Abstract
Background
Although Ras-association domain family of gene 2 (RASSF2) has been shown to undergo promoter methylation at high frequency in some cancer types and in brain metastases, its clinical utility as a useful prognostic molecular marker remains unclear in gastric cancer.
Methods
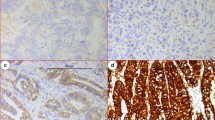
Prognostic significance of RASSF2 expression was retrospectively analysed by immunohistochemically in 105 patients with gastric cancer who underwent curative gastrectomy.
Results
Low RASSF2 expression was detected in 58 (55 %) patients, whereas 47 patients (45 %) had high RASSF2 expression. Lymph node involvement, pT stage, TNM stage, vascular invasion, perineural invasion and the presence of recurrence were found to be significantly related to RASSF2 expression levels. Low PRL-3 expression was closely correlated with lymph node metastasis (p = 0.001), advanced pT stage (p = 0.021), advanced TNM stage (p < 0.001), the presence of vascular invasion (p < 0.001), perineural invasion (p = 0.018) and high prevalence of recurrence (p = 0.003) compared with high RASSF2 expression. The median disease-free survival (DFS) time for patients with low RASSF2 expression was significantly worse than that of patients with high RASSF2 expression (10.2 vs. 50.6 months, p < 0.001). In addition, patients with high RASSF2 expression had the higher overall survival (OS) interval compared to patients with low RASSF2 expression (NR vs. 14.9 months, p < 0.001). In the multivariate analysis, the rate of RASSF2 expression levels was an independent prognostic factor, for DFS [p < 0.001, HR 0.12 (0.10–0.88)] and OS [p < 0.001, HR 0.10 (0.04–0.46)], as were pT stage and TNM stage, respectively.
Conclusions
RASSF2 may be an important molecular marker for carcinogenesis, prognosis and progression in gastric cancer, but the potential value of RASSF2 expression as a useful molecular marker in gastric cancer progression should be evaluated, comprehensively. It would be possible to develop treatments targeting RASSF2 and advance new treatment strategies for gastric cancer.



Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30.
Hartgrink HH, Jansen EP, van Grieken NC, van de Velde CJ. Gastric cancer. Lancet. 2009;374:477.
Kataoka Y, Okabe H, Yoshizawa A, Minamiguchi S, Yoshimura K, Haga H, et al. HER2 expression and its clinicopathological features in resectable gastric cancer. Gastric Cancer. 2013;16:84–93.
Sheng WQ, Huang D, Ying JM, Lu N, Wu HM, Liu YH, et al. HER2 status in gastric cancers: a retrospective analysis from four Chinese representative clinical centers and assessment of its prognostic significance. Ann Oncol. 2013;24:2360–4.
Malumbres M, Barbacid M. RAS oncogenes: the first 30 years. Nat Rev Cancer. 2003;3:459–65.
Liu C, Pan Y, Wang X, Lu J, Huang B, Li X. Activation of RASSF2A by p300 induces late apoptosis through histone hyperacetylation. Cell Biol Int. 2010;34:1133–9.
Luo D, Ye T, Li TQ, Tang P, Min SD, Zhao GF, et al. Ectopic expression of RASSF2 and its prognostic role for gastric adenocarcinoma patients. Exp Ther Med. 2012;3:391–6.
Cooper WN, Hesson LB, Matallanas D, Dallol A, von Kriegsheim A, Ward R, et al. RASSF2 associates with and stabilizes the proapoptotic kinase MST2. Oncogene. 2009;28:2988–98.
Richter AM, Pfeifer GP, Dammann RH. The RASSF proteins in cancer; from epigenetic silencing to functional characterization. Biochim Biophys Acta. 2009;1796:114–28.
Vos MD, Ellis CA, Elam C, Ulku AS, Taylor BS, Clark GJ. RASSF2 is a novel K-Ras-specific effector and potential tumor suppressor. J Biol Chem. 2003;2(78):28045–51.
Cooper WN, Dickinson RE, Dallol A, Grigorieva EV, Pavlova TV, Hesson LB, et al. Epigenetic regulation of the ras effector/tumour suppressor RASSF2 in breast and lung cancer. Oncogene. 2008;27:1805–11.
Hesson LB, Wilson R, Morton D, Adams C, Walker M, Maher ER, et al. CpG island promoter hypermethylation of a novel Ras-effector gene RASSF2A is an early event in colon carcinogenesis and correlates inversely with K-ras mutations. Oncogene. 2005;24:3987–94.
