Abstract
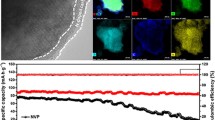
Carbonaceous materials have long been considered promising anode materials for Na-ion batteries. However, the electrochemical performance of conventional carbon anodes is generally poor because the sodium ion storage solely relies on the disordered region of the carbon materials in a carbonate-based electrolyte. The solvent co-intercalation mechanism for Na ions has been recently reported in natural graphite anodes for Na-ion batteries with ether-based electrolytes, but their capacities are still unsatisfactory. We show here for the first time that by combining regular Na ion storage in the disordered carbon layer and solvent co-intercalation mechanism in the graphitized layer of a commercial N330 carbon black as an anode material for Na-ion batteries in ether-based electrolyte, the reversible capacity could be fully realized and doubled in magnitude. This unique sodium intercalation process resulted in a significantly improved electrochemical performance for the N330 electrode with an initial reversible capacity of 234 mAh·g–1 at 50 mA·g–1 and a superior rate capability of 105 mAh·g–1 at 3,200 mA·g–1. When cycled at 3,200 mA·g–1 over 2,000 cycles, the electrode still exhibited a highly reversible capacity of 72 mAh·g–1 with a negligible capacity loss per cycle (0.0167%). Additionally, surface-sensitive C K-edge X-ray absorption spectroscopy, with the assistance of electrochemical and physicochemical characterizations, helped in identifying the controlled formation and evolution of a thin and robust solid electrolyte interphase film. This film not only reduced the resistance for sodium ion diffusion, but also maintained the structural stability of the electrode for extended cycle reversibility. The superior electrochemical performance of N330 carbon black strongly demonstrated the potential of applying ether-based electrolytes for a wide range of carbon anodes apart from natural graphite.

Similar content being viewed by others
References
Tarascon, J. M. Is lithium the new gold? Nat. Chem. 2010, 2, 510.
Kundu, D.; Talaie, E.; Duffort, V.; Nazar, L. F. The emerging chemistry of sodium ion batteries for electrochemical energy storage. Angew. Chem., Int. Ed. 2015, 54, 3431–3448.
Yabuuchi, N.; Kubota, K.; Dahbi, M.; Komaba, S. Research development on sodium-ion batteries. Chem. Rev. 2014, 114, 11636–11682.
Palomares, V.; Serras, P.; Villaluenga, I.; Hueso, K. B.; Carretero-González, J.; Rojo, T. Na-ion batteries, recent advances and present challenges to become low cost energy storage systems. Energy Environ. Sci. 2012, 5, 5884–5901.
Pan, H. L.; Hu, Y.-S.; Chen, L. Q. Room-temperature stationary sodium-ion batteries for large-scale electric energy storage. Energy Environ. Sci. 2013, 6, 2338–2360.
Hong, S. Y.; Kim, Y.; Park, Y.; Choi, A.; Choi, N.-S.; Lee, K. T. Charge carriers in rechargeable batteries: Na ions vs. Li ions. Energy Environ. Sci. 2013, 6, 2067–2081.
Okoshi, M.; Yamada, Y.; Yamada, A.; Nakai, H. Theoretical analysis on de-solvation of lithium, sodium, and magnesium cations to organic electrolyte solvents. J. Electrochem. Soc. 2013, 160, A2160–A2165.
Ong, S. P.; Chevrier, V. L.; Hautier, G.; Jain, A.; Moore, C.; Kim, S.; Ma, X. H.; Ceder, G. Voltage, stability and diffusion barrier differences between sodium-ion and lithium-ion intercalation materials. Energy Environ. Sci. 2011, 4, 3680–3688.
Asher, R. C. A lamellar compound of sodium and graphite. J. Inorg. Nucl. Chem. 1959, 10, 238–249.
DiVincenzo, D. P.; Mele, E. J. Self-consistent effective-mass theory for intralayer screening in graphite intercalation compounds. Phys. Rev. B 1984, 29, 1685–1694.
Ge, P.; Fouletier, M. Electrochemical intercalation of sodium in graphite. Solid State Ionics 1988, 28–30, 1172–1175.
Nobuhara, K.; Nakayama, H.; Nose, M.; Nakanishi, S.; Iba, H. First-principles study of alkali metal-graphite intercalation compounds. J. Power Sources 2013, 243, 585–587.
Stevens, D. A.; Dahn, J. R. High capacity anode materials for rechargeable sodium-ion batteries. J. Electrochem. Soc. 2000, 147, 1271–1273.
Doeff, M. M.; Ma, Y. P.; Visco, S. J.; De Jonghe, L. C. Electrochemical insertion of sodium into carbon. J. Electrochem. Soc. 1993, 140, L169–L170.
Xie, X. Q.; Ao, Z. M.; Su, D. W.; Zhang, J. Q.; Wang, G. X. MoS2/graphene composite anodes with enhanced performance for sodium-ion batteries: The role of the two-dimensional heterointerface. Adv. Funct. Mater. 2015, 25, 1393–1403.
