Abstract
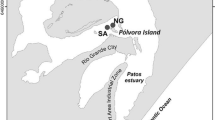
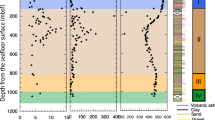
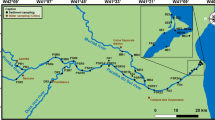
The arsenic (As) contents of sediment cores from the Patos Lagoon in southern Brazil were measured to better understand the biogeochemical cycling of As in the estuary. Sediment cores (ca. 60 cm) were obtained from three locations within the estuary to capture possible changes in As content across the salinity gradient (i.e., where saline, brackish, and freshwater dominated). Two sediment cores were collected at each location, one beneath open water and the other from the fringing salt marsh. Along with As, we quantified the particle size; redox potential (Eh); manganese (Mn), iron (Fe), total organic carbon (TOC), and free (dissolved) sulfide concentrations; and acid volatile sulfide (AVS) and chromium reducible sulfide (CRS) contents. Bioturbation supports oxygen penetration to depths between 20 and 30 cm below the salt marsh surface, where an As- and Fe-rich zone was identified (3-fold higher As than mean As contents of the sediments). A similar subsurface peak of As, Fe, and Mn occurs in the open-water cores, albeit at greater depths between 40 and 50 cm below the surface. The subsurface peak has As concentrations that are 2-fold higher than the average for each core. Vertical profiles of Eh, free sulfides, and CRS for the shallow open-water cores showed similar distribution at depths of 50 cm, suggesting that pyrite formation is an important sink for As in the open-water cores. The data demonstrate clear differences in the geochemical conditions for salt marshes and shallow open waters that can have important implications for As distribution in estuarine sediments.






Similar content being viewed by others
Notes
SiMCosta is a Brazilian Coast Monitoring System, which consist of an integrated network of floating or fixed platforms, equipped with instruments and sensors, with autonomous operation and capacity to collect real-time oceanographic and meteorological variables, transmitting them to a processing center at Federal University of Rio Grande (FURG)
References
Beck, M., O. Dellwig, B. Schnetger, and H.J. Brumsack. 2008. Cycling of trace metals (Mn, Fe, Mo, U, V, Cr) in deep pore waters of intertidal flat sediments. Geochimica et Cosmochimica Acta 72 (12): 2822–2840. https://doi.org/10.1016/j.gca.2008.04.013.
Bednar, A.J., J.R. Garbarino, J.F. Ranville, and T.R. Wildeman. 2005. Effects of iron on arsenic speciation and redox chemistry in acid mine water. Journal of Geochemical Exploration 85 (2): 55–62. https://doi.org/10.1016/j.gexplo.2004.10.001.
Berner, R.A. 1981. A new geochemical classification of sedimentary environments. Journal of Sedimentary Petrology 51. https://doi.org/10.1306/212F7C7F-2B24-11D7-8648000102C1865D.
Bhattacharyya, R., D. Chatterjee, B. Nath, J. Jana, G. Jacks, and M. Vahter. 2003. High arsenic groundwater: mobilization, metabolism and mitigation - an overview in the Bengal Delta Plain. Molecular and Cellular Biochemistry 253 (1/2): 347–355. https://doi.org/10.1023/A:1026001024578.
Bone, S.E., M.E. Gonneea, and M.A. Charette. 2006. Geochemical cycling of arsenic in a coastal aquifer. Environmental Science & Technology 40 (10): 3273–3278. https://doi.org/10.1021/es052352h.
Brannon, J.M., and W.H. Patrick. 1987. Fixation, transformation, and mobilization of arsenic in sediments. Environmental Science & Technology 21 (5): 450–459. https://doi.org/10.1021/es00159a005.
Brooks, K.M. 2001. An evaluation of the relationship between salmon farm biomass, organic inputs to sediments, physiochemical changes associated with those inputs and the infaunal response - with emphasis on total sediment sulfides, total volatile solids, and oxidation-reduction potential as surrogate endpoints for biological monitoring, final report. Port Townsend: Kenneth M. Brooks.
Burdige, D.J. 1993. The biogeochemistry of manganese and iron reduction in marine sediments. Earth-Science Reviews 35 (3): 249–284. https://doi.org/10.1016/0012-8252(93)90040-E.
