Abstract
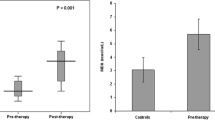
Conflicting data are available regarding oral iron therapy in iron deficiency (ID), iron deficiency anemia (IDA) and its relation to DNA damage, oxidative stress and antioxidant markers. Our aim was assessment of DNA damage, oxidative stress and anti-oxidant markers in children with ID and IDA before and after low dose iron therapy. The study was conducted in two stages, first stage was assessment of DNA damage using comet assay, malondialdehyde (MDA) and anti-oxidant enzymes levels (superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) & total antioxidant capacity (TAC) in thirty-nine children with IDA, forty-five children with ID without anemia and sixty healthy controls. Second stage was assessment of previous markers together with hematological response following oral therapy with 10 mg/day ferric ammonium citrate for 8 weeks. Before treatment, there was no significant difference between the three groups regarding MDA, GPx, SOD, CAT and TAC. A significant increase was detected in the DNA damage in the 2 groups compared to control (p < 0.005). Following iron therapy, hematological parameters was improved together with a significant increase in GPx (P = 0.04), SOD (p = 0.002), TAC (P = 0.001) and non-significant reduction in DNA damage in IDA group. There was a significant increase in SOD (p = 0.001) & TAC (p = 0.001) and significant decrease in DNA damage (p = 0.001) in ID group. Low dose iron therapy could be sufficient to improve antioxidant status and DNA damage together with correction of hematologic indices.

Similar content being viewed by others
Abbreviations
- ID:
-
Iron deficiency
- IDA:
-
Iron deficiency anemia
- BMI:
-
Body mass index
- CRP:
-
C Reactive Protein
- Retics:
-
Reticulocyte count
- TIBC:
-
Total Iron Binding Capacity
- MDA:
-
Malondialdehyde
- TBARS:
-
Thiobarbituric acid reactive substances
- GPx:
-
Glutathion Peroxidase
- SOD:
-
Superoxide Dismutase
- CAT:
-
Catalase
- TAC:
-
Total Antioxidant Capacity
- DMSO:
-
Dimethyl sulphoxide
- RPMI:
-
Roswell Park Memorial Institute
References
Zaka-Ur-Rab Z, Mohammad A, Najmul I (2016) Effect of oral iron on markers of oxidative stress and antioxidant status in children with iron deficiency anemia. J Clin Diagn Res 10(10):Sc13–Sc19
Stiban J, So M, Kaguni LS (2016) Iron-sulfur clusters in mitochondrial metabolism: multifaceted roles of a simple cofactor. Biochemistry 81:1066–1080. https://doi.org/10.1134/S0006297916100059
Amanda L, Jack T, Shohreh M, Morgan N, Greg T, Giuseppe V, Michael L (2018) Dysregulation of neuronal iron homeostasis as an alternative unifying effect of mutations causing familial Alzheimer’s disease. Front. Neurosci. https://doi.org/10.3389/fnins.2018.00533
Rose RC, Bode AM (1993) Biology of free radical scavengers: an evaluation of ascorbate. FASEB J 7(12):1135–1142
Ye H, Rouault TA (2010) Human iron-sulfur cluster assembly, cellular iron homeostasis and disease. Biochemistry 49(24):4945–4956
Joy GM, Nagababu E, Joseph M (2014) Rifkind Red blood cell oxidative stress impairs oxygen delivery and induces red blood cell aging. Front Physiol. 5:84
Nagababu E, Gulyani S, Earley CJ, Cutler RG, Mattson MP, Rifkind JM (2008) Iron-deficiency anaemia enhances red blood cell oxidative stress. Free Radic Res 42(9):824–829
Bagdagul A, Canan H, Solen H, Yildiz D, Eylem E, Sami H, Tulay A (2010) Leukocyte DNA damage in children with iron deficiency anemia: effect of iron supplementation. Eur J Pediatr 169:951–956
WHO (2001) Iron deficiency: assessment, prevention, and control. A guide for programme managers. Geneva, World Health Organization
