Abstract
Objective
This study aimed to compare the diagnostic value of 123I-metaiodobenzylguanidine (MIBG) myocardial scintigraphy, N-isopropyl-p[123I]iodoamphetamine (IMP) brain perfusion single-photon emission computed tomography (SPECT), and brain magnetic resonance imaging (MRI) voxel-based morphometry (VBM) for the differentiation of dementia with Lewy bodies (DLB) and Alzheimer’s disease (AD).
Methods
Thirty-five and 34 patients with probable DLB and probable AD, respectively, were enrolled. All patients underwent 123I-MIBG myocardial scintigraphy, 123I-IMP brain perfusion SPECT, and brain MRI. For 123I-MIBG imaging, we calculated early and delayed heart-to-mediastinum (H/M) uptake ratios. Three-dimensional stereotactic surface projections (3D-SSP) were used to analyze the results of 123I-IMP SPECT. VBM with statistical parametric mapping 8 plus diffeomorphic anatomical registration using exponentiated Lie algebra (DARTEL) was used to analyze the brain MRI data.
Results
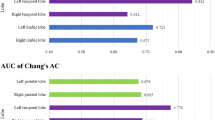
The area under the receiver operating characteristic curves (AUC) for discriminating DLB and AD was highest (0.882) for the delayed H/M ratio on 123I-MIBG scintigraphy. AUC for z-score measurement in the occipital lobe was 0.818 and that for the extent of gray matter (GM) atrophy in the whole brain was 0.788. AUC for the combination of 3D-SSP and VBM analysis was 0.836. The respective sensitivities and specificities for distinguishing DLB from AD were 97.1 and 100 % for the delayed H/M ratio using 123I-MIBG scintigraphy; 88.6 and 73.5 % for the occipital lobe z-score using 3D-SSP analysis; 85.7 and 64.7 % for the extent of whole brain GM atrophy using voxel-based MRI morphometry; and 91.4 and 76.5 % for the combination of 3D-SSP analysis and VBM.
Conclusions
123I-MIBG myocardial scintigraphy was superior to brain perfusion SPECT and brain MRI using an advanced statistical technique to differentiate DLB and AD.







Similar content being viewed by others
References
McKeith I, Mintzer J, Aarsland D, Burn D, Chiu H, Cohen-Mansfield J, et al; International Psychogeriatric Association Expert Meeting on DLB. Dementia with Lewy bodies. Lancet Neurol. 2004;3:19–28.
McKeith IG, Dickson DW, Lowe J, Emre M, O’Brien JT, Feldman H, et al; Consortium on DLB. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65:1–10.
Watanabe H, Ieda T, Katayama T, Takeda A, Aiba I, Doyu M, et al. Cardiac 123I-meta-iodobenzylguanidine (MIBG) uptake in dementia with Lewy bodies: comparison with Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2001;70:781–3.
Yoshita M, Taki J, Yamada M. A clinical role for [123I]MIBG myocardial scintigraphy in the distinction between dementia of the Alzheimer’s-type and dementia with Lewy bodies. J Neurol Neurosurg Psychiatry. 2001;71:583–8.
Oide T, Tokuda T, Momose M, Oguchi K, Nakamura A, Ohara S, et al. Usefulness of [123I] metaiodobenzylguanidine ([123I]MIBG) myocardial scintigraphy in differentiating between Alzheimer’s disease and dementia with Lewy bodies. Intern Med. 2003;42:686–90.
Orimo S, Amino T, Itoh Y, Takahashi A, Kojo T, Uchihara T, et al. Cardiac sympathetic denervation precedes neuronal loss in the sympathetic ganglia in Lewy body disease. Acta Neuropathol. 2005;109:583–8.
Inui Y, Toyama H, Manabe Y, Sato T, Sarai M, Kosaka K, et al. Evaluation of probable or possible dementia with Lewy bodies using 123I-IMP brain perfusion SPECT, 123I-MIBG, and 99mTc-MIBI myocardial SPECT. J Nucl Med. 2007;48:1641–50.
Treglia G, Cason E. Diagnostic performance of myocardial innervation imaging using MIBG scintigraphy in differential diagnosis between dementia with Lewy bodies and other dementias: a systematic review and a meta-analysis. J Neuroimaging. 2012;22:111–7.
McKeith I, O’Brien J, Walker Z, Tatsch K, Booij J, Darcourt J, DLB Study Group, et al. Sensitivity and specificity of dopamine transporter imaging with 123I-FP-CIT SPECT in dementia with Lewy bodies: a phase III, multicentre study. Lancet Neurol. 2007;6:305–13.