Canfield, D.E., and B. Thamdrup. 2009. Towards a consistent classification scheme for geochemical environments, or, why we wish the term “suboxic” would go away: Editorial. Geobiology. 7 (4): 385–392. https://doi.org/10.1111/j.1472-4669.2009.00214.x.
Chaillou, G., J. Schäfer, P. Anschutz, G. Lavaux, and G. Blanc. 2003. The behaviour of arsenic in muddy sediments of the Bay of Biscay (France). Geochimica et Cosmochimica Acta 67 (16): 2993–3003. https://doi.org/10.1016/S0016-7037(03)00204-7.
Charette, M.A., and E.R. Sholkovitz. 2002. Oxidative precipitation of groundwater-derived ferrous iron in the subterranean estaury of a coastal bay. Geophysical Research Letters 29 (10). https://doi.org/10.1029/2001GLO4512.
Choi, S., P.A. O’Day, and J.G. Hering. 2009. Natural attenuation of arsenic by sediment sorption and oxidation. Environmental Science & Technology 43 (12): 4253–4259. https://doi.org/10.1021/es802841x.
Clark, M.W., D. McConchie, D.W. Lewis, and P. Saenger. 1998. Redox stratification and heavy metal partitioning in Avicennia-dominated mangrove sediments: a geochemical model. Chemical Geology 149 (3-4): 147–171. https://doi.org/10.1016/S0009-2541(98)00034-5.
Costa, C.S.B., J.C. Marangoni, and A.M.G. Azevedo. 2003. Plant zonation in irregularly flooded salt marshes: relative importance of stress tolerance and biological interactions. Journal of Ecology. https://doi.org/10.1046/j.1365-2745.2003.00821.x.
Costa, L., N. Mirlean, and F. Garcia. 2017. Arsenic environmental threshold surpass in estuarine sediments: effects of bioturbation. Bulletin of Environmental Contamination and Toxicology 98 (4): 521–524. https://doi.org/10.1007/s00128-016-2024-z.
D’Incao, F., et al. 1992. Responses of Chasmagnathus granulata Dana (Decapoda: Grapsidae) to salt-marsh environmental variations. Journal of Experimental Marine Biology and Ecology 161 (2): 179–188. https://doi.org/10.1016/0022-0981(92)90095-R.
Dixit, S., and J.G. Hering. 2003. Comparison of arsenic(V) and arsenic (III) sorption onto iron oxide minerals: Implications for arsenic mobility. Environmental Science & Technology 37 (18): 4182–4189. https://doi.org/10.1021/es030309t.
Doyle, M.O., and M.L. Otte. 1997. Organism-induced accumulation of iron, zinc and arsenic in wetland soils. Environmental Pollution 96 (1): 1–11. https://doi.org/10.1016/S0269-7491(97)00014-6.
Edenborn, H.M., N. Belzile, A. Mucci, J. Lebel, and N. Silverberg. 1986. Observations on the diagenetic behavior of arsenic in a deep coastal sediment. Biogeochemistry. 2 (4): 359–376. https://doi.org/10.1007/BF02180326.
Elderfield, H., and A. Hepworth. 1975. Diagenesis, metals and pollution in estuaries. Marine Pollution Bulletin 6 (6): 85–87. https://doi.org/10.1016/0025-326X(75)90149-6.
Fitz, W.J., and W.W. Wenzel. 2003. Environmental chemistry of arsenic. Journal of Environmental Quality 32 (4): 1572–157a. https://doi.org/10.2134/jeq2003.1572a.
Flora, S. J. S. (2015) Handbook of Arsenic Toxicology, Handbook of arsenic toxicology. https://doi.org/10.1016/C2013-0-08322-3.
Fossing, H., and B.B. Jørgensen. 1989. Measurement of bacterial sulfate reduction in sediments: evaluation of a single-step chromium reduction method. Biogeochemistry. 8 (3). https://doi.org/10.1007/BF00002889.
Froelich, P.N., G.P. Klinkhammer, M.L. Bender, N.A. Luedtke, G.R. Heath, D. Cullen, and P. Dauphin. 1979. Early oxidation of organic matter in pelagic sediments of the eastern equitorial Atlantic: suboxic diagenesis. Geochimica et Cosmochimica Acta 43 (7): 1075–1090.
Gaillard, J.-F., H. Pauwels, and G. Michard. 1989. Chemical diagenesis in coastal marine sediments. Oceanologica Acta 12 (3): 175-187.
