btm404
Li S, Liu J, Liu Z et al (2014) HEAT-INDUCED TAS1 TARGET1 mediates thermotolerance via heat stress transcription factor A1a-directed pathways in arabidopsis. Plant Cell 26:1764–1780. https://doi.org/10.1105/tpc.114.124883
Lin JS, Kuo CC, Yang IC et al (2018) MicroRNA160 modulates plant development and heat shock protein gene expression to mediate heat tolerance in Arabidopsis. Front Plant Sci 9:1–16. https://doi.org/10.3389/fpls.2018.00068
Liu SR, Zhou JJ, Hu CG et al (2017) MicroRNA-mediated gene silencing in plant defense and viral counter-defense. Front Microbiol 8:1–12. https://doi.org/10.3389/fmicb.2017.01801
Liu YF, Wang LM, Zhao LZ et al (2021) Genome-wide identification and evolutionary analysis of argonaute genes in hexaploid bread wheat. Biomed Res Int. https://doi.org/10.1155/2021/9983858
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods. https://doi.org/10.1006/meth.2001.1262
Madsen CT, Stephens J, Hornyik C et al (2009) Identification and characterization of barley RNA-directed RNA polymerases. Biochim Biophys Acta Gene Regul Mech 1789:375–385. https://doi.org/10.1016/j.bbagrm.2009.03.003
Mallory A, Vaucheret H (2010) Form, function, and regulation of ARGONAUTE proteins. Plant Cell 22:3879–3889. https://doi.org/10.1105/tpc.110.080671
Marchler-Bauer A, Lu S, Anderson JB et al (2011) CDD: A Conserved Domain Database for the functional annotation of proteins. Nucleic Acids Res 39:225–229. https://doi.org/10.1093/nar/gkq1189
Margis R, Fusaro AF, Smith NA et al (2006) The evolution and diversification of Dicers in plants. FEBS Lett 580:2442–2450. https://doi.org/10.1016/j.febslet.2006.03.072
Metsalu T, Vilo J (2015) ClustVis: a web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic Acids Res 43:W566–W570. https://doi.org/10.1093/nar/gkv468
Montgomery TA, Howell MD, Cuperus JT et al (2008) Specificity of ARGONAUTE7-miR390 Interaction and dual functionality in TAS3 trans-acting siRNA formation. Cell 133:128–141. https://doi.org/10.1016/j.cell.2008.02.033
Muthusamy SK, Dalal M, Chinnusamy V, Bansal KC (2016) Differential regulation of genes coding for organelle and cytosolic ClpATPases under biotic and abiotic stresses in Wheat. Front Plant Sci. https://doi.org/10.3389/fpls.2016.00929
Nagasaki H, Itoh JI, Hayashi K et al (2007) The small interfering RNA production pathway is required for shoot meristem initiation in rice. Proc Natl Acad Sci 104:14867–14871. https://doi.org/10.1073/pnas.0704339104
Nakasugi K, Crowhurst RN, Bally J et al (2013) De Novo Transcriptome sequence assembly and analysis of rna silencing genes of Nicotiana benthamiana. PLoS ONE. https://doi.org/10.1371/journal.pone.0059534
Ozhuner E, Eldem V, Ipek A et al (2013) Boron stress responsive micrornas and their targets in barley. PLoS ONE. https://doi.org/10.1371/journal.pone.0059543
Parker JS, Roe SM, Barford D (2010) Europe PMC funders group structural insights into mRNA recognition from a PIWI domain—siRNA guide complex. Nature 434:663–666. https://doi.org/10.1038/nature03462.Structural
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D et al (1971) Cytoscape: a software environment for integrated models. Genome Res 13:426. https://doi.org/10.1101/gr.1239303.metabolite
Potter SC, Luciani A, Eddy SR et al (2018) HMMER web server: 2018 update. Nucleic Acids Res 46:W200–W204. https://doi.org/10.1093/nar/gky448
Pratt AJ, MacRae IJ (2009) The RNA-induced silencing complex: a versatile gene-silencing machine. J Biol Chem 284:17897–17901. https://doi.org/10.1074/jbc.R900012200
Ronemus M, Vaughn MW, Martienssen RA (2006) MicroRNA-targeted and small interfering RNA-mediated mRNA degradation is regulated by argonaute, dicer, and RNA-dependent RNA polymerase in Arabidopsis. Plant Cell 18:1559–1574. https://doi.org/10.1105/tpc.106.042127
Sabbione A, Daurelio L, Vegetti A et al (2019) Genome-wide analysis of AGO, DCL and RDR gene families reveals RNA-directed DNA methylation is involved in fruit abscission in Citrus sinensis. BMC Plant Biol 19:1–13. https://doi.org/10.1186/s12870-019-1998-1
Sharma P, Mehta G, Shefali et al (2021) Development and validation of heat-responsive candidate gene and miRNA gene based SSR markers to analysis genetic diversity in wheat for heat tolerance breeding. Mol Biol Rep 48:381–393. https://doi.org/10.1007/s11033-020-06059-1
Shen D, Wang S, Chen H et al (2009) Molecular phylogeny of miR390-guided trans-acting siRNA genes (TAS3) in the grass family. Plant Syst Evol 283:125–132. https://doi.org/10.1007/s00606-009-0221-5
