Abstract
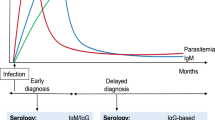
Tuberculosis is a significant problem globally for domestic animals as well as captive and free ranging wild life. Rapid point of care (POC) serology kits are well suited for the diagnosis of TB in wild animals. However, wild animals are invariably exposed to environmental non-pathogenic mycobacterium species with the development of cross reacting antibodies. In the present study, POC TB diagnosis kit was developed using a combination of pathogenic Mycobacteria specific recombinant antigens and purified protein derivatives of pathogenic and non-pathogenic Mycobacteria. To benchmark the TB antibody detection kit, particularly in respect to specificity which could not be determined in wildlife due to the lack of samples from confirmed uninfected animals, we first tested well-characterized sera from 100 M. bovis infected and 100 uninfected cattle. Then we investigated the kit’s performance using sera samples from wildlife, namely Sloth Bears (n = 74), Elephants (n = 9), Cervidae (n = 14), Felidae (n = 21), Cape buffalo (n = 2), Wild bear (n = 1) and Wild dog (n = 1).In cattle, a sensitivity of 81% and a specificity of 90% were obtained. The diagnostic sensitivity of the kit was 94% when the kit was tested using known TB positive sloth bear sera samples. 47.4% of the in-contact sloth bears turned seropositive using the rapid POC TB diagnostic kit. Seropositivity in other wild animals was 25% when the sera samples were tested using the kit. A point of care TB sero-diagnostic kit with the combination of proteins was developed and the kit was validated using the sera samples of wild animals.





Similar content being viewed by others
References
Larsen RS, Salman MD, Mikota SK, Isaza R, Montali RJ, Triantis J (2000) Evaluation of a multiple-antigen enzyme-linked immunosorbent assay for detection of Mycobacterium tuberculosis infection in captive elephants. J Zoo Wildl Med 31:291–302. https://doi.org/10.1638/10427260(2000)031[0291:EOAMAE]2.0.CO;2
Lyashchenko KP, Greenwald R, Esfandiari J, Olsen JH, Ball R, Dumonceaux G, Dunker F, Buckley C, Richard M, Murray S, Payeur JB, Andersen P, Pollock JM, Mikota S, Miller M, Sofranko D, Waters WR (2006) Tuberculosis in Elephants: antibody responses to defined antigens of Mycobacterium tuberculosis, potential for early diagnosis, and monitoring of treatment. Clin Vaccine Immunol 13:722–732. https://doi.org/10.1128/CVI.00133-06
Greenwald R, Lyashchenko O, Esfandiari J, Miller M, Mikota S, Olsen JH, Ball R, Dumonceaux G, Schmitt D, Moller T, Payeur JB, Harris B, Sofranko D, Waters WR, Lyashchenko KP (2009) Highly accurate antibody assays for early and rapid detection of tuberculosis in African and Asian elephants. Clin Vaccine Immunol 16:605–612. https://doi.org/10.1128/CVI.00038-09
Mehrotra J, Mittal A, Rastogi AK, Jaiswal AK, Bhandari NK, Sinha S (1990) Antigenic definition of plasma membrane proteins of Bacillus Calmette-Guerin, predominant activation of human T cells by low-molecular-mass integral proteins. Scand J Immunol 50:411–419. PMID:10520182
Rishikesavan R, Sha AA, Chandranaik BM, Basavarajappa K, Giridhar P, Renukaprasad C (2006) Study on prevalence of Tuberculosis in rescued captive Sloth Bears (Melursus ursinus). J Vet Pub Health 6:53–54
Rietkerk FE, Griffin FT, Wood B, Mubarak SM, Delima EC, Badri OM, Lindsay NB, Williamson D (1993) Treatment of Bovine Tuberculosis in an Arabian Oryx (Oryx leucoryx). J Zoo Wildl Med 24:523–527
Lyashchenko KP, Greenwald R, Esfandiari J, Chambers MA, Vicente J, Gortazar C, Santos N, Correia-Neves M, Buddle BM, Jackson R, O’Brien DJ, Schmitt S, Palmer MV, Delahay RJ, Waters WR (2008) Animal-side serologic assay for rapid detection of Mycobacterium bovis infection in multiple species of free-ranging. Wildl Vet Microbiol 132:283–292. https://doi.org/10.1016/j.vetmic.2008.05.029