Garelick, H., et al. 2008. Arsenic pollution sources. Reviews of Environmental Contamination and Toxicology. https://doi.org/10.1007/978-0-387-79284-2_2.
Hach, C. 2007. DR 2800 spectrophotometer user manual. Hach Company. https://doi.org/10.3928/01477447-20101221-06.
Harrington, J.M., et al. 1998. Phase associations and mobilization of iron and trace elements in Coeur d’Alene Lake, Idaho. Environmental Science and Technology. https://doi.org/10.12693/APhysPolA.133.447.
Hatje, V., S.M. Macedo, R.M. de Jesus, G. Cotrim, K.S. Garcia, A.F. de Queiroz, and S.L.C. Ferreira. 2010. Inorganic As speciation and bioavailability in estuarine sediments of Todos os Santos Bay, BA, Brazil. Marine Pollution Bulletin 60 (12): 2225–2232. https://doi.org/10.1016/j.marpolbul.2010.08.014.
He, Y.T., A.G. Fitzmaurice, A. Bilgin, S. Choi, P. O’Day, J. Horst, J. Harrington, H. James Reisinger, D.R. Burris, and J.G. Hering. 2010. Geochemical processes controlling arsenic mobility in groundwater: a case study of arsenic mobilization and natural attenuation. Applied Geochemistry 25 (1): 69–80. https://doi.org/10.1016/j.apgeochem.2009.10.002.
Hedges, J.I., and R.G. Keil. 1995. Sedimentary organic matter preservation: an assessment and speculative synthesis. Marine Chemistry 49 (2-3): 81–115. https://doi.org/10.1016/0304-4203(95)00008-F.
Helz, G.R., and J.A. Tossell. 2008. Thermodynamic model for arsenic speciation in suldific waters: a novel use of ab initio computations. Geochimica et Cosmochimica Acta 72 (18): 4457–4468.
Huerta-Diaz, M.A., and J.W. Morse. 1992. Pyritization of trace metals in anoxic marine sediments. Geochimica et Cosmochimica Acta 56 (7): 2681–2702. https://doi.org/10.1016/0016-7037(92)90353-K.
Huerta-Diaz, M.A., A. Tessier, and R. Carignan. 1998. Geochemistry of trace metals associated with reduced sulfur in freshwater sediments. Applied Geochemistry 13 (2): 213–233. https://doi.org/10.1016/S0883-2927(97)00060-7.
Hyacinthe, C., P. Anschutz, P. Carbonel, J.M. Jouanneau, and F.J. Jorissen. 2001. Early diagenetic processes in the muddy sediments of the bay of Biscay. Marine Geology 177 (1-2): 111–128. https://doi.org/10.1016/S0025-3227(01)00127-X.
Iribarne, O., F. Botto, P. Martinetto, and J.L. Gutierrez. 2000. The role of burrows of the SW Atlantic intertidal crab Chasmagnathus granulata in trapping debris. Marine Pollution Bulletin 40 (11): 1057–1062. https://doi.org/10.1016/S0025-326X(00)00058-8.
Jørgensen, B.B., and S. Kasten. 2006. Sulfur cycling and methane oxidation. In Marine Geochemistry. https://doi.org/10.1007/3-540-32144-6_8.
Kjerfve, B. 1994. Coastal lagoons. Elsevier Oceanography Series. https://doi.org/10.1016/S0422-9894(08)70006-0.
Kneebone, P.E., P.A. O'Day, N. Jones, and J.G. Hering. 2002. Deposition and fate of arsenic in iron- and arsenic-enriched reservoir sediments. Environmental Science & Technology 36 (3): 381–386. https://doi.org/10.1021/es010922h.
Krantzberg, G. 1985. The influence of bioturbation on physical, chemical and biological parameters in aquatic environments: a review. Environmental Pollution. Series A, Ecological and Biological. https://doi.org/10.1016/0143-1471(85)90009-1.
La Force, M.J., C.M. Hansel, and S. Fendorf. 2000. Arsenic speciation, seasonal transformations and co-distribution with iron in a mine waste-influenced palustrine emergent wetland. Environmental Science & Technology 34 (18): 3937–3943. https://doi.org/10.1021/es0010150.
