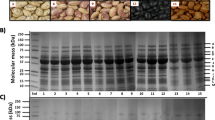
Abstract
Trypsin inhibitor activity (TIA) in soybean is attributed to two polypeptides, namely, Kunitz trypsin inhibitor (KTI) and Bowman-Birk inhibitor (BBI). Standard spectrophotometric protocol widely followed for estimation of TIA is cumbersome and does not distinguish KTI from BBI. In the present investigation, extraction conditions for KTI were optimized and different forms of this polypeptide were resolved in 180 soybean genotypes of Indian and exotic origin through native PAGE. This led to the identification of three KTI alleles, namely, Tia, Tib, and Tic, with Tia occurring in most of the Indian genotypes. Trypsin-KTI complex assay exhibited binding of Tia polypeptide with 2.51 fold concentration of trypsin. Subsequently, seeds of selected genotypes were subjected to estimation of KTI and BBI activity through densitometry and enzyme-linked immunosorbent assay (ELISA), respectively; and total TIA through standard spectrophotometric protocol. Summation of KTI and BBI was significantly (P < 0.05) lower than that of TIA determined through the spectrophotometric method.



Similar content being viewed by others
References
Aviles-Gaxiola S, Chuck-Hernandez C, Serna Saldivar SO (2018) Inactivation methods of trypsin inhibitor in legumes: a review. J Food Sci 83(1):17–29. https://doi.org/10.1111/1750-3841.13985
Brandon DL, Bates AH, Friedman M (1991) ELISA analysis of soybean trypsin inhibitors in processed foods. Adv Exp Med Biol 289:321–337. https://doi.org/10.1007/978-1-4899-2626-5
Brandon DL, Bates AH, Friedman M (2004) Immunoassays for Bowman–Birk and Kunitz soybean trypsin inhibitors in infant formula. J Food Sci 69(1):vi–67. https://doi.org/10.1111/j.1365-2621.2004.tb17849.x
Brune MFSS, Pinto MO, Peluzio MCG, Moreira MA, Barros EG (2010) Biochemical and nutritional evaluation of a free soybean line Kunitz trypsin inhibitor and lectins. Food Sci Technol 30(3):657–663. https://doi.org/10.1590/s0101-20612010000300014
Chen Y, Xu Z, Zhang C, Kong X, Hua Y (2014) Heat-induced inactivation mechanisms of Kunitz trypsin inhibitor and Bowman-Birk inhibitor in soymilk processing. Food Chem 154:108–116
Cucu T, Deverese B, Kerkaert B, Rogge M, Vercruysse L, De Meulenae B (2012) ELISA based detection of soybean proteins: a comparative study using antibodies against modified and native proteins. Food Anal Methods 5:1121–1130. https://doi.org/10.1007/s1261-011-9341-y
Galati G, Brien PJO (2004) Potential toxicity of flavonoids and other dietary phenolics: significance for their chemo preventive and anticancer properties. Free Radic Biol Med 37(3):287–303. https://doi.org/10.1016/j.freeradbiomed.2004.04.034
Hammerstrand GE, Black LT, Glover JD (1981) Trypsin inhibitors in soy products. Modification of the standard analytical procedure. Cereal Chem 58(1):42–45
Hymowitz T, Hadly HH (1972) Inheritance of trypsin inhibitor variant in seed protein of soybean. Crop Sci 12(2):197–198. https://doi.org/10.1086/336690
Kakade ML, Simons N, Liener IE (1969) An evaluation of natural VS synthetic substrates for measuring antitryptic activity of soybean samples. Soybeans: natural vs synthetic substrates 46:518–526
Kim SH, Hara S, Hase S, Ikenaka T, Toda H, Kitampura K, Kaizuma N (1985) Comparative study on amino acid sequences of Kunitz-type soybean trypsin inhibitors, Ti a, Ti b, and Ti c. J Biochem 98(2):435–448
Liener IE (1994) Implications of anti-nutritional components in soybean foods. J Critical Rev in Food Sci Nutr 34:31–67. https://doi.org/10.1086/336690
Liu K, Markakis P (1989) An improved colorimetric method for determining antitryptic activity in soybean products. Cereal Chem 66(5):415–422
Pesic MB, Vacelic-Radovic BV, Barac MB, Stanojevic SP, Nedovie VA (2007) Influence of different genotype on trypsin inhibitor levels and activity in soybeans. Sensors 7:67–74
Onesti S, Brick P, Blow DM (1991) Crystal structure of a Kunitz-type trypsin inhibitor from Erythrina caffra seeds. J Mol Bio 217(1):153–176. https://doi.org/10.1016/00222836(91)90618-G153-176
Orf JH, Hymowitz T (1977) Inheritance of a second trypsin inhibitor variant in seed protein of soybeans. Crop Sci 17(5):811–813
Peric V, Srebric M, Dragicevic V, Nikolic A, Mikic A, Drinic SM (2014) Development of soybean varieties with specific nutritional composition of grain. J Hyg Eng Design 8:174–177
Pauchar-Menacho LM, Berhow MA, Mandarino JMG, Gonzalez DE, Mejia E, Chang YK (2010) Optimization of germination time and temperature on the concentration of bioactive compounds in Brazilian soybean cultivar BRS 133 using response surface methodology. Food Chem 119:636–642. https://doi.org/10.1016/j.foodchem.2009.07.011
Vagadia BH, Vanga SK, Raghavan VSS (2017) Inactivation methods of soybean trypsin inhibitor—a review. Trends in Food Sci Tech 64:115–125. https://doi.org/10.2016/j.tifs.2017.02.003
Wang KJ, Takahata Y, Kono Y, Kaizuma N (2008) Allelic differentiation of Kunitz trypsin inhibitor in wild soybean (Glycine soja). Theor Appl Genet 117:565–573. https://doi.org/10.1007/s00122-008-0800-9
Zhou T, Han S, Li Z, He P, (2017) Purification and quantification of Kunitz trypsin inhibitor in soybean using two-dimensional liquid chromatography. Food Anal Methods doi:https://doi.org/10.1007/s1216-017-0902-6
Funding
This work was supported by Food Safety and Standards Authority of India (FSSAI), Ministry of Health and Family Welfare, Government of India, under Sanction No. F.No.65/R&D/Soybean/RARD/2016/FSSAI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Vineet Kumar declares that he has no conflict of interest. Anita Rani declares that she has no conflict of interest. Mohd Shuaib declares that he has no conflict of interest. Priyanka Mittal declares that she has no conflict of interest.
Ethical Approval
This article does not contain any studies involving human participants or animals performed by any of the authors.
Informed Consent
Not applicable as this study does not include any human participants.
Rights and permissions
About this article
Cite this article
Kumar, V., Rani, A., Shuaib, M. et al. Comparative Assessment of Trypsin Inhibitor vis-à-vis Kunitz Trypsin Inhibitor and Bowman-Birk Inhibitor Activities in Soybean. Food Anal. Methods 11, 2431–2437 (2018). https://doi.org/10.1007/s12161-018-1227-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1227-9