Abstract
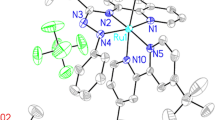
Two novel mononuclear heteroleptic Ru(II) photosensitizers with 2-(1H-benzo[d]imidazol-2-yl)quinoline derivatives were designed and successfully synthesized. The facile synthesis and their photophysical properties are investigated. Both the Ru(II) complexes i.e., [RuII(bpy)2(L1)](ClO4) : [1](ClO4) and [RuII(bpy)2(L2)](ClO4)2 : [2](ClO4)2 {L1 = 2-(1H-benzo[d]imidazol-2-yl)quinoline and L2 = 2-(1-methyl-1H-benzo[d]imidazol-2-yl)quinoline} have been meticulously characterized by different spectroscopic and analytical techniques such as FT–IR, 1H NMR, ESI mass spectra, UV–vis and fluorescence spectroscopy, etc. Molecular structures of [1](ClO4) and [2](ClO4)2 have been determined by a single-crystal X-ray structure diffraction study. Redox and spectral properties of the synthesized Ru(II) complexes were examined along with their corresponding ligands and compared with the classic homoleptic [RuII(bpy)3](PF6)2. The effects on substituents in the ligand backbone were scrutinised. The emission behaviour of both [1](ClO4) and [2](ClO4)2 revealed relatively long-lived emissive 3MLCT and bathochromic shift (~ 715 nm) while compared with [RuII(bpy)3](PF6)2 (~ 605 nm). Fairly weak quantum yields for [1](ClO4) and [2](ClO4)2 : ∼ 0.00299 and ∼ 0.00295 with half-lives 181.57 ns and 198.89 ns, respectively, suggested different non-radiative emission pathways. Additionally, for [1](ClO4) and [2](ClO4)2, electrochemical reduction of carbon dioxide (CO2) in dry acetonitrile solvent was performed and showed great promises for future designing of electrochemical reduction of CO2.
Graphical abstract
Two mononuclear heteroleptic Ru(II) photosensitizers [1](ClO4) and [2](ClO4)2 containing 2-(1H-benzo[d]imidazol-2-yl)quinoline ligand were synthesized and investigated via FT–IR, 1H NMR, ESI mass spectra, UV–vis, time-resolved photoluminescence spectroscopy, X-ray structure, etc. Both [1](ClO4) and [2](ClO4)2 showed great promises for electrochemical reduction of carbon dioxide in dry acetonitrile.










Similar content being viewed by others
References
Smith P T, Nichols E M, Cao Z and Chang C J 2020 Hybrid Catalysts for Artificial Photosynthesis: Merging Approaches from Molecular, Materials, and Biological Catalysis Acc. Chem. Res. 53 575
Waki M, Shirai S, Yamanaka K et al 2020 Heterogeneous water oxidation photocatalysis based on periodic mesoporous organosilica immobilizing a tris(2,2′-bipyridine)ruthenium sensitizer RSC Adv. 10 13960
Motimani N M, Ngubane S and Smith G S 2022 Polynuclear heteroleptic ruthenium(II) photoredox catalysts: Evaluation in blue-light-mediated, regioselective thiol-ene reactions Polyhedron 212 115616
Hoffmann N 2008 Photochemical Reactions as Key Steps in Organic Synthesis Chem. Rev. 108 1052
Kaur N 2018 Photochemical reactions as key steps in five-membered N-heterocycle synthesis Synth. Commun. 48 1259
Narayanam J M R, Tucker J W and Stephenson C R J 2009 Electron-Transfer Photoredox Catalysis: Development of a Tin-Free Reductive Dehalogenation Reaction J. Am. Chem. Soc. 131 8756
Prier C K, Rankic D A and MacMillan D W C 2013 Visible Light Photoredox Catalysis with Transition Metal Complexes: Applications in Organic Synthesis Chem. Rev. 113 5322
Su Y, Straathof N J W, Hessel V and Noël T 2014 Photochemical Transformations Accelerated in Continuous-Flow Reactors: Basic Concepts and Applications Chem. Eur. J. 20 10562
Chen J, Cen J, Xu X and Li X 2016 The application of heterogeneous visible light photocatalysts in organic synthesis Catal. Sci. Technol. 6 349
Xuan J and Xiao W-J 2012 Visible-Light Photoredox Catalysis Angew. Chem. Int. Ed. 51 6828
Shaw M H, Twilton J and MacMillan D W C 2016 Photoredox Catalysis in Organic Chemistry J. Org. Chem. 81 6898
Hockin B M, Li C, Robertson N and Zysman-Colman E 2019 Photoredox catalysts based on earth-abundant metal complexes Catal. Sci. Technol. 9 889
McCusker J K 2019 Electronic structure in the transition metal block and its implications for light harvesting Science 363 484
Noyori R 2009 Synthesizing our future Nature Chem. 1 5
Gualandi A, Matteucci E, Monti F et al 2017 Photoredox radical conjugate addition of dithiane-2-carboxylate promoted by an iridium(III) phenyl-tetrazole complex: a formal radical methylation of Michael acceptors Chem. Sci. 8 1613
Huo H, Wang C, Harms K and Meggers E 2015 Enantioselective, Catalytic Trichloromethylation through Visible-Light-Activated Photoredox Catalysis with a Chiral Iridium Complex J. Am. Chem. Soc. 137 9551