Lebron, I., and D.L. Suarez. 2010. Mechanisms and precipitation rate of rhodochrosite at 25°C as affected by PCO2 and organic ligands. Soil Science Society of America Journal 63 (3). https://doi.org/10.2136/sssaj1999.03615995006300030019x.
Leoni, L., and F. Sartori. 1996. Heavy metals and arsenic in sediments from the continental shelf of the Northern Tyrrhenian/Eastern Ligurian seas. Marine Environmental Research 41 (1): 73–98. https://doi.org/10.1016/0141-1136(94)00153-7.
Machado, W., R.E. Santelli, M.F. Carvalho, M.M. Molisani, R.C. Barreto, and L.D. Lacerda. 2008. Relation of reactive sulfides with organic carbon, iron, and manganese in anaerobic mangrove sediments: implications for sediment suitability to trap trace metals. Journal of Coastal Research 4: 25–32. https://doi.org/10.2112/06-0736.1.
Magalhães, M.C.F. 2007. Arsenic. An environmental problem limited by solubility. Pure and Applied Chemistry 74 (10): 1843–1850. https://doi.org/10.1351/pac200274101843.
Marangoni, J.C., and C.S.B. Costa. 2012. Short- and long-term vegetative propagation of two Spartina species on a salt marsh in southern Brazil. Estuaries and Coasts 35 (3): 763–773. https://doi.org/10.1007/s12237-011-9474-7.
Marques, W.C., E.H. Fernandes, I.O. Monteiro, and O.O. Möller. 2009. Numerical modeling of the Patos Lagoon coastal plume, Brazil. Continental Shelf Research 29 (3): 556–571. https://doi.org/10.1016/j.csr.2008.09.022.
Marques, W.C., E.H.L. Fernandes, B.C. Moraes, O.O. Möller, and A. Malcherek. 2010. Dynamics of the Patos Lagoon coastal plume and its contribution to the deposition pattern of the southern Brazilian inner shelf. Journal of Geophysical Research, Oceans 115 (10). https://doi.org/10.1029/2010JC006190.
Masscheleyn, P.H., R.D. Delaune, and W.H. Patrick. 1991. Effect of redox potential and pH on arsenic speciation and solubility in a contaminated soil. Environmental Science & Technology 25 (8): 1414–1419. https://doi.org/10.1021/es00020a008.
Masscheleyn, P.H., R.D. Delaune, and W.H. Patrick. 2010. Arsenic and selenium chemistry as affected by sediment redox potential and pH. Journal of Environmental Quality 20 (3). https://doi.org/10.2134/jeq1991.00472425002000030004x.
Mayer, L.M. 1994. Relationships between mineral surfaces and organic carbon concentrations in soils and sediments. Chemical Geology 114 (3-4): 347–363. https://doi.org/10.1016/0009-2541(94)90063-9.
Mermillod-Blondin, F. 2011. The functional significance of bioturbation and biodeposition on biogeochemical processes at the water–sediment interface in freshwater and marine ecosystems. Journal of the North American Benthological Society 30 (3): 770–778. https://doi.org/10.1899/10-121.1.
Mermillod-Blondin, F., R. Rosenberg, F. François-Carcaillet, K. Norling, and L. Mauclaire. 2004. Influence of bioturbation by three benthic infaunal species on microbial communities and biogeochemical processes in marine sediment. Aquatic Microbial Ecology 36: 271–284. https://doi.org/10.3354/ame036271.
Mirlean, N., and C.S.B. Costa. 2017. Geochemical factors promoting die-back gap formation in colonizing patches of Spartina densiflora in an irregularly flooded marsh. Estuarine, Coastal and Shelf Science 189: 104–114. https://doi.org/10.1016/j.ecss.2017.03.006.
Mirlean, N., V.E. Andrus, P. Baisch, G. Griep, and M.R. Casartelli. 2003. Arsenic pollution in Patos Lagoon estuarine sediments, Brazil. Marine Pollution Bulletin 46 (11): 1480–1484. https://doi.org/10.1016/S0025-326X(03)00257-1.
Mirlean, N., S. Medeanic, F.A. Garcia, M.P. Travassos, and P. Baisch. 2012. Arsenic enrichment in shelf and coastal sediment of the Brazilian subtropics. Continental Shelf Research 35: 129–136. https://doi.org/10.1016/j.csr.2012.01.006.
