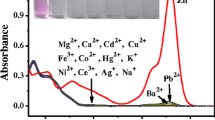
Abstract
We report the design and synthesis of a novel chemosensor (RGPS) rhodamine-based indicator for selective detection of Cu2+ ion. Sensing behavior toward various metal ions (M=Hg2+, Cu2+, Co2+, K+, Cs+, Ag+, Pb2+, Zn2+, Mg2+, Fe3+, Ni2+, Li+ and Al3+) was investigated by UV-Vis and fluorescence spectroscopy in acetonitrile:water (CH3CN-H2O, v/v, 9:1) solution. The indicator exhibited highly selective and sensitive colorimetric and “turn-on” fluorescent responses toward Cu2+ ions based on the ring-opening mechanism of the rhodamine spirolactam in an CH3CN-H2O (v/v, 9:1) solution. The obvious change from colorless to pale pink upon the addition of Cu2+ could make it a suitable “naked eye” indicator for Cu2+. More significantly, the sensor displayed a remarkable colorless to yellowish green fluorescence switch in the presence of Cu2+ ions. The limit of detection is proved 1.405×10−7 M from titration and 1:1 binding ratio is proved from jobs plot method. Finally, we proposed a reversible ring-opening mechanism (OFF-ON) of the rhodamine spirolactam induced by Cu2+ binding and forming a 1:1 stoichiometric complex of RGPS-Cu2+ which is supported by the material studio theory calculations.
Similar content being viewed by others
References
A. L. Sommer, Plant Physiol., 6, 339 (1931).
U. Ricardo, O. Manuel, and G. Mauricio, Am. J. Clin. Nutr., 67, 9525 (1998).
E. J. Underwood in “Copper in Trace Elements in Humans and Animals Nutrition”, 4th ed. (E. J. Underwood), pp.56–233, Academic Press New York, 1971.
R. R. Crichton, D. T. Dexter, and R. J. Warc, Coord. Chem. Rev., 252, pp252–1189 (2008).
S. Adrian in “US Environmental Protection Agency”, Washington, DC, USA, 1996.
S. Kousar and M. Javed, Int. J. Agric. Biol., 14, 801 (2012).
T. Gomes, C. G. Pereira, C. Cardoso, J. P. Pinheiro, I. Cancioand, M. J. BebianAquat.Toxicol., 72, 118 (2012)
E. J. Jun, H. N. Won, J. S. Kim, K. H. Lee, and J. Yoon, Tetrahedron Lett., 47, 4577 (2006).
Z. Xu, J. Yoon, and D. R. Spring, Chem. Commun., 46, 2563 (2010).
Z. Xu, J. Pan, D. R. Spring, J. Cui, and J. Yoon, Tetrahedron, 66, 1678 (2010).
J. H. Jung, M. H. Lee, H. J. Kim, H. S. Jung, S. Y. Lee, N. R. Shin, K. No, and J. S. Kim, Tetrahedron Lett., 50, 2013 (2009).
D. Maity, D. Karthigeyan, T. K. Kundu, and T. Govindaraju, Sens. Actuator B-Chem., 176, 831 (2013).
T. W. Lin and S. D. Huang, Anal. Chem., 73, 4319 (2001).
N. Pourreza and R. Hoveizavi, Anal. Chim. Acta, 549, 124 (2005).
Y. Lin, P. Liang, and L. Guo, Talanta, 68, 25 (2005).
J. S. Becker, M. V. Zoriy, C. Pickhardt, G. N. Palomer, and K. Zilles, Anal. Chem., 77, 3208 (2005).
L. Shi, C. Jing, W. Ma, D. W. Li, J. E. Halls, F. Marken, and Y. T. Long, Angew. Chem.-Int. Edit., 125, 6127 (2013).
T. Shtoyko, S. Conklin, A. T. Maghasi, J. N. Richardson, A. Piruska, C. J. Seliskar, and W. R. Heineman, Anal. Chem., 76, 1466 (2004).
A. A. Ensafi, T. Khayamiam, A. Benvidi, and E. Mirmomtaz, Anal. Chim. Acta, 251, 225 (2006).
K. P. Prathish, D. James, J. Jaisy, and T. P. Rao, Anal. Chim. Acta, 647, 84 (2009).
S. Bhattachaya and M. Thomas, Tetrahedron Lett., 41, 10313 (2000).
X. B. Zhang, J. Peng, C. L. He, G. L. Shen, and R. Q. Yu, Anal. Chim. Acta, 567, 189 (2006).
O. Oter, K. Ertekin, C. Kirilmis, and M. Koca, Anal. Chim. Acta, 584, 308 (2007).
Y. K. Jang, U. C. Nam, H. L. Kwon, I. H. Hwang, and C. Kim, Dyes Pigment., 99, 6 (2013).
C. X. Yin, L. J. Qua, and F. J. Huo, Chinese Chem. Lett., 25, 1230 (2014).
N. Aksuner, E. Henden, I. Yilmaz, and A. Cukurovali, Dyes Pigment., 83, 217 (2009).
L. Zhang, J. Fan, and X. Peng, Spectrochim. Acta A, 73, 398 (2009).
X. Chen, T. Pradhan, F. Wang, J. S. Kim, and J. Yoon, Chem. Rev., 112, 1910 (2012).
D. T. Quang, J. S. Wu, N. D. Luyen, T. Duong, N. D. Dan, N. C. Bao, and P. T. Quy, Spectrochim. Acta A, 78, 753 (2011).
V. B. Bojinov, A. I. Venkova, and N. I. Georgiev, Sens. Actuator B-Chem., 143, 42 (2009).
L. F. Zhang, J. L. Zhao, X. Zeng, L. Mu, X. K. Jiang, M. Deng, J. X. Zhang, and G. Wei, Sens. Actuator B-Chem., 160, 662 (2011).
