Abstract
Ischemic heart disease is caused by coronary artery occlusion. Despite the increasing number and success of interventions for restoring coronary artery perfusion, myocardial ischemia–reperfusion (I/R) injury remains a significant cause of morbidity and mortality worldwide. Inspired by the impact of I/R on the Cx43 trafficking to the intercalated discs (ICDs), we aim to explore the potential mechanisms underlying the downregulation of Cx43 in ICDs after myocardial I/R. Gene set enrichment analysis (GSEA), Western blotting, and immunofluorescence experiments showed that Myocardial I/R activated the P38MAPK signaling pathway and promoted microtubule depolymerization. Inhibition of P38MAPK signaling pathway activation attenuated I/R-induced microtubule depolymerization. The ability of SB203580 to recover the distribution of Cx43 and electrophysiological parameters in I/R myocardium depended on microtubule stability. Our study suggests that microtubule depolymerization caused by the activation of the P38MAPK signaling pathway is an important mechanism underlying the downregulation of Cx43 in ICDs after myocardial I/R.
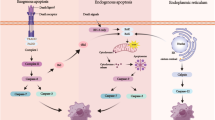
Graphical Abstract








Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- DMEM:
-
Dulbecco’s modified Eagle’s medium; DMSO: Dimethyl sulfoxide;
- ECG:
-
Electrocardiography; GOBP: Gene Ontology Biological Process;
- GSEA:
-
Gene set enrichment analysis; ICDs: intercalated discs;
- IHD:
-
Ischemic heart disease; I/R: Ischemia–reperfusion;
- MAPs:
-
Microtubule-associated proteins;
- MIRI:
-
Myocardial ischemic-reperfusion injury; SPF: Specific pathogen-free
References
Moran AE, Forouzanfar MH, Roth GA, et al. The global burden of ischemic heart disease in 1990 and 2010: the Global Burden of Disease 2010 study. Circulation. 2014;129:1493–501. https://doi.org/10.1161/circulationaha.113.004046.
Davidson SM, Ferdinandy P, Andreadou I, et al. Multitarget Strategies to Reduce Myocardial Ischemia/Reperfusion Injury: JACC Review Topic of the Week. J Am Coll Cardiol. 2019;73:89–99. https://doi.org/10.1016/j.jacc.2018.09.086.
Yi J, Duan H, Chen K, et al. Cardiac Electrophysiological Changes and Downregulated Connexin 43 Prompts Reperfusion Arrhythmias Induced by Hypothermic Ischemia-Reperfusion Injury in Isolated Rat Hearts. J Cardiovasc Transl Res. 2022;15:1464–73. https://doi.org/10.1007/s12265-022-10256-7.
Li W, Gao H, Gao J, et al. Upregulation of MMP-9 and CaMKII prompts cardiac electrophysiological changes that predispose denervated transplanted hearts to arrhythmogenesis after prolonged cold ischemic storage. Biomed Pharmacother. 2019;112:108641. https://doi.org/10.1016/j.biopha.2019.108641.
Boengler K, Schulz R. Connexin 43 and Mitochondria in Cardiovascular Health and Disease. Exp Biol Med. 2017;982:227–46. https://doi.org/10.1007/978-3-319-55330-6_12.
Leybaert L, Lampe PD, Dhein S, et al. Connexins in Cardiovascular and Neurovascular Health and Disease: Pharmacological Implications. Pharmacol Rev. 2017;69:396–478. https://doi.org/10.1124/pr.115.012062.
Akar FG, Nass RD, Hahn S, et al. Dynamic changes in conduction velocity and gap junction properties during development of pacing-induced heart failure. Am J Physiol Heart Circ Physiol. 2007;293:H1223–30. https://doi.org/10.1152/ajpheart.00079.2007.
Beardslee MA, Lerner DL, Tadros PN, et al. Dephosphorylation and intracellular redistribution of ventricular connexin43 during electrical uncoupling induced by ischemia. Circ Res. 2000;87:656–62. https://doi.org/10.1161/01.res.87.8.656.