Mladenov, N., Y. Zheng, B. Simone, T.M. Bilinski, D.M. McKnight, D. Nemergut, K.A. Radloff, M.M. Rahman, and K.M. Ahmed. 2015. Dissolved organic matter quality in a shallow aquifer of Bangladesh: implications for arsenic mobility. Environmental Science & Technology 49 (18): 10815–10824. https://doi.org/10.1021/acs.est.5b01962.
Moller, O.O., et al. 1996. The Patos Lagoon summertime circulation and dynamics. Continental Shelf Research 16 (3): 335–351. https://doi.org/10.1016/0278-4343(95)00014-R.
Moller, O.O., et al. 2001. The influence of local and non-local forcing effects on the subtidal circulation of Patos Lagoon. Estuaries. 24 (2): 297. https://doi.org/10.2307/1352953.
Moore, J.N., W.H. Ficklin, and C. Johns. 1988. Partitioning of arsenic and metals in reducing sulfidic sediments. Environmental Science & Technology 22 (4): 432–437. https://doi.org/10.1021/es00169a011.
Nickson, R.T., J.M. McArthur, P. Ravenscroft, W.G. Burgess, and K.M. Ahmed. 2000. Mechanism of arsenic release to groundwater, Bangladesh and West Bengal. Applied Geochemistry 15 (4): 403–413. https://doi.org/10.1016/S0883-2927(99)00086-4.
Niencheski, L.F., H.L. Windom, and R. Smith. 1994. Distribution of particulate trace metal in Patos Lagoon estuary (Brazil). Marine Pollution Bulletin 28 (2): 96–102. https://doi.org/10.1016/0025-326X(94)90545-2.
Niencheski, L.F., W.S. Moore, and H.L. Windom. 2014. History of human activity in coastal southern Brazil from sediment. Marine Pollution Bulletin 78 (1–2): 209–212. https://doi.org/10.1016/j.marpolbul.2013.10.042.
Nóbrega, G.N., T.O. Ferreira, R.E. Romero, A.G.B. Marques, and X.L. Otero. 2013. Iron and sulfur geochemistry in semi-arid mangrove soils (Ceará, Brazil) in relation to seasonal changes and shrimp farming effluents. Environmental Monitoring and Assessment 185 (9): 7393–7407. https://doi.org/10.1007/s10661-013-3108-4.
Nordstrom, D.K. 2002. Worldwide occurrences of arsenic in ground water. In Science compass. https://doi.org/10.1126/science.1072375.
O’Day, P.A., et al. 2004. The influence of sulfur and iron on dissolved arsenic concentrations in the shallow subsurface under changing redox conditions. Proceedings of the National Academy of Sciences 101 (38): 13703–13708. https://doi.org/10.1073/pnas.0402775101.
Otero, X.L., and F. Macias. 2003. Spatial variation in pyritization of trace metals in salt-marsh soils. Biogeochemistry. 62 (1): 59–86. https://doi.org/10.1023/A:1021115211165.
Pearcy, R.W., and S.L. Ustin. 1984. Effects of salinity on growth and photosynthesis of three California tidal marsh species. Oecologia. 62 (1): 68–73. https://doi.org/10.1007/BF00377375.
Pierce, M.L., and C.B. Moore. 1982. Adsorption of arsenite and arsenate on amorphous iron hydroxide. Water Research. https://doi.org/10.1016/0043-1354(82)90143-9.
Planer-Friedrich, B., J. London, R.B. McCleskey, D.K. Nordstrom, and D. Wallschläger. 2007. Thioarsenates in geothermal waters of Yellowstone National Park: Determination, preservation, and geochemical importance. Environmental Science & Technology 41 (15): 5245–5251.
Presley, B.J., R.J. Taylor, and P.N. Boothe. 1992. Trace metal concentrations in sediments of the Eastern Mississippi Bight. Marine Environmental Research 33 (4): 267–282. https://doi.org/10.1016/0141-1136(92)90142-9.
Ravenscroft, P., H. Brammer, and K. Richards. 2009. Arsenic pollution: a global synthesis. Chichester: Wiley.
Rickard, D., and G.W. Luther. 2007. Chemistry of iron sulfides. Chemical Reviews 107 (2): 514–562. https://doi.org/10.1021/cr0503658.
Rickard, D., and J.W. Morse. 2005. Acid volatile sulfide (AVS). Marine Chemistry 97 (3-4): 141–197. https://doi.org/10.1016/j.marchem.2005.08.004.
