Abstract
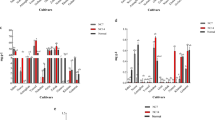
To curb the increasing demand for nitrogenous fertilizers, it is imperative to develop new cultivars with comparatively greater nitrogen use efficiency (NUE). Nonetheless, so far very meager information is available concerning the variances among barley (Hordeum vulgare L.) varieties for their response to nitrogen deprivation. The current study was carried out to explore the potential of barley genotypes for higher NUE. A hydroponic experiment was conducted at seedling stage to compare the performance of four barley genotypes, ZD9 and XZ149 (with higher NUE) and HXRL and XZ56 (with lower NUE) in response to low (0.1 mM) and normal nitrogen (2 mM) levels. Under low N, all the genotypes expressed less number of tillers, decreased soluble proteins, chlorophyll and N concentrations in both roots and shoots, in comparison with normal N supply. However, significant differences were found among the genotypes. The genotypes with high NUE (ZD9 and XZ149) showed higher N concentration, increased number of tillers, improved chlorophyll and soluble proteins in both roots and shoots as compared to the inefficient ones (HXRL and XZ56). Furthermore, nitrate transporter gene (NRT2.1) showed higher expression under low N, both in roots and leaves of N efficient genotypes, as compared to the N inefficient ones. However, N assimilatory genes (GS1 and GS2) showed higher expression under normal and low N level, in leaves and roots respectively. The outcome of the study revealed that genotypes with higher NUE (ZD9 and XZ149) performed better under reduced N supply, and may require relatively less N fertilizer for normal growth and development, as compared to those with lower NUE. The study also revealed a time-specific expression pattern of studied genes, indicating the duration of low N stress. The current study suggested that future work must involve the time course as a key factor while studying expression patterns of these genes to better understand the genetic basis of low-N tolerance.




Similar content being viewed by others
References
Abdel-Ghani AH, Kumar B, Reyes-Matamoros J, Gonzalez-Portilla PJ, Jansen C, Martin JPS, Lee M, Lübberstedt T (2013) Genotypic variation and relationships between seedling and adult plant traits in maize (Zea mays L.) inbred lines grown under contrasting nitrogen levels. Euphytica 189:123–133
Anbessa Y, Juskiw P, Good A, Nyachiro J, Helm J (2009) Genetic variability in nitrogen use efficiency of spring barley. Crop Sci 49:1259–1269
Bernard SM, Moller AL, Dionisio G, Kichey T, Jahn TP, Dubois F, Baudo M, Lopes MS, Tercé-Laforgue T, Foyer CH, Parry MA, Forde BG, Araus JL, Hirel B, Schjoerring JK, Habash DZ (2008) Gene expression, cellular localisation and function of glutamine synthetase isozymes in wheat (Triticum aestivumL.). Plant Mol Biol 67:89–105
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Bukhari SAH, Wang R, Wang W, Ahmed IA, Zheng W, Cao F (2016) Genotype-dependent effect of exogenous 24-epibrassinolide on chromium-induced changes in ultrastructure and physicochemical traits in tobacco seedlings. Environ Sci Pollut Res 23:18229–18238
Crawford NM, Forde BG (2002) Molecular and developmental biology of inorganic nitrogen nutrition. In: Meyerowitz E, Somerville C (eds) The arabidopsis book. American Soci Plant Biologists, Rockville. https://doi.org/10.1199/tab.0011
Crawford NM, Glass ADM (1998) Molecular and physiological aspects of nitrate uptake in plants. Trends Plant Sci 3:389–395
Dai F, Nevo E, Wu D, Comadran J, Zhou M, Qiu L, Chen Z, Beiles A, Chen G, Zhang G (2012) Tibet is one of the centers of domestication of cultivated barley. Proc Natl Acad Sci USA 109:16969–16973
Dawson JC, Huggins DR, Jones SS (2008) Characterizing nitrogen use efficiency to improve crop performance in organic and sustainable agricultural systems. Field Crops Res 107:89–101
Edwards JW, Walker EL, Coruzzi GM (1990) Cell-specific expression in transgenic plants reveals nonoverlapping roles for chloroplast and cytosolic glutamine synthetase. Proc Natl Acad Sci USA 87:3459–3463
