Abstract
Purpose
Familial aggregation among patients with several hematological malignancies has been revealed. This emphasizes the importance of genetic factors. Only few genes predisposing to familial hematological malignancies have been reported until now due to the low occurrence. We have described in previous study PRF1 and CEBPA variants that might contribute to the background of genetic factors, which encourage us to extend our investigations to other cooperating genes. The aim of this study is to determine whether germline additional sex combs-like 1 (ASXL1) gene mutations may be involved?
Methods/patients
In this study, we investigated the candidate gene ASXL1 by direct sequencing in 88 unrelated Tunisian and French families with aggregated hematological malignancies.
Results
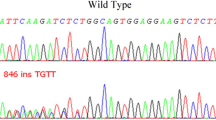
We report a new p.Arg402Gln germline missense substitution in two related Tunisian patients which has not been previously described. We identified here this variant for the first time in non-Hodgkin lymphoma. The p.Arg402Gln variant was not found in 200 control chromosomes. In silico analysis has predicted potential deleterious effect on ASXL1 protein.
Conclusions
From an extended candidate genes analyzed in the field of familial hematological malignancies, ASXL1 might be involved. This variant should be considered since a potential damaging effect was predicted by in silico analysis, with a view to develop functional assay in order to investigate the biological assessment.


Similar content being viewed by others
References
Cho YS, Kim EJ, Park UH, Sin HS, Um SJ. Additional sex comb-like 1 (ASXL1), in cooperation with SRC-1, acts as a ligand-dependent coactivator for retinoic acid receptor. JBC. 2006;281:17588–98.
Aravind L, Iyer LM. The HARE-HTH and associated domains: novel modules in the coordination of epigenetic DNA and protein modifications. Cell Cycle. 2012;11:119–31.
Lee SW, Cho YS, Na JM, Park UH, Kang M, Kim EJ, et al. ASXL1 represses retinoic acid receptor mediated transcription through associating with HP1 and LSD1. JBC. 2010;285:18–29.
Huang HS, Matevossian A, Whittle C, Kim SY, Schumacher A, Baker SP, et al. Prefrontal dysfunction in schizophrenia involves mixed-lineage leukemia 1-regulated histone methylation at GABAergic gene promoters. J Neurosci. 2007;27:11254–62.
Ringrose L, Paro R. Polycomb/Trithorax response elements and epigenetic memory of cell identity. Development. 2007;134:223–32.
Schwartz YB, Pirrotta V. Polycomb silencing mechanisms and the management of genomic programmes. Nature Rev Genet. 2007;8:9–22.
Carbuccia N, Murati A, Trouplin V, Brecqueville M, Adélaïde J, Rey J, et al. Mutations of ASXL1 gene in myeloproliferative neoplasms. Leukemia. 2009;23:2183–6.
Gelsi-Boyer V, Trouplin V, Adelaide J, Bonansea J, Cervera N, Carbuccia N, et al. Mutations of polycomb-associated gene ASXL1 in myelodysplastic syndromes and chronic myelomonocytic leukaemia. Br J Haematol. 2009;145:788–800.
Chen TC, Hou HA, Chou WC, Tang JL, Kuo YY, Chen CY, et al. Dynamics of ASXL1 mutation and other associated genetic alterations during disease progression in patients with primary myelodysplastic syndrome. Blood Cancer J. 2014;4:177–85.
Hoischen A, van Bon BW, Rodriguez-Santiago B, Gilissen C, Vissers LE, de Vries P, et al. De novo nonsense mutations in ASXL1 cause Bohring-Opitz syndrome. Nature Genet. 2011;43:729–31.
Magini P, Della Monica M, Uzielli ML, Mongelli P, Scarselli G, Gambineri E, et al. Two novel patients with Bohring-Opitz syndrome caused by de novo ASXL1 mutations. Am J Med Genet. 2012;158:917–21.
Thol F, Friesen I, Damm F, Yun H, Weissinger EM, Krauter J, et al. Prognostic significance of ASXL1 mutations in patients with myelodysplastic syndromes. J Clin Oncol. 2011;29:2499–506.
Pratcorona M, Abbas S, Sanders M, Koenders JE, Kavelaars FG, Erpelinck-Verschueren CA, et al. Acquired mutations in ASXL1 in acute myeloid leukemia: prevalence and prognostic value. Hematologica. 2012;97:388–92.
Gelsi-Boyer V, Brecqueville M, Devillier R, Murati A, Mozziconacci MJ, Birnbaum D. Mutations in ASXL1 are associated with poor prognosis across the spectrum of malignant myeloid diseases. J Hematol Oncol. 2012;5:12–7.