Thibodeau IL, Xu J, Li Q, et al. Paradigm of genetic mosaicism and lone atrial fibrillation: physiological characterization of a connexin 43-deletion mutant identified from atrial tissue. Circulation. 2010;122:236–44. https://doi.org/10.1161/circulationaha.110.961227.
Marais E, Genade S, Huisamen B, et al. Activation of p38 MAPK induced by a multi-cycle ischaemic preconditioning protocol is associated with attenuated p38 MAPK activity during sustained ischaemia and reperfusion. J Mol Cell Cardiol. 2001;33:769–78. https://doi.org/10.1006/jmcc.2001.1347.
Ahmad F, Tomar D, Aryal ACS, et al. Nicotinamide riboside kinase-2 alleviates ischemia-induced heart failure through P38 signaling. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165609. https://doi.org/10.1016/j.bbadis.2019.165609.
Xu L, Yates CC, Lockyer P, et al. MMI-0100 inhibits cardiac fibrosis in myocardial infarction by direct actions on cardiomyocytes and fibroblasts via MK2 inhibition. J Mol Cell Cardiol. 2014;77:86–101. https://doi.org/10.1016/j.yjmcc.2014.09.011.
Wang M, Li Z, Zhang X, et al. Rosuvastatin attenuates atrial structural remodelling in rats with myocardial infarction through the inhibition of the p38 MAPK signalling pathway. Heart Lung Circ. 2015;24:386–94. https://doi.org/10.1016/j.hlc.2014.11.012.
Li M, Liu F, Sang M, et al. Effects of atorvastatin on p38 phosphorylation and cardiac remodeling after myocardial infarction in rats. Exp Ther Med. 2018;16:751–7. https://doi.org/10.3892/etm.2018.6201.
Prompunt E, Sanit J, Barrère-Lemaire S, et al. The cardioprotective effects of secretory leukocyte protease inhibitor against myocardial ischemia/reperfusion injury. Exp Ther Med. 2018;15:5231–42. https://doi.org/10.3892/etm.2018.6097.
Rogers SL, Gelfand VI. Membrane trafficking, organelle transport, and the cytoskeleton. Curr Opin Cell Biol. 2000;12:57–62. https://doi.org/10.1016/s0955-0674(99)00057-5.
Mitchison T, Kirschner M. Dynamic instability of microtubule growth. Nature. 1984;312:237–42. https://doi.org/10.1038/312237a0.
Dehmelt L, Halpain S. The MAP2/Tau family of microtubule-associated proteins. Genome biol. 2005;6:204. https://doi.org/10.1186/gb-2004-6-1-204.
Halpain S, Dehmelt L. The MAP1 family of microtubule-associated proteins. Genome biol. 2006;7:224. https://doi.org/10.1186/gb-2006-7-6-224.
Zhang J, Li L, Zhang Q, et al. Phosphorylation of Microtubule- Associated Protein 4 Promotes Hypoxic Endothelial Cell Migration and Proliferation. Front Pharmacol. 2019;10:368. https://doi.org/10.3389/fphar.2019.00368.
Shaw RM, Fay AJ, Puthenveedu MA, et al. Microtubule plus-end-tracking proteins target gap junctions directly from the cell interior to adherens junctions. Cell. 2007;128:547–60. https://doi.org/10.1016/j.cell.2006.12.037.
Smyth JW, Hong TT, Gao D, et al. Limited forward trafficking of connexin 43 reduces cell-cell coupling in stressed human and mouse myocardium. J Clin Investig. 2010;120:266–79. https://doi.org/10.1172/jci39740.
Ligon LA, Holzbaur EL. Microtubules tethered at epithelial cell junctions by dynein facilitate efficient junction assembly. Traffic. 2007;8:808–19. https://doi.org/10.1111/j.1600-0854.2007.00574.x.