Waters WR, Palmer MV, Bannantine JP, Greenwald R, Esfandiari J, Andersen P, McNair J, Pollock JM, Lyashchenko KP (2005) Antibody responses in reindeer (Rangifer tarandus) infected with Mycobacterium bovis. Clin Diagn Lab Immunol 12:727–735. https://doi.org/10.1128/CDLI.12.6.727-735.2005
Clifton-Hadley RS, Wilesmith JW, Stuart FA (1993) Mycobacterium bovis in the European badger (Meles meles) Epidemiological findings in tuberculosis badgers from a naturally infected population. Epidemiol Infect 111:9–19. https://doi.org/10.1017/S0950268800056624
Chambers MA, Pressling WA, Cheeseman CL, Clifton-Hadley RS, Hewinson RG (2002) Value of existing serological tests for identifying badgers that shed Mycobacterium bovis. Vet Microbiol 86:183–189. https://doi.org/10.1016/S0378-1135(02)00012-3
Mann PC, Bush M, Janssen DL, Frank ES, Montali RJ (1981) Clinicopathologic correlations of tuberculosis in large zoo mammals. J Am Vet Med Assoc 179:1123–1129. PMID:7035420
Cleaveland S, Mlengeya T, Kazwala RR, Michel A, Kaare MT, Jones SL, Eblate E, Shirima GM, Packer C (2005) Tuberculosis in Tanzanian wildlife. J Wildl Dis 41:446–453. https://doi.org/10.7589/0090-3558-41.2.446
Donnelly CA, Woodroffe R, Cox DR, Bourne FJ, Cheeseman CL, Clifton-Hadley RS, Wei G, Gettinby G, Gilks P, Jenkins H, Johnston WT, Le Fevre AM, McInerney JP, Morrison WI (2006) Positive and negative effects of widespread badger culling on tuberculosis in cattle. Nature 439:843–846. https://doi.org/10.1038/nature04454
Frost PA (2006) Tuberculosis in nonhuman primates with an emphasis on Mycobacterium bovis. In: Thoen CO, Steele JH, Gilsdorf MJ (eds) Mycobacterium bovis infection in Animals and Humans, 2nd edn. Blackwell Publishing Ltd, Oxford
De Lisle GW, Mackintosh CG, Bengis RG (2001) Mycobacterium bovis in free-living and captive wildlife, including farmed deer. Revue Scientifique et Technique de 1’ Office International des Epizooties 20:86–111. PMID:11288522
Thoen CO, Lobue PA, Enarson DA, Kaneene JB, De kantor IN (2009) Tuberculosis a re-emerging disease in animals and humans. Vet Ital 45:135-181. PMID: 20391396
Alvarez J, Perez AM, Bezos J, Casal C, Romero B, Rodriguez-Campos S, Saez-Llorente JL, Diaz R, Carpintero J, de Juan L, Domínguez L (2012) Eradication of bovine tuberculosis at a herd-level in Madrid, Spain study of within-herd transmission dynamics over a 12 year period. BMC Vet 8:100. https://doi.org/10.1186/1746-6148-8-100
De Garine WM, Caron A, Kock R, Tschopp R, Munyeme M, Hofmeyr M, Michel A (2013) A review of bovine tuberculosis at the wildlife-livestock human interface in sub-saharan Africa. Epidemiol Infect 141:1342–1356. https://doi.org/10.1017/S0950268813000708
Mancuso JD, Tribble D, Mazurek GH, Li Y, Olsen C, Aronson NE, Geiter L, Goodwin D, Keep LW (2011) Impact of targeted testing for latent tuberculosis infection using commercially available diagnostics. Clin Infect Dis 53:234–244. https://doi.org/10.1093/cid/cir321
Stevens JB, Thoen CO, Rohonczy EB, Tessaro S, Kelly HA, Duncan JR (1998) The immunological response of llamas (Lama glama) following experimental infection with Mycobacterium bovis. Can J Vet Res 62:102–109.PMCID:PMC1189455
Parsons LM, Somoskövi A, Gutierrez C, Lee E, Paramasivan CN, Abimiku A, Spector S, Roscigno G, Nkengasong J (2011) Laboratory diagnosis of tuberculosis in resource-poor countries challenges and opportunities. Clin Microbiol Rev 24:314–350. https://doi.org/10.1128/CMR.00059-10
Veerasami M, Reddy DS, Sugumar P, Naidu SS, Bahekar V, Mahesh kumar EK, Mukherjee F, Rana SK, Chandran D, Das D, Srinivasan VA (2012) Multi-antigen print immunoassay for sero-epidemiological surveillance of bovine tuberculosis on Indian cattle farms. Vet Ital 48:253–267