FAO (2018) World Food and Agriculture—statistical pocketbook 2018. Rome. pp 8. http://www.fao.org/economic/ess/ess-publications/ess-yearbook/en/#.XTZhXK_lYdU
Farida M, Ali S, Rizwan M, Ali Q, Abbas F, Bukhari SAH, Saeed R, Wu L (2017) Citric acid assisted phytoextraction of chromium by sunflower; morphophysiological and biochemical alterations in plants. Ecotoxicol Environ Saf 145:90–102
Filleur S, Daniel-Vedele F (1999) Expression analysis of a high-affinity nitrate transporter isolated from Arabidopsis thaliana by differential display. Planta 207:461–469
Galangau F, Danielvedele F, Moureaux T, Dorbe MF, Leydecker MT, Caboche M (1988) Expression of leaf nitrate reductase from tomato and tobacco in relation to light-dark regimes and nitrate supply. Plant Physiol 88:383–388
Garnett T, Conn V, Kaiser BN (2009) Root based approaches to improving nitrogen use efficiency in plants. Plant Cell Environ 32:1272–1283
Gleadow RM, Evans JR, McCaffery S, Cavagnaro TR (2009) Growth and nutritive value of cassava (Manihot esculenta Cranz) are reduced when grown in elevated CO2. Plant Biol 11:76–82
Goodall AJ, Kumar P, Tobin AK (2013) Identification and expression analyses of cytosolic glutamine synthetase genes in barley (Hordeum vulgare L.). Plant Cell Physiol 54:492–505
Hawkesford M, Horst W, Kichey T, Lambers H, Schjoerring J, Skrumsager Møller I, White P (2012) Functions of macronutrients. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, 3rd edn. Elsevier, Amsterdam, pp 135–189
Ikram S, Bedu M, Daniel-Vedele F, Chaillou S, Chardon F (2012) Natural variation of Arabidopsis response to nitrogen availability. J Exp Bot 63:91–105
Jones JB Jr (1991) Kjeldahl method for nitrogen (N) determination. Micro-Macro Publishing, Athens
Kant S, Bi YM, Rothstein SJ (2011) Understanding plant response to nitrogen limitation for the improvement of crop nitrogen use efficiency. J Exp Bot 62:1499–1509
Kessel B, Schierholt A, Becker HC (2012) Nitrogen use efficiency in a genetically diverse set of winter oilseed rape (Brassica napus L.). Crop Sci 52:2546–2554
Kiba T, Feria-Bourrellier AB, Lafouge F, Lezhneva L, Boutet-Mercey S, Orsel M, Bréhaut V, Miller A, Daniel-Vedele F, Sakakibara H, Krapp A (2012) The Arabidopsis nitrate transporter NRT2.4 plays a double role in roots and shoots of nitrogen starved plants. Plant Cell 24:245–258
Kraiser T, Gras DE, Gutiérrez AG, González B, Gutiérrez RA (2011) A holistic view of nitrogen acquisition in plants. J Exp Bot 62:1455–1466
Kronzucker HJ, Glass ADM, Siddiqi MY (1999) Inhibition of nitrate uptake by ammonium in barley. Analysis of component fluxes. Plant Physiol 120:283–291
Krouk G, Tillard P, Gojon A (2006) Regulation of the high-affinity NO3 uptake system by NRT1.1-mediated NO3 demand signaling in Arabidopsis. Plant Physiol 142:1075–1086
Le Gouis J, Beghin D, Heumez E, Pluchard P (2000) Genetic differences for nitrogen uptake and nitrogen utilization efficiency in winter wheat. Eur J Agron 12:163–173
Lea PJ, Azevedo RA (2007) Nitrogen use efficiency. 2. amino acid metabolism. Ann Appl Biol 151:269–275
Lewis OAM, James DM, Hewitt EJ (1982) Nitrogen assimilation in barley (Hordeum vulgare L Cv. Mazurka) in response to nitrate and ammonium nutrition. Ann Bot 49:39–49
Lezhneva L, Kiba T, Feria-Bourrellier AB, Lafouge F, Boutet-Mercey S, Orsel M, Bréhaut V, Miller A, Daniel-Vedele F, Sakakibara H, Krapp A (2014) The Arabidopsis nitrate transporter NRT2.5 plays a role in nitrate acquisition and remobilization in nitrogen-starved plants. Plant J 80:230–241
Li BZ, Xin WJ, Sun SB, Shen QR, Xu GH (2006) Physiological and molecular responses of nitrogen-starved rice plants to re-supply of different nitrogen sources. Plant Soil 287:145–159
Little DY, Rao H, Oliva S, Daniel-Vedele F, Krapp A, Malamy JE (2005) The putative high-affinity nitrate transporter NRT2.1 represses lateral root initiation in response to nutritional cues. Proc Natl Acad Sci USA 102:13693–13698
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative pcr and the 2−ΔΔCT method. Methods 25:402–408