Xie, X. Q.; Zhao, M.-Q.; Anasori, B.; Maleski, K.; Ren, C. E.; Li, J. W.; Byles, B. W.; Pomerantseva, E.; Wang, G.; Gogotsi, Y. Porous heterostructured MXene/carbon nanotube composite paper with high volumetric capacity for sodiumbased energy storage devices. Nano Energy 2016, 26, 513–523.
Xie, X. Q.; Makaryan, T.; Zhao, M. Q.; Van Aken, K. L.; Gogotsi, Y.; Wang, G. X. MoS2 nanosheets vertically aligned on carbon paper: A freestanding electrode for highly reversible sodium-ion batteries. Adv. Energy Mater. 2016, 6, 1502161.
Bommier, C.; Ji, X. L. Recent development on anodes for Na-ion batteries. Isr. J. Chem. 2015, 55, 486–507.
Kim, Y.; Ha, K. H.; Oh, S. M.; Lee, K. T. High-capacity anode materials for sodium-ion batteries. Chem.—Eur. J. 2014, 20, 11980–11992.
Ponrouch, A.; Dedryvère, R.; Monti, D.; Demet, A. E.; Ateba Mba, J. M.; Croguennec, L.; Masquelier, C.; Johansson, P.; Palacín, M. R. Towards high energy density sodium ion batteries through electrolyte optimization. Energy Environ. Sci. 2013, 6, 2361–2369.
Ponrouch, A.; Marchante, E.; Courty, M.; Tarascon, J.-M.; Palacín, M. R. In search of an optimized electrolyte for Na-ion batteries. Energy Environ. Sci. 2012, 5, 8572–8583.
Jache, B.; Adelhelm, P. Use of graphite as a highly reversible electrode with superior cycle life for sodium-ion batteries by making use of Co-intercalation phenomena. Angew. Chem., Int. Ed. 2014, 53, 10169–10173.
Kim, H.; Hong, J.; Park, Y.-U.; Kim, J.; Hwang, I.; Kang, K. Sodium storage behavior in natural graphite using ether-based electrolyte systems. Adv. Funct. Mater. 2015, 25, 534–541.
Kim, H.; Hong, J.; Yoon, G.; Kim, H.; Park, K.-Y.; Park, M.-S.; Yoon, W.-S.; Kang, K. Sodium intercalation chemistry in graphite. Energy Environ. Sci. 2015, 8, 2963–2969.
Jung, S. C.; Kang, Y.-J.; Han, Y.-K. Origin of excellent rate and cycle performance of Na+-solvent cointercalated graphite vs. poor performance of Li+-solvent case. Nano Energy 2017, 34, 456–462.
Zhu, Y.-E.; Yang, L. P.; Zhou, X. L.; Li, F.; Wei, J. P.; Zhou, Z. Boosting the rate capability of hard carbon with an ether-based electrolyte for sodium ion batteries. J. Mater. Chem. A 2017, 5, 9528–9532.
Seidl, L.; Bucher, N.; Chu, E.; Hartung, S.; Martens, S.; Schneider, O.; Stimming, U. Intercalation of solvated Na-ions into graphite. Energy Environ. Sci. 2017, 10, 1631–1642.
Zhang, J.; Wang, D.-W.; Lv, W.; Zhang, S. W.; Liang, Q. H.; Zheng, D. Q.; Kang, F. Y.; Yang, Q.-H. Achieving superb sodium storage performance on carbon anodes through an ether-derived solid electrolyte interphase. Energy Environ. Sci. 2017, 10, 370–376.
Jache, B.; Binder, J. O.; Abe, T.; Adelhelm, P. A comparative study on the impact of different glymes and their derivatives as electrolyte solvents for graphite co-intercalation electrodes in lithium-ion and sodium-ion batteries. Phys. Chem. Chem. Phys. 2016, 18, 14299–14316.
Guan, Z. X.; Shen, X.; Yu, R. C.; Wang, Z. X.; Chen, L. Q. Chemical intercalation of solvated sodium ions in graphite. Electrochim. Acta 2016, 222, 1365–1370.
Hasa, I.; Dou, X. W.; Buchholz, D.; Shao-Horn, Y.; Hassoun, J.; Passerini, S.; Scrosati, B. A sodium-ion battery exploiting layered oxide cathode, graphite anode and glymebased electrolyte. J. Power Sources 2016, 310, 26–31.
Zhu, Z. Q.; Cheng, F. Y.; Hu, Z.; Niu, Z. Q.; Chen, J. Highly stable and ultrafast electrode reaction of graphite for sodium ion batteries. J. Power Sources 2015, 293, 626–634.
Wen, Y.; He, K.; Zhu, Y. J.; Han, F. D.; Xu, Y. H.; Matsuda, I.; Ishii, Y.; Cumings, J.; Wang, C. S. Expanded graphite as superior anode for sodium-ion batteries. Nat. Commun. 2014, 5, 4033.