Riedel, G.F., J.G. Sanders, and R.W. Osman. 1987. The effect of biological and physical disturbances on the transport of arsenic from contaminated estuarine sediments. Estuarine, Coastal and Shelf Science 25 (6): 693–706. https://doi.org/10.1016/0272-7714(87)90016-3.
Rittle, K.A., J.I. Drever, and P.J.S. Colberg. 1995. Precipitation of arsenic during bacterial sulfate reduction. Geomicrobiology Journal 13 (1): 1–11. https://doi.org/10.1080/01490459509378000.
Roman, C.T., W.A. Niering, and R.S. Warren. 1984. Salt marsh vegetation change in response to tidal restriction. Environmental Management 8 (2): 141–149. https://doi.org/10.1007/BF01866935.
Rosa, L.C., and C.E. Bemvenuti. 2005. Effects of the burrowing crab Chasmagnathus granulata (Dana) on meiofauna of estuarine intertidal habitats of Patos Lagoon, Southern Brazil. Brazilian Archives of Biology and Technology 48 (2): 267–274. https://doi.org/10.1590/S1516-89132005000200014.
Roy, M., J.B. Martin, J. Cherrier, J.E. Cable, and C.G. Smith. 2010. Influence of sea level rise on iron diagenesis in an east Florida subterranean estuary. Geochimica et Cosmochimica Acta 74 (19): 5560–5573.
Roy, M., J.B. Martin, C.G. Smith, and J.E. Cable. 2011. Reactive-transport modeling of iron diagenesis and associated organic matter mineralization in a Florida (USA) subterranean estuary. Earth and Planetary Science Letters 304 (1-2): 191–201.
Sá, F., C.J. Sanders, S.R. Patchineelam, E.C. Machado, and A.T. Lombardi. 2015. Arsenic fractionation in estuarine sediments: Does coastal eutrophication influence As behavior? Marine Pollution Bulletin 96 (1-2): 496–501. https://doi.org/10.1016/j.marpolbul.2015.04.037.
Sailo, L., and C. Mahanta. 2014. Arsenic mobilization in the Brahmaputra plains of Assam: groundwater and sedimentary controls. Environmental Monitoring and Assessment 186 (10): 6805–6820. https://doi.org/10.1007/s10661-014-3890-7.
Secrieru, D. and G. Oaie 2009. The relation between the grain size composition of the sediments from the NW Black Sea and their Total Organic Cabbon (ToC) content. Geo-Eco-Marina 15 (1): 5-11.
Sharma, V.K., and M. Sohn. 2009. Aquatic arsenic: Toxicity, speciation, transformations, and remediation. Environment International 35 (4): 743–759. https://doi.org/10.1016/j.envint.2009.01.005.
Shimada, N. 1996. Geochemical conditions enhancing the solubilization of arsenic into groundwater in Japan. Applied Organometallic Chemistry 10 (9): 667–674. https://doi.org/10.1002/(SICI)1099-0739(199611)10:9<667::AID-AOC545>3.0.CO;2-I.
Smedley, P.L., and D.G. Kinniburgh. 2002. A review of the source, behaviour and distribution of arsenic in natural waters. Applied Geochemistry 17 (5): 517–568. https://doi.org/10.1016/S0883-2927(02)00018-5.
Smith, A.H., E.O. Lingas, and M. Rahman. 2000. Contamination of drinking-water by arsenic in Bangladesh: a public health emergency. Bulletin of the World Health Organization. https://doi.org/10.1590/S0042-96862000000900005.
Smith, E., J. Smith, and R. Naidu. 2006. Distribution and nature of arsenic along former railway corridors of South Australia. The Science of the Total Environment 363 (1-3): 175–182. https://doi.org/10.1016/j.scitotenv.2005.05.039.
Stucker, V.K., D.R. Silverman, K.H. Williams, J.O. Sharp, and J.F. Ranville. 2014. Thioarsenic species associated with increased arsenic release during biostimulated subsurface sulfate reduction. Environmental Science & Technology 48 (22): 13367–13375.
Sullivan, K.A., and R.C. Aller. 1996. Diagenetic cycling of arsenic in Amazon shelf sediments. Geochimica et Cosmochimica Acta 60 (9): 1465–1477. https://doi.org/10.1016/0016-7037(96)00040-3.