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s disease. Neurology. 1984;34:939–44.
Solanski KK, Bomanji J, Moyes J, Mather SJ, Trainer PJ, Britton KE. A pharmacological guide to medicines which interfere with the biodistribution of radiolabelled meta-iodobenzylguanidine (MIBG). Nucl Med Commun. 1992;13:513–21.
Minoshima S, Berger KL, Lee KS, Mintun MA. An automated method for rotational correction and centering of three-dimensional functional brain images. J Nucl Med. 1992;33:1579–85.
Minoshima S, Frey KA, Koeppe RA, Foster NL, Kuhl DE. A diagnostic approach in Alzheimer’s disease using three-dimensional stereotractic surface projections of fluorine-18-FDG PET. J Nucl Med. 1995;36:1238–48.
Germano G, Erel J, Kiat H, Kavanagh PB, Berman DS. Quantitative LVEF and qualitative regional function from gated thallium-201 perfusion SPECT. J Nucl Med. 1997;38:749–54.
Mizumura S, Kumita S, Cho K, Ishihara M, Nakajo H, Toba M, et al. Development of quantitative analysis method for stereotactic brain image: assessment of reduced accumulation in extent and severity using anatomical segmentation. Ann Nucl Med. 2003;17:289–95.
Hirata Y, Matsuda H, Nemoto K, Ohnishi T, Hirao K, Yamashita F, et al. Voxel-based morphometry to discriminate early Alzheimer’s disease from controls. Neurosci Lett. 2005;382:269–74.
Matsuda H, Mizumura S, Nemoto K, Yamashita F, Imabayashi E, Sato N, et al. Automatic voxel-based morphometry of structural MRI by SPM8 plus diffeomorphic anatomic registration through exponentiated lie algebra improves the diagnosis of probable Alzheimer disease. Am J Neuroradiol. 2012;33:1109–14.
Ashburner J. A fast diffeomorphic image registration algorithm. Neuroimage. 2007;38:95–113.
McKeith IG, Del Ser T, Spano P, Emre M, Wesnes K, Anand R, et al. Efficacy of rivastigmine in dementia with Lewy bodies. A randomized double blind, placebo-controlled intermation study. Lancet. 2000;356:2031–6.
Ikeda M, Mori E, Kosaka K, Iseki E, Hashimoto M, Matsukawa N, et al; Donepezil-DLB Study Investigators. Long-term safety and efficacy of donepezil in patients with dementia with Lewy bodies: results from a 52-week, open-label, multicenter extension study. Dement Geriatr Cogn Disord. 2013;36:229–41.
McKeith I, Fairbairn A, Perry R, Thompson P, Perry E. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. Br J Psychiatry. 1992;305:673–8.
Albin RL, Minoshima S, D’Amato CJ, Frey KA, Kuhl DA, Sima AA. Fluoro-deoxyglucose positron emission tomography in diffuse Lewy body disease. Neurology. 1996;47:462–6.
Ishii K, Yamaji S, Kitagaki H, Immamura T, Hirono N, Mori E. Regional cerebral blood flow difference between dementia with Lewy bodies and AD. Neurology. 1999;53:413–6.
Minoshima S, Foster NL, Sima AA, Frey KA, Albin RL, Kuhl DE. Alzheimer’s disease versus dementia with Lewy bodies: cerebral metabolic distinction with autopsy confirmation. Ann Neurol. 2001;50:358–65.
Lobotesis K, Fenwick JD, Phipps A, Ryman A, Swann A, Ballard C, et al. Occipital hypoperfusion on SPECT in dementia with Lewy bodies but not AD. Neurology. 2001;56:643–9.
Colloby SJ, Fenwick JD, Williams ED, Paling SM, Lobotesis K, Ballard C, et al. A comparison of 99mTc-HMPAO SPET changes in dementia with Lewy bodies and Alzheimer’s disease using statistical parametric mapping. Eur J Nucl Med Mol Imaging. 2002;29:615–22.
Pasquier J, Michel BF, Brenot-Rossi I, Hassan-Sebbag N, Sauvan R, Gastaut JL. Value of 99mTc-ECD SPET for the diagnosis of dementia with Lewy bodies. Eur J Nucl Med Mol Imaging. 2002;29:1342–8.
Shimizu S, Hanyu H, Kanetake H, Iwamoto T, Koizumi K, Abe K. Differentiation of dementia with Lewy bodies from Alzheimer’s disease using brain SPECT. Dement Geriatr Cogn Disord. 2005;20:25–30.
Mori T, Ikeda M, Fukuhara R, Nestor PJ, Tanabe H. Correlation of visual hallucinations with occipital rCBF changes by donepezil in DLB. Neurology. 2006;66:935–7.
