Abstract
Objective
In preclinical studies, high-throughput positron emission tomography (PET) imaging, known as simultaneous multiple animal scanning, can reduce the time spent on animal experiments, the cost of PET tracers, and the risk of synthesis of PET tracers. It is well known that the image quality acquired by high-throughput imaging depends on the PET system. Herein, we investigated the influence of large field of view (FOV) PET scanner on high-throughput imaging.
Methods
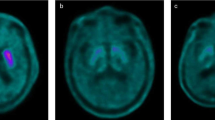
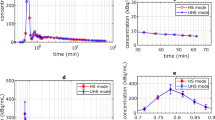
We investigated the influence of scanning four objects using a small animal PET scanner with a large FOV. We compared the image quality acquired by four objects scanned with the one acquired by one object scanned using phantoms and animals. We assessed the image quality with uniformity, recovery coefficient (RC), and spillover ratio (SOR), which are indicators of image noise, spatial resolution, and quantitative precision, respectively. For the phantom study, we used the NEMA NU 4-2008 image quality phantom and evaluated uniformity, RC, and SOR, and for the animal study, we used Wistar rats and evaluated the spillover in the heart and kidney.
Results
In the phantom study, four phantoms had little effect on imaging quality, especially SOR compared with that for one phantom. In the animal study as well, four rats had little effect on spillover from the heart muscle and kidney cortex compared with that for one rat.
Conclusions
This study demonstrated that an animal PET scanner with a large FOV was suitable for high-throughput imaging. Thus, the large FOV PET scanner can support drug discovery and bridging research through rapid pharmacological and pathological evaluation.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during this study are available from the corresponding author on reasonable request.
Abbreviations
- PET:
-
Positron emission tomography
- FOV:
-
Field of view
- RI:
-
Radioisotope
- DOI:
-
Depth of interaction
- PSF:
-
Point spread function
- LGSO:
-
Lutetium gadolinium oxy orthosilicate
- 3D:
-
Three dimensional
- DRAMA:
-
Dynamic row-action maximum likelihood algorithm
- NEMA:
-
National Electrical Manufacturers Association
- NU4IQ:
-
NU 4-2008 image quality
- STD:
-
Standard deviation
- ROI:
-
Region of interest
- VOI:
-
Volume of interest
- RC(s):
-
Recovery coefficient(s)
- SOR(s):
-
Spillover ratio(s)
- [18F] BCPP-BF:
-
2-tert-butyl-4-chloro-5-[6-(4-18F-fluorobutoxy)-pyridin-3-ylmethoxy]-2H-pyridazin-3-one
- LW:
-
Left heart wall
- AS:
-
Atrial septum
References
Visser EP, Disselhorst JA, Brom M, Laverman P, Gotthardt M, Oyen WJG, et al. Spatial resolution and sensitivity of the Inveon small-animal PET scanner. J Nucl Med. 2009;50(1):139–47.
Kitamura K. Development of “Clairvivo PET” small animal PET scanner. Shimadzu Hyoron. 2008;64(3–4):107–15.
Nagy K, Tóth M, Major P, Patay G, Egri G, Häggkvist J, et al. Performance evaluation of the small-animal nanoscan PET/MRI system. J Nucl Med. 2013;54(10):1825–32.
Cheng TE, Yoder KK, Normandin MD, Risacher SL, Converse AK, Hampel JA, et al. A rat head holder for simultaneous scanning of two rats in small animal PET scanners: design, construction, feasibility testing and kinetic validation. J Neurosci Methods. 2009;176(1):24–33.
Aide N, Desmonts C, Briand M, Meryet-Figuiere M, Poulain L. High-throughput small animal PET imaging in cancer research: evaluation of the capability of the Inveon scanner to image four mice simultaneously. Nucl Med Commun. 2010;31(10):851–8.
Siepel FJ, Van Lier MGJTB, Chen M, Disselhorst JA, Meeuwis APW, Oyen WJG, et al. Scanning multiple mice in a small-animal PET scanner: influence on image quality. Nucl Instrum Methods Phys Res Sect A Accel Spectrom Detect Assoc Equip. 2010;621(1–3):605–10.
Aide N, Desmonts C, Beauregard JM, Beyer T, Kinross K, Roselt P, et al. High throughput static and dynamic small animal imaging using clinical PET/CT: potential preclinical applications. Eur J Nucl Med Mol Imaging. 2010;37(5):991–1001.
Efthimiou N, Wright JD, Clayton L, Renard I, Zagni F, Caribé PRRV, et al. Influence of multiple animal scanning on image quality for the sedecal superArgus2R preclinical PET scanner. Front Phys. 2021. https://doi.org/10.3389/fphy.2020.531662.
Greenwood HE, Nyitrai Z, Mocsai G, Hobor S, Witney TH. High-throughput PET/CT imaging using a multiple-mouse imaging system. J Nucl Med. 2020;61(2):292–7.
Kyushima H, Shimoi H, Atsumi A, Ito M, Oba K, Yoshizawa Y. The development of flat panel PMT. In: Proc. IEEE nuclear imaging conf. rec.; 2000. pp 15–20
Ota R. Photon counting detectors and their applications ranging from particle physics experiments to environmental radiation monitoring and medical imaging. Radiol Phys Technol. 2021;14(2):134–48.