Peled, E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems-the solid electrolyte interphase model. J. Electrochem. Soc. 1979, 126, 2047–2051.
Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 2014, 114, 11503–11618.
Xiao, L. F.; Cao, Y. L.; Henderson, W. A.; Sushko, M. L.; Shao, Y. Y.; Xiao, J.; Wang, W.; Engelhard, M. H.; Nie, Z. M.; Liu, J. Hard carbon nanoparticles as high-capacity, highstability anodic materials for Na-ion batteries. Nano Energy 2016, 19, 279–288.
Lotfabad, E. M.; Kalisvaart, P.; Kohandehghan, A.; Karpuzov, D.; Mitlin, D. Origin of non-SEI related coulombic efficiency loss in carbons tested against Na and Li. J. Mater. Chem. A 2014, 2, 19685–19695.
Zhou, J. G.; Zhou, X. T.; Li, R. Y.; Sun, X. L.; Ding, Z. F.; Cutler, J.; Sham, T.-K. Electronic structure and luminescence center of blue luminescent carbon nanocrystals. Chem. Phys. Lett. 2009, 474, 320–324.
Zhou, J. G.; Zhou, X. T.; Sun, X. H.; Li, R. Y.; Murphy, M.; Ding, Z. F.; Sun, X. L.; Sham, T.-K. Interaction between Pt nanoparticles and carbon nanotubes—An X-ray absorption near edge structures (XANES) study. Chem. Phys. Lett. 2007, 437, 229–232.
Bouchet-Fabre, B.; Pinault, M.; Pichot, V.; Launois, P.; Mayne-L'Hermite, M.; Parent, P.; Laffon, K.; Durand, D.; Reynaud, C. NEXAFS and X-ray scattering study of structure changes after post-annealing treatments of aligned MWNTs. Diamond Relat. Mater. 2005, 14, 881–886.
Roy, S. S.; Papakonstantinou, P.; Okpalugo, T. I. T.; Murphy, H. Temperature dependent evolution of the local electronic structure of atmospheric plasma treated carbon nanotubes: Near edge X-ray absorption fine structure study. J. Appl. Phys. 2006, 100, 053703.
Banerjee, S.; Hemraj-Benny, T.; Balasubramanian, M.; Fischer, D. A.; Misewich, J. A.; Wong, S. S. Ozonized single-walled carbon nanotubes investigated using NEXAFS spectroscopy. Chem. Commun. 2004, 772–773.
Kuznetsova, A.; Popova, I.; Yates Jr, J. T.; Bronikowski, M. J.; Huffman, C. B.; Liu, J.; Smalley, R. E.; Hwu, H. H.; Chen, J. G. Oxygen-containing functional groups on single-wall carbon nanotubes: NEXAFS and vibrational spectroscopic studies. J. Am. Chem. Soc. 2001, 123, 10699–10704.
Nyberg, M.; Hasselström, J.; Karis, O.; Wassdahl, N.; Weinelt, M.; Nilsson, A.; Pettersson, L. G. M. The electronic structure and surface chemistry of glycine adsorbed on Cu(110). J. Chem. Phys. 2000, 112, 5420–5427.
Seh, Z. W.; Sun, J.; Sun, Y. M.; Cui, Y. A highly reversible room-temperature sodium metal anode. ACS Cent. Sci. 2015, 1, 449–455.
Stevens, D. A.; Dahn, J. R. The mechanisms of lithium and sodium insertion in carbon materials. J. Electrochem. Soc. 2001, 148, A803–A811.
Bommier, C.; Surta, T. W.; Dolgos, M.; Ji, X. L. New mechanistic insights on Na-ion storage in nongraphitizable carbon. Nano Lett. 2015, 15, 5888–5892.
Jaouen, F.; Dodelet, J.-P. Non-noble electrocatalysts for O2 reduction: How does heat treatment affect their activity and structure? Part I. Model for carbon black gasification by NH3: Parametric calibration and electrochemical validation. J. Phys. Chem. C 2007, 111, 5963–5970.
Li, Y. L.; Li, X. F.; Geng, D. S.; Tang, Y. J.; Li, R. Y.; Dodelet, J.-P.; Lefèvre, M.; Sun, X. L. Carbon black cathodes for lithium oxygen batteries: Influence of porosity and heteroatom-doping. Carbon 2013, 64, 170–177.
Acknowledgements
This research was supported by the Natural Science and Engineering Research Council of Canada (NSERC), the Canada Research Chair Program (CRC), the Canada Foundation for Innovation (CFI), and the University of Western Ontario (UWO). J. L. is grateful to the financial support from NSERC Postdoctoral Fellowships Program. The ALS is supported by the Director, Office of Science, Office of Basic Energy Sciences, of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xiao, W., Sun, Q., Liu, J. et al. Utilizing the full capacity of carbon black as anode for Na-ion batteries via solvent co-intercalation. Nano Res. 10, 4378–4387 (2017). https://doi.org/10.1007/s12274-017-1852-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1852-4