Liao X, Siu MK, Chan KY, Wong ES, Ngan HY, Chan QK, et al. Hypermethylation of RAS effector related genes and DNA methyltransferase 1 expression in endometrial carcinogenesis. Int J Cancer. 2008;123:296–302.
Maruyama R, Akino K, Toyota M, Suzuki H, Imai T, Ohe-Toyota M, et al. Cytoplasmic RASSF2A is a proapoptotic mediator whose expression is epigenetically silenced in gastric cancer. Carcinogenesis. 2008;29:1312–8.
Schagdarsurengin U, Richter AM, Hornung J, Lange C, Steinmann K, Dammann RH. Frequent epigenetic inactivation of RASSF2 in thyroid cancer and functional consequences. Mol Cancer. 2010;9:264.
Zhang X, Ma Y, Wu Y, Lin L, Ma X, Zhang Y. Aberrant promoter methylation and silencing of RASSF2A gene in cervical cancer. J Obstet Gynaecol Res. 2014;40:1375–81.
Edge SB, Byrd DR, Compton CC. American Joint Committee on Cancer Staging Manual. 7th ed. New York: Springer; 2010. p. 117.
Association Japanese Gastric Cancer. Japanese classification of gastric carcinoma. 2nd English ed. Gastric Cancer. 1998;1:10–24.
Qu Y, Dang S, Hou P. Gene methylation in gastric cancer. Clin Chim Acta. 2013;424:53–65.
Calcagno DQ, de Arrodo Cardoso Smith M, Burbano RR. Cancer type-specific epigenetic changes: gastric cancer. Methods Mol Biol. 2015;1238:79–101.
Agathanggelou A, Cooper WN, Latif F. Role of the ras-association domain family 1 tumor suppressor gene in human cancers. Cancer Res. 2005;65:3497–508.
Schubbert S, Bollag G, Shannon K. Deregulated Ras signaling in developmental disorders: new tricks for an old dog. Curr Opin Genet Dev. 2007;17:15–22.
Cooper WN, Dickinson RE, Dallol A, Grigorieva EV, Pavlova TV, Hesson LB, et al. Nuclear localisation and epigenetic inactivation of the ras effector/tumour suppressor RASSF2A in multiple human cancers. Oncogene. 2008;27:1805–11.
Cox AD, Der CJ. The dark side of Ras: regulation of apoptosis. Oncogene. 2003;22:8999–9006.
Park HW, Kang HC, Kim IJ, Jang SG, Kim K, Yoon HJ, et al. Correlation between hypermethylation of the RASSF2A promoter and K-ras/BRAF mutations in microsatellite-stable colorectal cancers. Int J Cancer. 2007;120:7–12.
Zhang Z, Sun D, Van do N, Tang A, Hu L, Huang G. Inactivation of RASSF2A by promoter methylation correlates with lymph node metastasis in nasopharyngeal carcinoma. Int J Cancer. 2007;120:32–8.
Endoh M, Tamura G, Honda T, Homma N, Terashima M, Nishizuka S, et al. RASSF2, a potential tumour suppressor, is silenced by CpG island hypermethylation in gastric cancer. Br J Cancer. 2005;93:1395–9.
Imai T, Toyota M, Suzuki H, Akino K, Ogi K, Sogabe Y, et al. Epigenetic inactivation of RASSF2 in oral squamous cell carcinoma. Cancer Sci. 2008;99:958–66.
Steinmann K, Sandner A, Schagdarsurengin U, Dammann RH. Frequent promoter hypermethylation of tumor-related genes in head and neck squamous cell carcinoma. Oncol Rep. 2009;22:1519–26.
Ren J, He W, Zhang R, Li Z, Cao W, Yao J, et al. RASSF2A promoter methylation in hepatitis B virus-related hepatocellular carcinogenesis and its correlation with elevated serum alpha-fetoprotein level. J Huazhong Univ Sci Technol Med Sci. 2009;29:309–12.
Gharanei S, Brini AT, Vaiyapuri S, Alholle A, Dallol A, Arrigoni E, et al. RASSF2 methylation is a strong prognostic marker in younger age patients with Ewing sarcoma. Epigenetics. 2013;8:893–8.
Mezzanotte JJ, Hill V, Schmidt ML, Shinawi T, Tommasi S, Krex D, et al. RASSF6 exhibits promoter hypermethylation in metastatic melanoma and inhibits invasion in melanoma cells. Epigenetics. 2014;9:1496–503.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Aydin, D., Bilici, A., Kayahan, S. et al. Prognostic importance of RASSF2 expression in patients with gastric cancer who had undergone radical gastrectomy. Clin Transl Oncol 18, 608–616 (2016). https://doi.org/10.1007/s12094-015-1405-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1405-9