Badawi R, Marsden P. Developments in component-based normalization for 3D PET. Phys Med Biol. 1999;44(2):571–94.
Tanaka E, Kudo H. Subset-dependent relaxation in block-iterative algorithms for image reconstruction in emission tomography. Phys Med Biol. 2003;48:1405.
Panin VY, Kehren F, Michel C, Casey M. Fully 3-D PET reconstruction with system matrix derived from point source measurements. IEEE Trans Med Imaging. 2006;25(7):907–21.
Sato K, Shidahara M, Watabe H, Watanuki S, Ishikawa Y, Arakawa Y, et al. Performance evaluation of the small-animal PET scanner ClairvivoPET using NEMA NU 4–2008 standards. Phys Med Biol. 2015;61(2):696–711.
Yu AR, Kim JS, Kang JH, Lim SM. Comparison of reconstruction methods and quantitative accuracy in Siemens Inveon PET scanner. J Instrum. 2015. https://doi.org/10.1088/1748-0221/10/04/P04001.
National Electrical Manufacturers Association (NEMA). NEMA standards publication NU 4–2008. Rosslyn: National Electrical Manufacturers Association; 2008.
Henze E, Huang SC, Ratib O, Hoffman E, Phelps ME, Schelbert HR. Measurements of regional tissue and blood-pool radiotracer concentrations from serial tomographic images of the heart. J Nucl Med. 1983;24(11):987–96.
Herrero P, Kim J, Sharp TL, Engelbach JA, Lewis JS, Gropler RJ, et al. Assessment of myocardial blood flow using 15 O-water and 1–11 C-acetate in rats with small-animal PET. J Nucl Med. 2006;47(3):477–85.
Zheng X, Tian G, Huang SC, Feng D. A hybrid clustering method for ROI delineation in small-animal dynamic PET images: application to the automatic estimation of FDG input functions. IEEE Trans Inf Technol Biomed. 2011;15(2):195–205.
Harada N, Nishiyama S, Kanazawa M, Tsukada H. Development of novel PET probes, [18F]BCPP-EF, [18F]BCPP-BF, and [11C]BCPP-EM for mitochondrial complex 1 imaging in the living brain. J Label Compd Radiopharm. 2013;56(11):553–61.
Tsukada H, Nishiyama S, Fukumoto D, Kanazawa M, Harada N. Novel PET probes 18F-BCPP-EF and 18F-BCPP-BF for mitochondrial complex I: a PET study in comparison with 18F-BMS-747158-02 in rat brain. J Nucl Med. 2014;55(3):473–80.
Ohba H, Kanazawa M, Kakiuchi T, Tsukada H. Effects of acetaminophen on mitochondrial complex I activity in the rat liver and kidney: a PET study with 18F-BCPP-BF. EJNMMI Res. 2016;6(1):82.
Wiant D, Gersh JA, Bennett M, Bourland JD. Evaluation of the spatial dependence of the point spread function in 2D PET image reconstruction using LOR-OSEM. Med Phys. 2010;37(3):1169–82.
Murata T, Miwa K, Miyaji N, Wagatsuma K, Hasegawa T, Oda K, et al. Evaluation of spatial dependence of point spread function-based PET reconstruction using a traceable point-like 22Na source. EJNMMI Phys. 2016. https://doi.org/10.1186/s40658-016-0162-3.
Kidera D, Kihara K, Akamatsu G, Mikasa S, Taniguchi T, Tsutsui Y, et al. The edge artifact in the point-spread function-based PET reconstruction at different sphere-to-background ratios of radioactivity. Ann Nucl Med. 2016;30(2):97–103.
Tsutsui Y, Awamoto S, Himuro K, Umezu Y, Baba S, Sasaki M. Edge artifacts in point spread function-based PET reconstruction in relation to object size and reconstruction parameters. Asia Ocean J Nucl Med Biol. 2017;5(2):134–43.
Ríha H, Papoušek F, Neckár J, Pirk J, Oštádal B. Effects of isoflurane concentration on basic echocardiographic parameters of the left ventricle in rats. Physiol Res. 2012;61(4):419–23.
Buys-Gonçalves GF, Sampaio FJB, Pereira-Sampaio MA, Silva MEM, De Souza DB. Histomorphometric and biochemical data of rat kidney submitted to warm ischemia associated with resveratrol treatment. Data Brief. 2020;30:105545.
Acknowledgements
We would like to thank Masakatsu Kanazawa for PET probe synthesis and Dai Fukumoto for the animal study. All members are employees of Hamamatsu Photonics K.K. We all have no control over this work by the company and declare no conflict of interest.
Author information
Authors and Affiliations
Contributions
YT designed this study, did the phantom and animal studies, analyzed the data, and wrote the manuscript draft. YO did the phantom study and wrote the manuscript draft. FH designed this study and advised on analysis. KO advised on study design. TO oversaw the system description. HO did the phantom study and advised on study design. All the authors read, revised, and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This animal study was approved with permission No. HPK-2022-17 by the Institutional Animal Care and Use Committee at the Hamamatsu Photonics.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tomonari, Y., Onishi, Y., Hashimoto, F. et al. Animal PET scanner with a large field of view is suitable for high-throughput scanning of rodents. Ann Nucl Med (2024). https://doi.org/10.1007/s12149-024-01937-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12149-024-01937-1