Mack G (1995) Organ -specific changes in the activity and subunit composition of glutamine-synthetase isoforms of barley (Hordeum vulgare L) after growth on different levels of NH4. Planta 196:231–238
Mc Nally SF, Hirel B, Gadal P, Mann AF, Stewart GR (1983) Glutamine synthetases of higher plants evidence for a specific isoform content related to their possible physiological role and their compartmentation within the leaf. Plant Physiol 72:22–25
Muchow RC (1998) Nitrogen utilization efficiency in maize and grain sorghum. Field Crops Res 56:209–216
Munos S, Cazettes C, Fizames C, Gaymard F, Tillard P, Lepetit M, Lejay L, Gojon A (2004) Transcript profiling in the chl1-5 mutant of Arabidopsis reveals a role of the nitrate transporter NRT1.1 in the regulation of another nitrate transporter, NRT2.1. Plant Cell 16:2433–2447
Namai S, Toriyama K, Fukuta Y (2009) Genetic variations in dry matter production and physiological nitrogen use efficiency in rice (Oryza sativa L.) varieties. Breed Sci 59:269–276
Okamoto M, Vidmar JJ, Glass AD (2003) Regulation of NRT1 and NRT2 gene families of Arabidopsis thaliana: responses to nitrate provision. Plant Cell Physiol 44:304–317
Ortiz-Monasterio RJI, Sayre KD, Rajaram S, McMa-hon M (1997) Genetic progress in wheat yield and nitrogen use efficiency under four nitrogen rates. Crop Sci 37:898–904
Peat LJ, Tobin AK (1996) The effect of nitrogen nutrition on the cellular localization of glutamine synthetase isoforms in barley roots. Plant Physiol 111:1109–1117
Peng M, Hudson D, Schofield A, Tsao R, Yang R, Gu H, Bi Y-M, Rothstein SJ (2008) Adaptation of Arabidopsis to nitrogen limitation involves induction of anthocyanin synthesis which is controlled by the NLA gene. J Exp Bot 59(11):2933–2944
Presterl T, Seitz G, Landbeck M, Thiemt EM, Schmidt W, Geiger HH (2003) Improving nitrogen use efficiency in European maize: estimation of quantitative genetic parameters. Crop Sci 43:1259–1265
Quan X, Zeng J, Ye L, Chen G, Han Z, Shah JM, Zhang G (2016) Transcriptome profiling analysis for two Tibetan wild barley genotypes in responses to low nitrogen. BMC Plant Biol 16:30
Raven JA, Handley LL, Andrews M (2004) Global aspects of C/N interactions determining plant environment interactions. J Exp Bot 55:11–25
Robredo A, Pérez-López U, Miranda-Apodaca J, Lacuesta M, Mena-Petite A, Muñoz-Rueda A (2011) Elevated CO2 reduces the drought effect on nitrogen metabolism in barley plants during drought and subsequent recovery. Environ Exp Bot 71:399–408
Shah JM, Bukhari SAH, Jianbin Z, Xiaoyan Q, Ali E, Noor M, Zhang G (2017a) Nitrogen (N) metabolism related enzyme activities, cell ultrastructure and nutrient contents as affected by N level and barley genotype. J Integr Agric 16:190–198
Shah JM, Jianbin Z, Xiaoyan Q, Essa A, Imran HS, Zhang G (2017b) Growth and physiological characterization of low nitrogen responses in Tibetan wild and cultivated barleys. J Plant Nutr 40:861–868
Stitt M (1999) Nitrate regulation of metabolism and growth. Curr Opin Plant Biol 2:178–186
Tobin AK, Yamaya T (2001) Cellular compartmentation of ammonium assimilation in rice and barley. J Exp Bot 52:591–604
Wei D, Cui K, Ye G, Pan J, Xiang J, Huang J, Nie L, Estavillo JM (2012) QTL mapping for nitrogen use efficiency and nitrogen deficiency tolerance traits in rice. Plant Soil 359:281–295
Yang X, Wu J, Ziegler TE, Yang X, Zayed A, Rajani MS, Zhou D, Basra AS, Schachtman DP, Peng M, Armstrong CL, Caldo RA, Morrell JA, Lacy M, Staub JM (2011) Gene expression biomarkers provide sensitive indicators of in planta nitrogen status in maize. Plant Physiol 157:1841–1852
Zhao XQ, Shi WM (2006) Expression analysis of the glutamine synthetase and glutamate synthase gene families in young rice (Oryza sativa) seedlings. Plant Sci 170:748–754
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shah, J.M., Muntaha, S.t., Ali, E. et al. Comparative study of the genetic basis of nitrogen use efficiency in wild and cultivated barley. Physiol Mol Biol Plants 25, 1435–1444 (2019). https://doi.org/10.1007/s12298-019-00714-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-019-00714-z