Telfeyan, K., A. Breaux, J. Kim, J.E. Cable, A.S. Kolker, D.A. Grimm, and K.H. Johannesson. 2017. Arsenic, vanadium, iron, and manganese biogeochemistry in a deltaic wetland, southern Louisiana, USA. Marine Chemistry 192: 32–48. https://doi.org/10.1016/j.marchem.2017.03.010.
USEPA, U. S. E. P. A. 1991. Draft analytical method for determination of acid volatile sulfide in sediment. EPA-821-R-91-100, Washington, DC.
USEPA, U. S. E. P. A 1996. Method 3050B - acid digestion of sediments, sludges, and soils., 1996. https://doi.org/10.1117/12.528651.
Van Cappellen, P., and Y. Wang. 1996. Cycling of iron and manganese in surface sediments: a general theory for the coupled transport and reaction of carbon, nitrogen, sulfur, iron, and managense. American Journal of Science 296 (3): 197–243.
Vidal-Durà, A., I.T. Burke, D.I. Stewart, and R.J.G. Mortimer. 2018. Reoxidation of estuarine sediments during simulated resuspension events: effects on nutrient and trace metal mobilisation. Estuarine, Coastal and Shelf Science 207: 40–55. https://doi.org/10.1016/j.ecss.2018.03.024.
Wallner-Kersanach, M., N. Mirlean, M.G.Z. Baumgarten, L.D.F. Costa, and P. Baisch. 2016. Temporal evolution of the contamination in the southern area of the Patos Lagoon estuary, RS, Brazil. Revista de Gestão Costeira Integrada 16 (3): 263–279. https://doi.org/10.5894/rgci596.
Wang, S., L. Xu, Z. Zhao, S. Wang, Y. Jia, H. Wang, and X. Wang. 2012. Arsenic retention and remobilization in muddy sediments with high iron and sulfur contents from a heavily contaminated estuary in China. Chemical Geology 314-317: 57–65. https://doi.org/10.1016/j.chemgeo.2012.05.005.
Welch, A.H., D.B. Westjohn, D.R. Helsel, and R.B. Wanty. 2000. Arsenic in ground water of the United States: occurrence and geochemistry. Ground Water 38 (4): 589–604. https://doi.org/10.1111/j.1745-6584.2000.tb00251.x.
Widerlund, A., and J. Ingri. 1995. Early diagenesis of arsenic in sediments of the Kalix River estuary, northern Sweden. Chemical Geology 125 (3-4): 185–196. https://doi.org/10.1016/0009-2541(95)00073-U.
Windom, H.L., L.F. Niencheski, and R.G. Smith. 1999. Biogeochemistry of nutrients and trace metals in the estuarine region of the Patos lagoon (Brazil). Estuarine, Coastal and Shelf Science 48 (1): 113–123. https://doi.org/10.1006/ecss.1998.0410.
Yang, N., Z. Shen, S. Datta, and K.H. Johannesson. 2016. High arsenic (As) concentrations in the shallow groundwaters of southern Louisiana: Evidence of microbial controls on As mobilization from sediments. Journal of Hydrology: Regional Studies. 5: 100–113. https://doi.org/10.1016/j.ejrh.2015.11.023.
Ying, S.C., B.D. Kocar, and S. Fendorf. 2012. Oxidation and competitive retention of arsenic between iron- and manganese oxides. Geochimica et Cosmochimica Acta 96: 294–303. https://doi.org/10.1016/j.gca.2012.07.013.
Yücel, M., S.K. Konovalov, T.S. Moore, C.P. Janzen, and G.W. Luther III. 2010. Sulfur speciation in the upper Black Sea sediments. Chemical Geology 269 (3-4): 364–375. https://doi.org/10.1016/j.chemgeo.2009.10.010.
Acknowledgments
This study was supported by grant from “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil” (CAPES) as well as funds made available by Michael and Mathilda Cochran through the Cochran Family Professorship at Tulane University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Wen-Xiong Wang
Rights and permissions
About this article
Cite this article
Costa, L., Mirlean, N., Quintana, G. et al. Distribution and Geochemistry of Arsenic in Sediments of the World’s Largest Choked Estuary: the Patos Lagoon, Brazil. Estuaries and Coasts 42, 1896–1911 (2019). https://doi.org/10.1007/s12237-019-00596-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-019-00596-0