Abstract
Objective
To know the rotavirus burden associated with acute gastroenteritis along with circulating genotypes among under-five children and to find out possible associations with different demographic and clinical predictors in a tertiary care teaching hospital in Bhubaneswar, Odisha.
Methods
A prospective acute gastroenteritis surveillance conducted from February 2016 to June 2019 at a tertiary care pediatric hospital in Bhubaneswar has enrolled 850 children under five years of age. The stool samples were tested for VP6 antigen of rotavirus by enzyme immunoassay (EIA) and hemi-nested multiplex PCR to find out VP7 (G type) and VP4 (P type) genes. The data was presented using mean ± SD, median (IQR) along with frequencies and percentages.
Results
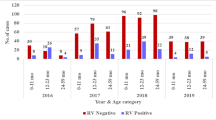
Rotavirus positivity was found in 246 children (28.9%) with male: female ratio of 3:1. An increasing trend of rotaviral diarrheal cases was seen during the winter months. History of vomiting for 2 d, age group of 12–23 mo, and fever were significantly associated with rotavirus diarrhea having odd ratios of 1.80 (95% CI, 1.48, and 1.69, respectively). Among the genotypes, G3 and P8 were found to be most common in the present study.
Conclusion
With the introduction of Rotavac in the state the overall rotaviral distribution has significantly changed. Children of 6–23 mo were the most affected age group in the study indicating the necessity of this vaccine in the early months of life.


Similar content being viewed by others
References
Clark B, Mckendrick M. A review of viral gastroenteritis. Curr Opin Infect Dis. 2004;17:461–9.
Kang G, Desai R, Arora R, et al. Diversity of circulating rotavirus strains in children hospitalized with diarrhea in India, 2005-2009. Vaccine. 2013;31:2879–83.
Anandan S, Peter R, Aramugam R, Ismail N, Veeraraghavan B, Kang G. Group A rotavirus gastroenteritis in older children and adults at a hospital in group A rotavirus gastroenteritis in older children and adults at a hospital in southern India. Vaccine [internet]. 2014;32:A33–5. Available at: https://doi.org/10.1016/j.vaccine.2014.03.008.
Iturriza-gómara M, Kang G, Gray J. Rotavirus genotyping: keeping up with an evolving population of human rotaviruses. J Clin Virol. 2004;31:259–65.
Dey SK, Ushijima H, Phathammavong O, et al. Seasonal trend and serotype distribution of rotavirus infection in Japan, 1981–2008. Pediatr Infect Dis J. 2010;29(2):166–7.
Parashar UD, Bresee JS, Gentsch JR, Glass RI, Control D. Rotavirus. Emerg Infect Dis. 1998;4(4):561–70.
Diarrhoeal Disesase Collaborators G 2016. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of diarrhoea in 195 countries: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis. 2018;18:1211–28.
WHO, UNICEF. Ending Preventable Child Deaths from Pneumonia and Diarrhoea by 2025 - The Integrated Global Action Plan for Pneumonia and Diarrhoea (GAPPD). 2013.
Giri S, Nair NP, Mathew A, et al. Rotavirus gastroenteritis in Indian children < 5 years hospitalized for diarrhoea, 2012 to 2016. BMC Public Health. 2019;19(1):69.
Mohanty E, Dwibedi B. Kar SK, et al. Epidemiological features and genetic characterization of virus strains in rotavirus associated gastroenteritis in children of Odisha in Eastern India. Infect Genet Evol. 2017;53:77-84.
John J, Sarkar R, Muliyil J, Bhandari N, Bhan MK, Kang G. Rotavirus gastroenteritis in India , 2011–2013: revised estimates of disease burden and potential impact of vaccines. Vaccine [Internet]. 2014;32:A5–9. Available at: https://doi.org/10.1016/j.vaccine.2014.03.004.
Iturriza-gomara M, Green J, Brown DWG, Desselberger U, Gray JJ. Comparison of specific and random priming in the reverse transcriptase polymerase chain reaction for genotyping group A rotaviruses. J Virol Methods. 1999;78:93–103.
Banerjee I, Ramani S, Primrose B, et al. Modification of rotavirus multiplex RT-PCR for the detection of G12 strains based on characterization of emerging G12 rotavirus strains from south India. J Med Virol [Internet]. 2007;79:1413–21. Available at: http://www.interscience.wiley.com.
Levy K, Hubbard AE, Eisenberg JNS. Seasonality of rotavirus disease in the tropics: a systematic review and meta-analysis. Int J Epidemiol. 2009;38:1487–96.
Kawai K, O’Brien MA, Goveia MG, Mast TC, El Khoury AC. Burden of rotavirus gastroenteritis and distribution of rotavirus strains in Asia: a systematic review. Vaccine [Internet]. 2012;30:1244–54. Available at: http://www.elsevier.com/locate/vaccine.
Cook SM, Glass RI, Lebaron CW, Ho MS. Global seasonality of rotavirus infections. Bull World Health Organ. 1990;68(5059):171–7.
Paniker CKJ, Mathew S, Mathan M. Rotavirus and acute diarrhoeal disease in children in a southern Indian coastal town *. Bull World Health Organ. 1982;60(1):123–7.
Konno T, Suzuki H, Katsushima N, et al. Influence of temperature and relative humidity on human rotavirus infection in Japan. J Infect Dis [Internet]. 1983;147(1):125–8. Available at: http://www.jstor.org/stable/30115036.
Bhandari N, Rongsen-Chandola T, Bavdekar A, et al. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian infants: a randomised double-blind placebo controlled trial. Lancet. 2015;383(9935):2136–43.
Ansari SA, Springthorpe VS, Sattar SA. Survival and vehicular spread of human rotaviruses: possible relation to seasonality of outbreaks. Rev Infect Dis [Internet]. 1991;13:448–61. Available at: http://cid.oxfordjournals.org/.
Kang G, Arora R, Chitambar SD, et al. Multicenter, hospital-based surveillance of rotavirus disease and strains among Indian children aged ! 5 years. J Infect Dis. 2009;200:147–53.
Acknowledgements
The authors are grateful for the co-operation by all participants, parents/guardians and surveillance staff of the KIMS hospital. Their special thanks to Dr. RishabhPugalia and Dr. Mirabai Das for their help in data collection and counselling the parents. They thank the Indian Council of Medical Research, New Delhi; Ministry of Health and Family Welfare, Govt. of India and the State government of Odisha for their support. They also thank the team at Christian Medical College, Vellore for the support.
Funding
This work was supported by grants from the Bill and Melinda Gates Foundation to the Centers for Disease Control and Prevention, Atlanta, GA, USA (subcontract to Christian Medical College, Vellore grant no MOA#871-15SC) and the Translational Health Science and Technology Institute (grant no OPP1165083).
Author information
Authors and Affiliations
Contributions
VG, NKM, SRN: Study design, manuscript development; MKN, NM: Data acquisition, management and manuscript development; VG, NKM, NM, RK, SRN: Data analysis, literature review and manuscript writing; All authors approved the final manuscript. NKM will act as guarantor of the study.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghoshal, V., Nayak, M.K., Misra, N. et al. Surveillance and Molecular Characterization of Rotavirus Strains Circulating in Odisha, India after Introduction of Rotavac. Indian J Pediatr 88 (Suppl 1), 41–46 (2021). https://doi.org/10.1007/s12098-020-03622-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-020-03622-3