Abstract
Background
BRCA1/2 mutated breast cancer accounts for 3 to 12% of all women with breast cancer and significantly increases the lifetime risk of breast cancer. However, the optimal local treatment for breast cancer with BRCA germline mutation remains controversial. Here we present a meta-analysis to evaluate the impact of breast-conserving therapy (BCT) on the prognosis of breast cancer with BRCA mutation.
Methods
Two independent reviewers searched Pubmed, Embase and Cochrane Central Register of Controlled Trials databases for relevant studies on BCT and BRCA mutated breast cancer. Fixed or random effect models were used for meta-analyses based on whether significant heterogeneity existed among included studies. Funnel plot and Begg’s test were employed for the evaluation of publication bias.
Results
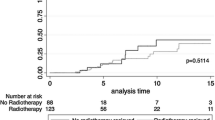
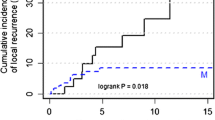
Totally, four studies with five cohorts and a totally 1254 patients were included for meta-analyses. The BCT group involved more T0/T1 (BCT 63.7% Vs. M 48.9%, p < 0.001), N0 (BCT 70.5% Vs. M 56.2%, p < 0.001) and ER negative (BCT 58.8% Vs. M 49.3% p < 0.01) tumors than M group. Patients who received M tended to have prophylactic contralateral mastectomy (BCT 16.5% Vs. M 35.8%, p < 0.001). BCT had a significant higher risk for local recurrence than M (HR 3.838, 95% CI = 2.376–6.201, p < 0.001). The pooled results revealed no significant impact of BCT on disease-free survival (DFS), metastasis-free survival (MFS), breast cancer-specific survival (BCSS) and overall survival (OS).
Conclusions
The present meta-analysis suggested that BCT had increasing local recurrence risk, but did not significantly impact patient survival in terms of DFS, MFS, BCSS and OS. BCT may serve as a safe alternative to mastectomy for breast cancer with BRCA mutation. Further high-quality randomized control trials are warranted to explore the optimal surgical management for BRCA mutation carriers.




Similar content being viewed by others
References
Tung NM, Boughey JC, Pierce LJ, Robson ME, Bedrosian I, Dietz JR, et al. Management of hereditary breast cancer: American Society of Clinical Oncology, American Society for Radiation Oncology, and Society of Surgical Oncology Guideline. J Clin Oncol. 2020;38(18):2080–106.
Copson ER, Maishman TC, Tapper WJ, Cutress RI, Greville-Heygate S, Altman DG, et al. Germline BRCA mutation and outcome in young-onset breast cancer (POSH): a prospective cohort study. Lancet Oncol. 2018;19(2):169–80.
Zhao W, Wiese C, Kwon Y, Hromas R, Sung P. The BRCA tumor suppressor network in chromosome damage repair by homologous recombination. Annu Rev Biochem. 2019;20(88):221–45.
Begg CB, Haile RW, Borg A, Malone KE, Concannon P, Thomas DC, et al. Variation of breast cancer risk among BRCA1/2 carriers. JAMA. 2008;299(2):194–201.
Struewing JP, Hartge P, Wacholder S, Baker SM, Berlin M, McAdams M, et al. The risk of cancer associated with specific mutations of BRCA1 and BRCA2 among Ashkenazi Jews. N Engl J Med. 1997;336(20):1401–8.
Honrado E, Osorio A, Palacios J, Benitez J. Pathology and gene expression of hereditary breast tumors associated with BRCA1, BRCA2 and CHEK2 gene mutations. Oncogene. 2006;25(43):5837–45.
Phillips KA. Current perspectives on BRCA1- and BRCA2-associated breast cancers. Intern Med J. 2001;31(6):349–56.
Valencia OM, Samuel SE, Viscusi RK, Riall TS, Neumayer LA, Aziz H. The role of genetic testing in patients with breast cancer: a review. JAMA Surg. 2017;152(6):589–94.
Balmaña J, Diez O, Rubio I, Castiglione M, ESMO Guidelines Working Group. BRCA in breast cancer: ESMO Clinical Practice Guidelines. Ann Oncol. 2010;21(Suppl 5):20–2.
Lort G, Chirivella I, Morales R, Serrano R, Sanchez AB, Teule A, et al. SEOM clinical guidelines in Hereditary Breast and ovarian cancer. Clin Transl Oncol. 2015;17(12):956–61.
Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353(17):1784–92.
Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002;347(16):1233–41.
Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A, et al. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med. 2002;347(16):1227–32.
van Maaren MC, de Munck L, de Bock GH, Jobsen JJ, van Dalen T, Linn SC, et al. 10 year survival after breast-conserving surgery plus radiotherapy compared with mastectomy in early breast cancer in the Netherlands: a population-based study. Lancet Oncol. 2016;17(8):1158–70.
Pierce LJ, Phillips K-A, Griffith KA, Buys S, Gaffney DK, Moran MS, et al. Local therapy in BRCA1 and BRCA2 mutation carriers with operable breast cancer: comparison of breast conservation and mastectomy. Breast Cancer Res Treat. 2010;121(2):389–98.
van Broek AJ, Schmidt MK, van Veer LJ, Oldenburg HSA, Rutgers EJ, Russell NS, et al. Prognostic impact of breast-conserving therapy versus mastectomy of BRCA1/2 mutation carriers compared with noncarriers in a consecutive series of young breast cancer patients. Ann Surg. 2019;270(2):364–72.
Huang X, Cai X-Y, Liu J-Q, Hao W-W, Zhou Y-D, Wang X, et al. Breast-conserving therapy is safe both within BRCA1/2 mutation carriers and noncarriers with breast cancer in the Chinese population. Gland Surg. 2020;9(3):775–87.