Patnaik MM, Itzykson R, Lasho TL, Kosmider O, Finke CM, Hanson CA, et al. ASXL1 and SETBP1 mutations and their prognostic contribution in chronic myelomonocytic leukemia: a two-center study of 466 patients. Leukemia. 2014;28:2206–12.
Abdel-Wahab O, Pardanani A, Patel J, Wadleigh M, Lasho T, Heguy A, et al. Concomitant analysis of EZH2 and ASXL1 mutations in myelofibrosis, chronic myelomonocytic leukemia and blast-phase myeloproliferative neoplasms. Leukemia. 2011;25:1200–2.
Quesada V, Conde L, Villamor N, Ordóñez GR, Jares P, Bassaganyas L, et al. Exome sequencing identifies recurrent mutations of the splicing factor SF3B1 gene in chronic lymphocytic leukemia. Nature Genet. 2011;44:47–52.
Tavtigian SV, Deffenbaugh AM, Yin L, Judkins T, Scholl T, Samollow PB, de Silva D, Thomas A, et al. Comprehensive statistical study of 452 BRCA1 missense substitutions with classification of eight recurrent substitutions as neutral. J Med Genet. 2006;43:295–305.
Tong W, Li L, Weng Z. Computational prediction of binding hotspots Conf Proc IEEE. Eng Med Biol Soc. 2004;4:2980–3.
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, et al. A method and server for predicting damaging missense mutations. Nat Methods. 2010;7:248–9.
Stecher G, Liu L, Sanderford M, Peterson D, Tamura K, Kumar S. MEGA-MD: Molecular Evolutionary Genetics Analysis software with mutational diagnosis of amino acid variation. Bioinformatics. 2014;30:1305–7.
Houlston RS, Catovsky D, Yuille MR. Genetic susceptibility to chronic lymphocytic leukemia. Leukemia. 2002;16:1008–14.
El Abed R, Bourdon V, Voskoboinik I, Omri H, Youssef YB, Laatiri MA, et al. Molecular study of the perforin gene in familial hematological malignancies. HCCP J. 2011;9:9–16.
El Abed R, Bourdon V, Huiart L, Eisinger F, Khelif A, Frenay M, et al. Molecular study of CEPBA in familial hematological malignancies. Fam Cancer. 2009;8:581–4.
Su L, Li X, Gao SJ, Yu P, Liu XL, Tan YH, et al. Cytogenetic and genetic mutation features of de novo acute myeloid leukemia in elderly chinese patients. Asian Pac J Cancer Prev. 2014;15:895–8.
Katoh M. Functional and cancer genomics of ASXL family members. Br J Cancer. 2013;109:299–306.
An Q, Wright SL, Moorman AV, Parker H, Griffiths M, Ross FM, et al. Heterogeneous breakpoints in patients with acute lymphoblastic leukemia and the dic(9;20)(p11-13;q11) show recurrent involvement of genes at 20q11.21. Hematologica. 2009;94:1164–9.
Ramanathan A, Mahmoud HAR, Hui LP, Mei NY, Valliappan V, Zain RB. Oral extranodal non Hodgkin’s lymphoma: series of forty two cases in Malaysia. Asian Pac J Cancer Prev. 2014;15:1633–7.
Cho YS, Kim EJ, Park UH, Sin HS, Um SJ. Additional sex comb-like1(ASXL1) in cooperation with SRC-1, acts as a ligand-dependent coactivator for retinoic acid receptor. J Biol Chem. 2006;281:17588–98.
Tang M, Hou YL, Kang QQ, Chen XY, Duan LQ, Shu J, et al. All-trans-retinoic acid promotes iodine uptake via upregulating the sodium iodide symporter in medullary thyroid cancer stem cells. Asian Pac J Cancer Prev. 2014;15:1859–62.
Acknowledgments
This work was supported by la Société Française d’Hématologie, le groupe Génétique et Cancer and Institut National du Cancer (INCa) and the Ministère de l’Enseignement Supérieur et de la Recherche Scientifique en Tunisie. It is a part of the GenHem INSERM/DGRS project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
This study was approved by the ethics committee of the hospital and all subjects gave their informed consent according to Helsinki Declaration.
Conflict of interest
We declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hamadou, W.S., Abed, R.E., Besbes, S. et al. Familial hematological malignancies: ASXL1 gene investigation. Clin Transl Oncol 18, 385–390 (2016). https://doi.org/10.1007/s12094-015-1379-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1379-7