El-Zanaty and Associates [Egypt], and ICF International (2015) Egypt demographic and health survey 2014. Ministry of Health and Population and ICF International, Cairo and Rockville
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometric assay of superoxide dismutase. Ind J Biochem Biophys 21:130–132
Sinha KA (1972) Colorimetric assay of catalase. Anal Biochem 47:389–394
Rotruck JJ, Pope AL, Ganther HE (1973) Selenium: biochemical rates as a component of glutathione peroxidase. Science 179:588–590
Erel O (2004) A novel automated method to measure total antioxidant response against potent free radical reactions. Clin Biochem 37:112–119
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Blasiak J, Gloe E, Wozniak K, Drzewoski J, Zadrozny M, Pertynski T (2003) Free radicals scavengers can differently modulate the genotoxicity of a msacrine in normal and cancer cells. Mutat Res 535:25–34
Moretti D, Goede JS, Zeder C, Jiskra M, Chatzinakou V, Tjalsma H et al (2015) Oral iron supplements increase hepcidin and decrease iron absorption from daily or twice-daily doses in iron-depleted young women. Blood 126(17):1981–1989 Medline
Schrier SL (2015) So you know how to treat iron deficiency anemia. Blood 126(17):1971 Medline
Aslan M, Horoz M, Kocyigit A, Ozgon¨ul S, Celik H, Celik M, Erel O (2006) Lymphocyte DNA damage and oxidative stress in patients with iron deficiency anemia. Mutat Res 601:144–149
Meral A, Tuncer P, Surmen-Gur E, Ozbek R, Oztürk E, Günay U (2000) Lipid peroxidation and antioxidant status in beta-thalassemia. Paediatr. Haematol Oncol. 17(8):687–693
Altun D, Kurekci AE, Gursel O, Hacıhamdioglu DO, Kurt I, Aydın A et al (2014) Malondialdehyde, antioxidant enzymes, and renal tubular functions in children with iron deficiency or iron-deficiency anaemia. Biol Trace Elem Res 161(1):48–56
Coghetto B, Lauerman L, Duarte M, Brandao V, Manfredini V, Peralba M, Silveira B (2009) Oxidative stress in older patients with iron deficiency anaemia. J Nutr Health Aging 13(8):666–670
Zhang Caiguo (2014) Essential functions of iron-requiring proteins in DNA replication, repair and cell cycle control. Protein Cell 5(10):750–760
Knutson MD, Walter PB, Ames BN, Viteri FE (2000) Both iron deficiency and daily iron supplements increase lipid peroxidation in rats. J Nutr 130:621–628
Yoo JH, Maeng HY, Sun YK, Kim YA, Park DW, Park TS et al (2009) Oxidative status in iron-deficiency anemia. J Clin Lab Anal 23(5):319–323
Gropper SS, Kerr S, Barksdale JM (2003) Non-anaemic iron deficiency, oral iron supplementation, and oxidative damage in college-aged females. J Nutr Biochem 14(7):409–415
Aycicek A, Koc A, Oymak Y, Selek S, Kaya C, Guzel B (2014) Ferrous sulfate (Fe2 +) had a faster effect than did ferric polymaltose (Fe3 +) on increased oxidant status in children with iron-deficiency anaemia. J Paediatr Haematol Oncol. 36(1):57–61
Mustapha UI, Shenshen Z, Jifei M, Hao W, Fudi W (2017) Antioxidants mediate both iron homeostasis and oxidative stress. Nutrients 9:671–690
Gambaro R, Analía S, Gisel P (2018) Oxidative stress and genomic damage induced in vitro in human peripheral blood by two preventive treatments of iron deficiency anemia. Biol Trace Elem Res 188:1–9
Acknowledgements
We acknowledge National Research Centre that funded this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hamed, H.M., Motawie, A.A., Abd Al-Aziz, A.M. et al. Low Dose Iron Therapy in Children with Iron Deficiency: DNA Damage and Oxidant Stress Markers. Indian J Hematol Blood Transfus 37, 287–294 (2021). https://doi.org/10.1007/s12288-020-01340-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-020-01340-6