Waters WR, Palmer MV, Thacker TC, Bannantine JP, Vordermeier HM, Hewinson RG, Greenwald R, Esfandiari J, McNair J, Pollock JM, Andersen P, Lyashchenko KP (2006) Early antibody responses to experimental Mycobacterium bovis infection of cattle. Clin Vaccine Immunol 13:648–654. https://doi.org/10.1128/CVI.00061-06
Lesellier S, Corner L, Costello E, Sleeman P, Lyashchenko K, Greenwald R, Esfandiari J, Singh M, Hewinson RG, Chambers M, Gormley E (2008) Antigen specific immunological responses of badgers (Meles meles) experimentally infected with Mycobacterium bovis. Vet Immunol Immunopathol 122:35–45. https://doi.org/10.1016/j.vetimm.2007.11.005
Duncan AE, Lyashchenko K, Greenwald R, Miller M, Ball R (2009) Application of Elephant TB STAT-PAK assay and MAPIA (multi-antigen print immunoassay) for detection of tuberculosis and monitoring of treatment in black rhinoceros (Diceros bicornis). J Zoo Wildl Med. 40:781–785. https://doi.org/10.1638/2009-0044.1
Vosloo W, Tippoo P, Hughes JE, Harriman N, Emms M, Beatty DW, Zappe H, Steyn LM (1997) Characterization of a lipoprotein in Mycobacterium bovis (BCG) with sequence similarity to the secreted protein MPB70. Gene 188:123–128. https://doi.org/10.1016/S0378-1119(96)00806-2
Corner LA, Barrett RH, Lepper AWD, Lewis V, Pearson CW (1981) A survey of mycobacteriosis of feral pigs in the Northern Territory. Aust Vet J 57:537–542. https://doi.org/10.1111/j.1751-0813.1981.tb00428.x
Beesley JE (2015) Immuno labelling and electron microscopy in cyto chemistry. Curr Opin Immunol 2:927–931
Rhodes SG, Gunn-Mooore D, Boschiroli ML, Schiller I, Esfandiari JR, Greenwald R, Lyaschenko KP (2011) Comparative study of IFN-gamma and antibody tests for feline tuberculosis. Vet Immunol Immunopathol 144:129–134. https://doi.org/10.1016/j.vetimm.2011.07.020
Ameni G, Tadesse K, Hailu E, Deresse Y, Medhin G, Aseffa A, Hewinson G, Vordermeier M, Berg S (2013) Transmission of M. tuberculosis between Farmers and Cattle in Central Ethiopia. PLoS One 8:e76891. https://doi.org/10.1371/journal.pone.0076891
Chen Y, Chao Y, Deng Q, Liu T, Xiang J, Chen J, Zhou J, Zhan Z, Kuang Y, Cai H, Chen H, Guo A (2009) Potential challenges to the stop plan for humans in china; cattle maintain M. bovis and M. tuberculosis. Tuberculosis 89:95–100. https://doi.org/10.1016/j.tube.2008.07.003
Bhanu Rekha V, Gunaseelan L, Ganesh P, Reza N, Sukumar B (2015) Molecular detection of Mycobacterium tuberculosis from bovine milk samples. J Adv Vet Anim Res 2:80–83. https://doi.org/10.5455/javar.2015.b44
Waters WR, Palmer MV, Bannantine JP, Whipple DL, Greenwald R, Esfandiari J, Andersen P, McNair J, Pollock JM, Lyashchenko KP (2004) Antigen recognition by serum antibodies in white-tailed deer (Odocoileus virginianus) experimentally infected with Mycobacterium bovis. Clin Diagn Lab Immunol 11:849–855. https://doi.org/10.1128/CDLI.11.5.849-855.2004
Renshaw PS, Lightbody KL, Veverka V, Muskett FW, Kelly G, Frenkiel TA, Gordon SV, Hewinson RG, Burke B, Norman J, Williamson RA, Carr MD (2005) Structure and function of the complex formed by the tuberculosis virulence factors CFP-10 and ESAT-6. EMBO 24:2491–2498. https://doi.org/10.1038/sj.emboj.7600732
Guinn KM, Hickey MJ, Mathur SK, Zakel KL, Grotzke JE, Lewinsohn DM, Smith S, Sherman DR (2004) Individual RD1-region genes are required for export of ESAT-6/CFP-10 and for virulence of Mycobacterium tuberculosis. Mol Microbiol 51:359–370. https://doi.org/10.1046/j.1365-2958.2003.03844.x
Harboe M, Oettinger T, Wiker HG, Rosenkrands I, Andersen P (1996) Evidence for occurrence of the ESAT‐6 protein in Mycobacterium tuberculosis and virulent Mycobacterium bovis and for its absence in Mycobacterium bovis BCG. Infect Immunol 64:16–22. PMCID:PMC173721
Vordermeier HM, Gareth J, Jones Bryce M, Buddle R, Hewinson G, Villarreal-Ramos Bernardo (2016) Bovine tuberculosis in cattle: vaccines, DIVA tests, and host biomarker discovery. Annu Rev Anim Biosci 4:87–109. https://doi.org/10.1146/annurev-animal-021815-11131