Hanyu H, Shimizu S, Hirao K, Kanetaka H, Iwamoto T, Chikamori T, et al. Comparative value of brain perfusion SPECT and [123I]MIBG myocardial scintigraphy in distinguishing between dementia with Lewy bodies and Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2006;33:248–53.
Tateno M, Kobayashi S, Shirasaka T, Furukawa Y, Fujii K, Morii H, et al. Comparison of the usefulness of brain perfusion SPECT and MIBG myocardial scintigraphy for the diagnosis of dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2008;26:453–7.
Kantarci K, Ferman TJ, Boeve BF, Weigand SD, Przybelski S, Vemuri P, et al. Focal atrophy on MRI and neuropathologic classification of dementia with Lewy bodies. Neurology. 2012;79:553–60.
Hashimoto M, Kitagaki H, Imamura T, Hirono N, Shimomura T, Kazui H, et al. Medial temporal and whole-brain atrophy in dementia with Lewy bodies. A volumetric MRI study. Neurology. 1998;51:357–62.
Barber R, Ballard C, McKeith IG, Gholkar A, O’Brien JT. MRI volumetric study of dementia with Lewy bodies: a comparison with AD and vascular dementia. Neurology. 2000;54:1304–9.
O’Brien JT, Paling S, Barber R, Williams ED, Ballard C, McKeith IG, et al. Progressive brain atrophy on serial MRI in dementia with Lewy bodies, AD, and vascular dementia. Neurology. 2001;56:1386–8.
Barber R, Gholkar A, Scheltens P, Ballard C, McKeith IG, O’Brien JT. Medial temporal lobe atrophy on MRI in dementia with Lewy bodies. Neurology. 1999;52:1153–8.
Takahashi R, Ishii K, Miyamoto N, Yoshikawa T, Shimada K, Ohkawa S, et al. Measurement of gray and white matter atrophy in dementia with Lewy bodies using diffeomorphic anatomical registration through exponentiated lie algebra: a comparison with conventional voxel-based morphometry. Am J Neuroradiol. 2010;31:1873–8.
Iwanaga K, Wakabayashi K, Yoshimoto M, Tomita I, Satoh H, Takashima H, et al. Lewy body-type degeneration in cardiac plexus in Parkinson’s and incidental Lewy body diseases. Neurology. 1999;52:1269–71.
Miyamoto T, Miyamoto M, Suzuki K, Nishibayashi M, Iwanami M, Hirata K. 123I-MIBG cardiac scintigraphy provides clues to the underlying neurodegenerative disorder in idiopathic REM sleep behavior disorder. Sleep. 2008;31:717–23.
Boeve BF, Silber MH, Fermen TJ, Lin SC, Benarroch EE, Schmeichel AM, et al. Clinicopathologic correlations in 172 cases of rapid eye movement sleep behavior disorder with or without a coexisting neurologic disorder. Sleep Med. 2013;14:754–62.
Colloby SJ, Firbank MJ, Pakrasi S, Lloyd JJ, Driver I, McKeith IG, et al. A comparison of 99mTc-exametazime and 123I-FP-CIT SPECT imaging in the differential diagnosis of Alzheimer’s disease and dementia with Lewy bodies. Int Psychogeriatr. 2008;20:1124–40.
Iranzo A, Valldeoriola F, Lomeña F, Molinuevo JL, Serradell M, Salamero M, et al. Serial dopamine transporter imaging of nigrostriatal function in patients with idiopathic rapid-eye-movement sleep behavior disorder: a prospective study. Lancet Neurol. 2011;10:797–805.
Walker Z, Jaros E, Walker RW, Lee L, Livingston G, Ince PG, et al. Dementia with Lewy bodies: a comparison of clinical diagnosis, FP-CIT single photon emission computed tomography imaging and autopsy. J Neurol Neurosurg Psychiatry. 2007;78:1176–81.
Treglia G, Cason E, Cortelli P, Gabellini A, Liguori R, Bagnato A, et al. Iodine-123 metaiodobenzylguanidine scintigraphy and iodine-123 ioflupane single photon emission computed tomography in Lewy body diseases: complementary or alternative techniques? J Neuroimaging. 2014;24:149–54.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Inui, Y., Toyama, H., Manabe, Y. et al. Comparison of 123I-MIBG myocardial scintigraphy, brain perfusion SPECT, and voxel-based MRI morphometry for distinguishing between dementia with Lewy bodies and Alzheimer’s disease. Ann Nucl Med 28, 796–804 (2014). https://doi.org/10.1007/s12149-014-0873-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-014-0873-2