STROBE Statement: Available checklists [Internet]. [cited 2020 Aug 8]. Available from: https://www.strobe-statement.org/index.php?id=available-checklists
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet Lond Engl. 2007;370(9596):1453–7.
Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;7(8):16.
Nilsson MP, Hartman L, Kristoffersson U, Johannsson OT, Borg Å, Henriksson K, et al. High risk of in-breast tumor recurrence after BRCA1/2-associated breast cancer. Breast Cancer Res Treat. 2014;147(3):571–8.
Mavaddat N, Peock S, Frost D, Ellis S, Platte R, Fineberg E, et al. Cancer risks for BRCA1 and BRCA2 mutation carriers: results from prospective analysis of EMBRACE. J Natl Cancer Inst. 2013;105(11):812–22.
Giuliano AE, Ballman KV, McCall L, Beitsch PD, Brennan MB, Kelemen PR, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: The ACOSOG Z0011 (Alliance) Randomized Clinical Trial. JAMA. 2017;318(10):918.
Yadav S, Jinna S, Pereira-Rodrigues O, Reeves A, Campian S, Sufka A, et al. Impact of preoperative BRCA1/2 testing on surgical decision making in patients with newly diagnosed breast cancer. Breast J. 2018;24(4):541–8.
Kurian AW, Li Y, Hamilton AS, Ward KC, Hawley ST, Morrow M, et al. Gaps in incorporating germline genetic testing into treatment decision-making for early-stage breast cancer. J Clin Oncol. 2017;35(20):2232–9.
Co M, Liu T, Leung J, Li CH, Tse T, Wong M, et al. Breast conserving surgery for BRCA mutation carriers-a systematic review. Clin Breast Cancer. 2020;20(3):e244–50.
Valachis A, Nearchou AD, Lind P. Surgical management of breast cancer in BRCA-mutation carriers: a systematic review and meta-analysis. Breast Cancer Res Treat. 2014;144(3):443–55.
Molina-Montes E, Pérez-Nevot B, Pollán M, Sánchez-Cantalejo E, Espín J, Sánchez M-J. Cumulative risk of second primary contralateral breast cancer in BRCA1/BRCA2 mutation carriers with a first breast cancer: a systematic review and meta-analysis. Breast Edinb Scotl. 2014;23(6):721–42.
Turner BC, Harrold E, Matloff E, Smith T, Gumbs AA, Beinfield M, et al. BRCA1/BRCA2 germline mutations in locally recurrent breast cancer patients after lumpectomy and radiation therapy: implications for breast-conserving management in patients with BRCA1/BRCA2 mutations. J Clin Oncol. 1999;17(10):3017–24.
Anderson SJ, Wapnir I, Dignam JJ, Fisher B, Mamounas EP, Jeong J-H, et al. Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in patients treated by breast-conserving therapy in five National Surgical Adjuvant Breast and Bowel Project protocols of node-negative breast cancer. J Clin Oncol. 2009;27(15):2466–73.
Wapnir IL, Anderson SJ, Mamounas EP, Geyer CE, Jeong JH, Chiu ET, et al. Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in five National Surgical Adjuvant Breast and Bowel Project node-positive adjuvant breast cancer trials. J Clin Oncol. 2006. https://doi.org/10.1200/JCO.2005.04.3273.
Fourquet A, Stoppa-Lyonnet D, Kirova YM, Sigal-Zafrani B, Asselain B, Institut Curie Breast Cancer Study Group, et al. Familial breast cancer: clinical response to induction chemotherapy or radiotherapy related to BRCA1/2 mutations status. Am J Clin Oncol. 2009;32(2):127–31.
Byrski T, Gronwald J, Huzarski T, Grzybowska E, Budryk M, Stawicka M, et al. Pathologic complete response rates in young women with BRCA1-positive breast cancers after neoadjuvant chemotherapy. J Clin Oncol. 2010;28(3):375–9.
Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. The Lancet. 2014;384(9938):164–72.
Wang C-J, Xu Y, Lin Y, Zhu H-J, Zhou Y-D, Mao F, et al. Platinum-based neoadjuvant chemotherapy for breast cancer with BRCA mutations: a meta-analysis. Front Oncol. 2020;10:592998.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions version 6.1 (updated September 2020). Cochrane. 2020. Available from www.training.cochrane.org/handbook.
Acknowledgements
This study was funded by Key Projects in the National Science and Technology Pillar Program during the Twelfth Five-year Plan Period (No.2014BAI08B00), Beijing Municipal Science and Technology Project (No. D161100000816005) and State Key Laboratory of Medicinal Chemical Biology (NanKai University) (No. 2019014). The funding agencies had no role in the design or conduct of the study.
Funding
This study was funded by Key Projects in the National Science and Technology Pillar Program during the Twelfth Five‐year Plan Period (No.2014BAI08B00), Beijing Municipal Science and Technology Project (No. D161100000816005) and State Key Laboratory of Medicinal Chemical Biology (NanKai University) (No. 2019014).
Author information
Authors and Affiliations
Contributions
CW, YZ and QS designed the project; CW, YL and QS performed the literature search and data acquisition; CW and YL performed data extraction; FM, HZ, XH and XZ performed the statistical analyses for heterogeneity investigation; CW, XC, HZ and YZ supported the writing of the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors have declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

About this article
Cite this article
Wang, C., Lin, Y., Zhu, H. et al. Breast-conserving therapy for breast cancer with BRCA mutations: a meta-analysis. Breast Cancer 29, 314–323 (2022). https://doi.org/10.1007/s12282-021-01312-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-021-01312-2