Y. Ma, X. Zhang, Z. Guan, Y. Cao, and J. Yao, J. Mater. Res., 16, 2928 (2001).
D. Maity, D. Karthigeyan, T. K. Kundu, and T. Govindaraju, Sens. Actuator B-Chem., 176, 831 (2013).
S. Y. Lee, H. J. Kim, J. S. Wu, K. No, and J. S. Kim, Tetrahedron Lett., 49, 6141 (2008).
Z. Xu, L. Zhang, R. Guo, T. Xiang, C. Wu, Z. Zhang, and F. Yang, Sens. Actuator B-Chem., 156, 546 (2011).
K. M. K. Swamy, S. K. Ko, S. K. Kwon, H. N. Lee, C. Mao, J. M. Kim, K. H. Lee, J. Kim, I. Shin, and J. Yoon, Chem. Commun., 5915 (2008).
J. F. Zhang, Y. Zhou, J. Yoon, Y. Kim, S. J. Kim, and S. H. Kim, Org. Lett., 12, 3852 (2010).
Z. Yang, M. She, J. Zhang, X. Chen, Y. Huang, H. Zhu, P. Liu, J. Li, and Z. Shi, Sens. Actuator B-Chem., 176, 482 (2013).
F. Huo, L. Wang, C. Yin, Y. Yang, H. Tong, J. Chao, and Y. Zhang, Sens. Actuator B-Chem., 188, 735 (2013).
A. R. Boddu, H. Kim, and Y. A. Son, Sens. Actuator BChem., 188, 847 (2013).
S. H. Lee, A. R. Boddu, and Y. A. Son, Sens. Actuator BChem., 196, 388 (2014).
H. S. So, A. R. Boddu, J. Hwang, K. Yesudas, and Y. A. Son, Sens. Actuator B-Chem., 202, 779 (2014).
H. Kim, S. H. Kim, D. H. Lee, and Y. A. Son, Supramol. Chem., 25, 87 (2013).
J. W. Jeong, A. R. Boddu, and Y. A. Son, Sens. Actuator BChem., 208, 75 (2015).
H. Kim, A. R. Boddu, J. W. Jeong, S. Mallick, S. M. Kang, J. S. Choi, C. S. Lee, Y. A. Son, Sens. Actuator B-Chem. (2015), DOI: http://dx.doi.org/doi: 10.1016/j.snb.2014.12.100.
S. H. Lee, A. R. Boddu, and Y. A. Son, Sens. Actuator BChem. (2015), DOI: http://dx.doi.org/doi:10.1016/j.snb.2015. 01.008.
Y. A. Son and J. Park, Textile Color. Finish., 24, 158 (2012).
A. R. Boddu, J. Y. Lee, and Y. A. Son, Supramol. Chem., 27, 191 (2015).
A. R. Boddu, J. Y. Lee, and Y. A. Son, Spectrochim. Acta A, 127, 268 (2014).
H. Kim, A. R. Boddu, and Y. A. Son, Fiber. Polym., 14, 2010 (2013).
E. M. Lee, S. Y. Gwon, Y. A. Son, and S. H. Kim, Spectrochim. Acta A, 95, 25 (2012).
M. W. Beukers, M. J. Wanner, J. K. V. F. Drabbe Kunzel, E. C. Klaasse, A. P. IJzerman, and G.-J. Koomen, J. Med. Chem., 46, 1492 (2003).
Z. Zhang, Y. Zheng, W. Hang, X. Yan, and Y. Zhao, Talanta, 85, 779 (2011).
H. Li, J. Fan, F. Song, H. Zhu, J. Du, S. Sun, and X. Peng, Chem. Eur. J., 16, 12349 (2010).
P. MacCarthy, Anal. Chem., 50, 2165 (1978).
Y. Zhou, F. Wang, Y. M. Kim, S. J. Kim, and J. Y. Yoon, Org. Lett., 11, 4442 (2009).
B. Delley, J. Chem. Phys., 92, 508 (1990).
B. Delley, J. Chem. Phys., 113, 7756 (2000).
A. D. Boese and N. C. Handy, J. Chem. Phys., 114, 5497 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, J., Rao, B.A. & Son, YA. “Turn-on” fluorescent and colorimetric determination of Cu2+ ions in aqueous media based on a Rhodamine-N-phenyl Semicarbazide derivative. Fibers Polym 16, 953–960 (2015). https://doi.org/10.1007/s12221-015-0953-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12221-015-0953-5