Levy JR, Holzbaur EL. Special delivery: dynamic targeting via cortical capture of microtubules. Dev Cell. 2007;12:320–2. https://doi.org/10.1016/j.devcel.2007.02.012.
Hu JY, Chu ZG, Han J, et al. The p38/MAPK pathway regulates microtubule polymerization through phosphorylation of MAP4 and Op18 in hypoxic cells. Cell Mol Life Sci. 2010;67:321–33. https://doi.org/10.1007/s00018-009-0187-z.
van Rijen HV, Eckardt D, Degen J, et al. Slow conduction and enhanced anisotropy increase the propensity for ventricular tachyarrhythmias in adult mice with induced deletion of connexin43. Circulation. 2004;109:1048–55. https://doi.org/10.1161/01.cir.0000117402.70689.75.
Fontes MS, van Veen TA, de Bakker JM, et al. Functional consequences of abnormal Cx43 expression in the heart. Biochim Biophys Acta Biomembr. 2012;1818:2020–9. https://doi.org/10.1016/j.bbamem.2011.07.039.
Smyth JW, Vogan JM, Buch PJ, et al. Actin Cytoskeleton Rest Stops Regulate Anterograde Traffic of Connexin 43 Vesicles to the Plasma Membrane. Circ Res. 2012;110:978–89. https://doi.org/10.1161/circresaha.111.257964.
Veeraraghavan R, Lin J, Hoeker GS, et al. Sodium channels in the Cx43 gap junction perinexus may constitute a cardiac ephapse: an experimental and modeling study. Pflügers Arch Eur J Physiol. 2015;467:2093–105. https://doi.org/10.1007/s00424-014-1675-z.
Matthew Rhett J, Ongstad EL, Jourdan J, et al. Cx43 Associates with Nav1.5 in the Cardiomyocyte Perinexus. J Membr Biol. 2012;245:411–22. https://doi.org/10.1007/s00232-012-9465-z.
Nasilli G, Lin X, Perez-Hernandez M et al Targeting microtubule dynamics improves cardiac sodium channel function in arrhythmogenic cardiomyopathy. European Heart Journal. (2023) 44: https://doi.org/10.1093/eurheartj/ehad655.3060
Smyth JW, Shaw RM. Autoregulation of connexin43 gap junction formation by internally translated isoforms. Cell Rep. 2013;5:611–8. https://doi.org/10.1016/j.celrep.2013.10.009.
Basheer WA, Xiao S, Epifantseva I, et al. GJA1-20k Arranges Actin to Guide Cx43 Delivery to Cardiac Intercalated Discs. Circ Res. 2017;121:1069–80. https://doi.org/10.1161/circresaha.117.311955.
Basheer WA, Fu Y, Shimura D et al Stress response protein GJA1–20k promotes mitochondrial biogenesis, metabolic quiescence, and cardioprotection against ischemia/reperfusion injury. JCI insight. (2018) 3: https://doi.org/10.1172/jci.insight.121900
Xiao S, Shimura D, Baum R, et al. Auxiliary trafficking subunit GJA1-20k protects connexin-43 from degradation and limits ventricular arrhythmias. J Clin Invest. 2020;130:4858–70. https://doi.org/10.1172/jci134682.
Chen L, Yuan J, Li H, et al. Trans-cinnamaldehyde attenuates renal ischemia/reperfusion injury through suppressing inflammation via JNK/p38 MAPK signaling pathway. Int Immunopharmacol. 2023;118:110088. https://doi.org/10.1016/j.intimp.2023.110088.
Liu XM, Chen QH, Hu Q, et al. Dexmedetomidine protects intestinal ischemia-reperfusion injury via inhibiting p38 MAPK cascades. Exp Mol Pathol. 2020;115:104444. https://doi.org/10.1016/j.yexmp.2020.104444.