Arend SM, de Haas P, Leyten E, Rosenkrands I, Rigouts L, Andersen P, Mijs W, van Dissel JT, van Soolingen D (2005) ESAT-6 and CFP-10 in clinical versus environmental isolates of Mycobacterium kansasii. J infect Dis 191:1301–1310. https://doi.org/10.1086/428950
Arend SM, Van Meijgaarden KE, De Boer K, De Palou EC, Van Soolingen D, Ottenhoff TH, Van Dissel JT (2012) Tuberculin skin testing and in vitro T cell responses to ESAT-6 and culture filtrate protein 10 after infection with Mycobacterium marinum or M. Kansasii. J Infect Dis 186:1797–1807. https://doi.org/10.1086/345760
Lyashchenko KP, Singh M, Colangeli R, Gennaro ML (2000) A multi-antigen print immunoassay for the development of serological diagnosis of infectious diseases. J Immunol Methods. 242:91–100. https://doi.org/10.1016/S0022-1759(00)00241-6
Sharma AK, Dhasmana N, Dubey N, Kumar N, Gangwal A, Gupta M, Singh Y (2017) Bacterial virulence factors: secreted for survival. Indian J Microbiol. 57:1–10. https://doi.org/10.1007/s12088-016-0625-1
Aurtenetxe O, Barral M, Vicente J, de la Fuente J, Gortazar C, Juste RA (2008) Development and validation of an enzyme-linked immunosorbent assay for antibodies against Mycobacterium bovis in European wild boar. BMC Vet Res 4:43. https://doi.org/10.1186/1746-6148-4-43
Da Silva EB, Silva BD, Leon JR, Kipnis A, Santos IK, Junqueira-Kipnis AP (2011) Using BCG and Ag85 as antigens in an indirect ELISA for the diagnosis of bovine tuberculosis. Vet J 187:276–278. https://doi.org/10.1016/j.tvjl.2009.11.017
Lyashchenko KP, Greenwald R, Esfandiari J, O’Brien DJ, Schmitt SM, Palmer MV, Waters WR (2013) Rapid detection of serum antibody by dual-path platform VetTB assay in white-tailed deer infected with Mycobacterium bovis. Clin Vaccine Immunol 20:907–911. https://doi.org/10.1128/CVI.00120-13
Lyashchenko KP, Greenwald R, Esfandiari J, Rhodes S, Dean G, de la Rua-Domenech R, Meylan M, Vordermeier HM, Zanolari P (2011) Diagnostic value of animal-side antibody assays for rapid detection of Mycobacterium bovis or Mycobacterium microti infection in South American camelids. Clin Vaccine Immunol 18:2143–2147. https://doi.org/10.1128/CVI.05386-11
Lyashchenko KP, Greenwald R, Esfandiari J, Greenwald D, Nacy CA, Gibson S, Didier PJ, Washington M, Szczerba P, Motzel S, Handt L, Pollock JM, McNair J, Andersen P, Langermans JA, Verreck F, Ervin S, Ervin F, McCombs C (2007) PrimaTB STAT-PAK assay, a novel, rapid lateral-flow test for tuberculosis in nonhuman primates. Clin Vaccine Immunol 14:1158–1164. https://doi.org/10.1128/CVI.00230-07
Cousins DV, Florisson N (2005) A review of tests available for use in the diagnosis of tuberculosis in non-bovine species. Rev Sci Technol 24:1039–1059. PMID:16642773
Chambers MA, Waterhouse S, Lyashchenko K, Delahay R, Sayers R, Hewinson RG (2009) Performance of TB immunodiagnostic tests in Eurasian badgers (Meles meles) of different ages and the influence of duration of infection on serological sensitivity. BMC Vet Res 5:42. https://doi.org/10.1186/1746-6148-5-42
Acknowledgements
We thank Karnataka Forests Department and Wildlife SOS for helping us with Sloth Bear sera samples. TRPVB acknowledges the DBT-BBSRC FADH funding scheme code:BT/IN/indo-uk/FADH/52/VM/2013 project titled “Development of recombinant BGG vaccine and complimentary diagnosis for TB control in cattle”.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The author(s) declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Veerasami, M., Venkataraman, K., Karuppannan, C. et al. Point of Care Tuberculosis Sero-Diagnosis Kit for Wild Animals: Combination of Proteins for Improving the Diagnostic Sensitivity and Specificity. Indian J Microbiol 58, 81–92 (2018). https://doi.org/10.1007/s12088-017-0688-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-017-0688-7