Singh M, Goel S, Meeley RB et al (2011) Production of viable gametes without meiosis in maize deficient for an ARGONAUTE protein. Plant Cell 23:443–458. https://doi.org/10.1105/tpc.110.079020
Song JJ, Smith SK, Hannon GJ, Joshua-Tor L (2004) Crystal structure of argonaute and its implications for RISC slicer activity. Science (80-) 305:1434–1437. https://doi.org/10.1126/science.1102514
Song X, Li P, Zhai J et al (2012) Roles of DCL4 and DCL3b in rice phased small RNA biogenesis. Plant J 69:462–474. https://doi.org/10.1111/j.1365-313X.2011.04805.x
Srivastava AK, Lu Y, Zinta G et al (2018) UTR-dependent control of gene expression in plants. Trends Plant Sci 23:248–259. https://doi.org/10.1016/j.tplants.2017.11.003
Stief A, Altmann S, Hoffmann K et al (2014) Arabidopsis miR156 regulates tolerance to recurring environmental stress through SPL transcription factors. Plant Cell 26:1792–1807. https://doi.org/10.1105/tpc.114.123851
Szaker HM, Gyula P, Szittya G, Csorba T (2020) Regulation of high-temperature stress response by small RNAs. Springer International Publishing, Berlin
Szklarczyk D, Franceschini A, Wyder S et al (2015) STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43:D447–D452. https://doi.org/10.1093/nar/gku1003
Teng C, Zhang H, Hammond R et al (2020) Dicer-like 5 deficiency confers temperature-sensitive male sterility in maize. Nat Commun 11:1–32. https://doi.org/10.1038/s41467-020-16634-6
Várallyay É, Válóczi A, Ágyi Á et al (2010) Plant virus-mediated induction of miR168 is associated with repression of ARGONAUTE1 accumulation. EMBO J 29:3507–3519. https://doi.org/10.1038/emboj.2010.215
Vaucheret H (2008) Plant ARGONAUTES. Trends Plant Sci 13:350–358. https://doi.org/10.1016/j.tplants.2008.04.007
Vaucheret H, Mallory AC, Bartel DP (2006) AGO1 Homeostasis Entails Coexpression of MIR168 and AGO1 and Preferential Stabilization of miR168 by AGO1. Mol Cell 22:129–136. https://doi.org/10.1016/j.molcel.2006.03.011
Vaucheret H, Vazquez F, Crété P, Bartel DP (2004) The action of ARGONAUTE1 in the miRNA pathway and its regulation by the miRNA pathway are crucial for plant development. Genes Dev 18:1187–1197. https://doi.org/10.1101/gad.1201404
Voorrips RE (2002) Mapchart: software for the graphical presentation of linkage maps and QTLs. J Hered 93:77–78. https://doi.org/10.1093/jhered/93.1.77
Wang H, Jiao X, Kong X et al (2016) A signaling cascade from miR444 to RDR1 in rice antiviral RNA silencing pathway. Plant Physiol 170:2365–2377. https://doi.org/10.1104/pp.15.01283
Wu J, Yang Z, Wang Y et al (2015) Viral-inducible Argonaute18 confers broad-spectrum virus resistance in rice by sequestering a host microRNA. Elife 2015:1–19. https://doi.org/10.7554/eLife.05733
Xu D, Yang H, Zou C et al (2016) Identification and functional characterization of the AGO1 ortholog in maize. J Integr Plant Biol 58:749–758. https://doi.org/10.1111/jipb.12467
Yan KS, Yan S, Farooq A et al (2003) Structure and conserved RNA binding of the PAZ domain. Nature 426:469–474. https://doi.org/10.1038/nature02129
Yu X, Wang H, Lu Y et al (2012) Identification of conserved and novel microRNAs that are responsive to heat stress in Brassica rapa. J Exp Bot 63:1025–1038. https://doi.org/10.1093/jxb/err337
Zhai J, Zhang H, Arikit S et al (2015) Spatiotemporally dynamic, cell-type-dependent premeiotic and meiotic phasiRNAs in maize anthers. Proc Natl Acad Sci U S A 112:3146–3151. https://doi.org/10.1073/pnas.1418918112
Zhonga SH, Liu JZ, Jin H et al (2013) Warm temperatures induce transgenerational epigenetic release of RNA silencing by inhibiting siRNA biogenesis in Arabidopsis. Proc Natl Acad Sci U S A 110:9171–9176. https://doi.org/10.1073/pnas.1219655110
Acknowledgements
The project work was supported by the grants from Indian Council of Agricultural Research, New Delhi. The authors acknowledge to Germplasm Resource Unit of ICAR-IIWBR, Karnal for supply of seed material. This is IIWBR paper contribution no. 179/430.
Funding
This work was supported by a grant from the ICAR-Core Research projects Grant-in-Aids to PS.
Author information
Authors and Affiliations
Contributions
PS conceived the idea and designed the experiments. SM and PS performed the experiments, analyse the data. SM, PS drafted the manuscript. PS, RS, OPA, GS edited the manuscript. All the authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mishra, S., Sharma, P., Singh, R. et al. Genome-wide identification of DCL, AGO, and RDR gene families in wheat (Triticum aestivum L.) and their expression analysis in response to heat stress. Physiol Mol Biol Plants 29, 1525–1541 (2023). https://doi.org/10.1007/s12298-023-01362-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-023-01362-0