Wang T, Liu C, Pan LH, et al. Inhibition of p38 MAPK Mitigates Lung Ischemia Reperfusion Injury by Reducing Blood-Air Barrier Hyperpermeability. Front Pharmacol. 2020;11:569251. https://doi.org/10.3389/fphar.2020.569251.
Zeng JJ, Shi HQ, Ren FF, et al. Notoginsenoside R1 protects against myocardial ischemia/reperfusion injury in mice via suppressing TAK1-JNK/p38 signaling. Acta Pharmacol Sin. 2023. https://doi.org/10.1038/s41401-023-01057-y.
Gudimchuk NB, McIntosh JR. Regulation of microtubule dynamics, mechanics and function through the growing tip. Nat Rev Mol Cell Biol. 2021;22:777–95. https://doi.org/10.1038/s41580-021-00399-x.
Etienne-Manneville S. From signaling pathways to microtubule dynamics: the key players. Curr Opin Cell Biol. 2010;22:104–11. https://doi.org/10.1016/j.ceb.2009.11.008.
Wloga D, Joachimiak E, Fabczak H Tubulin Post-Translational Modifications and Microtubule Dynamics. Int J Molecular Scie (2017) 18: https://doi.org/10.3390/ijms18102207
Kang J, Kang N, Lovatt D, et al. Connexin 43 hemichannels are permeable to ATP. J Neurosci: Official J Soc Neurosci. 2008;28:4702–11. https://doi.org/10.1523/jneurosci.5048-07.2008.
Ai X, Yan J, Pogwizd SM. Serine-threonine protein phosphatase regulation of Cx43 dephosphorylation in arrhythmogenic disorders. Cell Signal. 2021;86:110070. https://doi.org/10.1016/j.cellsig.2021.110070.
Xue J, Yan X, Yang Y, et al. Connexin 43 dephosphorylation contributes to arrhythmias and cardiomyocyte apoptosis in ischemia/reperfusion hearts. Basic Res Cardiol. 2019;114:40. https://doi.org/10.1007/s00395-019-0748-8.
Morel S, Christoffersen C, Axelsen LN, et al. Sphingosine-1-phosphate reduces ischaemia-reperfusion injury by phosphorylating the gap junction protein Connexin43. Cardiovasc Res. 2016;109:385–96. https://doi.org/10.1093/cvr/cvw004.
Kim B, Breton S. The MAPK/ERK signaling pathway regulates the expression and localization of Cx43 in mouse proximal epididymis†. Biol Reprod. 2022;106:919–27. https://doi.org/10.1093/biolre/ioac034.
Zhong C, Zhao H, Xie X, et al. Protein Kinase C-Mediated Hyperphosphorylation and Lateralization of Connexin 43 Are Involved in Autoimmune Myocarditis-Induced Prolongation of QRS Complex. Front Physiol. 2022;13:815301. https://doi.org/10.3389/fphys.2022.815301.
Acknowledgements
We thank Figdraw (www.figdraw.com) for their help in creating the schematic representation of the experimental protocol.
Funding
This study was supported by grants from the Science and Technology Fund Project of the Guizhou Provincial Health Commission (gzwkj2021-270, Gzwkj2022-131).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
No human studies were carried out by the authors for this article. The protocol of the experiments was approved by the Ethics Committee of Guizhou Medical University (approval number: 2303066). The animal care and use protocol was adhered to the Chinese National Laboratory Animal-Guideline for Ethical Review of Animal Welfare (GB/T 35892–2018).
Conflict of Interest
The authors declare that they have no conflict of interest.
Human subjects/informed consent statement
No human studies were carried out by the authors for this article.
Additional information
Communicated by Associate Editor Geert Boink oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Bai, X., Yi, J. et al. The activation of P38MAPK Signaling Pathway Impedes the Delivery of the Cx43 to the Intercalated Discs During Cardiac Ischemia–Reperfusion Injury. J. of Cardiovasc. Trans. Res. (2024). https://doi.org/10.1007/s12265-024-10515-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12265-024-10515-9