Tyson E L, Ament M S and Yoon T P 2013 Transition Metal Photoredox Catalysis of Radical Thiol-Ene Reactions J. Org. Chem. 78 2046
Fabry D C, Ronge M A, Zoller J and Rueping M 2015 C–H Functionalization of Phenols Using Combined Ruthenium and Photoredox Catalysis: In Situ Generation of the Oxidant. Angew. Chem. Int. Ed. 54 2801
Caspar J V and Meyer T J 1983 Photochemistry of tris(2,2/-bipyridine)ruthenium(2+) ion (Ru(bpy)32+). Solvent effects J. Am. Chem. Soc. 105 5583
Cruz A J, Kirgan R, Siam K et al 2010 Photochemical and photophysical properties of ruthenium(II) bis-bipyridine bis-nitrile complexes: Photolability Inorg. Chim. Acta 363 2496
Soupart A, Dixon I M, Alary F and Heully J-L 2018 DFT rationalization of the room-temperature luminescence properties of Ru(bpy)32+ and Ru(tpy)22+: 3MLCT–3MC minimum energy path from NEB calculations and emission spectra from VRES calculations Theor. Chem. Acc. 137 37
Dongare P, Myron B D B, Wang L et al 2017 [Ru(bpy)3]2+∗ revisited. Is it localized or delocalized? How does it decay? Coord. Chem. Rev. 345 86
Sun Y, Hudson Z M, Rao Y and Wang S 2011 Tuning and Switching MLCT Phosphorescence of [Ru(bpy)3]2+ Complexes with Triarylboranes and Anions Inorg. Chem. 50 3373
Sun Q, Dereka B, Vauthey E et al 2017 Ultrafast transient IR spectroscopy and DFT calculations of ruthenium(II) polypyridyl complexes Chem. Sci. 8 223
Xu S, Smith J E T and Weber J M 2016 The electronic spectrum of cryogenic ruthenium-tris-bipyridine dications in vacuo J. Chem. Phys. 145 024304
Thompson D W, Ito A and Meyer T J 2013 [Ru(bpy)3]2+* and other remarkable metal-to-ligand charge transfer (MLCT) excited states Pure Appl. Chem. 85 1257
Muniz-Miranda F, De Bruecker L, De Vos A et al 2019 Optical Properties of Isolated and Covalent Organic Framework-Embedded Ruthenium Complexes J. Phys. Chem. A 123 6854
Pal A K and Hanan G S 2014 Design, synthesis and excited-state properties of mononuclear Ru(II) complexes of tridentate heterocyclic ligands Chem. Soc. Rev. 43 6184
Balzani V, Ceroni P, Credi A and Venturi M 2021 Ruthenium tris(bipyridine) complexes: Interchange between photons and electrons in molecular-scale devices and machines Coord. Chem. Rev. 433 213758
Balzani V, Ceroni P, Juris A et al 2001 Dendrimers based on photoactive metal complexes. Recent advances Coord. Chem. Rev. 219–221 545
Motley T C, Troian-Gautier L, Brennaman M K and Meyer G J 2017 Excited-State Decay Pathways of Tris(bidentate) Cyclometalated Ruthenium(II) Compounds Inorg. Chem. 56 13579
Mede T, Jäger M and Schubert U S 2018 “Chemistry-on-the-complex”: functional RuII polypyridyl-type sensitizers as divergent building blocks Chem. Soc. Rev. 47 7577
O’Regan B and Grätzel M 1991 A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films Nature 353 737
Chung I, Lee B, He J et al 2012 All-solid-state dye-sensitized solar cells with high efficiency Nature 485 486
Rajab F M 2018 Assessment of Ruthenium Dye N719 Adsorption Kinetics in Mesoporous TiO2 Films of Dye-Sensitized Solar Cells via Nanoplasmonic Sensing J. Nanotechnol. 2018 e9808697
Portillo-Cortez K, Martínez A, Dutt A and Santana G 2019 N719 Derivatives for Application in a Dye-Sensitized Solar Cell (DSSC): A Theoretical Study J. Phys. Chem. A. 123 10930
Hussain M, El-Shafei A, Islam A and Han L 2013 Structure–property relationship of extended π-conjugation of ancillary ligands with and without an electron donor of heteroleptic Ru(II) bipyridyl complexes for high efficiency dye-sensitized solar cells Phys. Chem. Chem. Phys. 15 8401
Anderson P A, Strouse G F, Treadway J A et al 1994 Black MLCT Absorbers Inorg. Chem. 33 3863
Arias-Rotondo D M and McCusker J K 2016 The photophysics of photoredox catalysis: a roadmap for catalyst design Chem. Soc. Rev. 45 5803
Chen G, Waterhouse G I N, Shi R et al 2019 From Solar Energy to Fuels: Recent Advances in Light-Driven C1 Chemistry Angew. Chem. Int. Ed. 58 17528
Appel A M, Bercaw J E, Bocarsly A B et al 2013 Frontiers, Opportunities, and Challenges in Biochemical and Chemical Catalysis of CO2 Fixation Chem. Rev. 113 6621
Benson E E, Kubiak C P, Sathrum A J and Smieja J M 2008 Electrocatalytic and homogeneous approaches to conversion of CO2 to liquid fuels Chem. Soc. Rev. 38 89
Mikkelsen M, Jørgensen M and Krebs F C 2010 The teraton challenge. A review of fixation and transformation of carbon dioxide Energy Environ. Sci. 3 43
Francke R, Schille B and Roemelt M 2018 Homogeneously Catalyzed Electroreduction of Carbon Dioxide—Methods, Mechanisms, and Catalysts Chem. Rev. 118 4631
Goeppert A, Czaun M, Jones J-P et al 2014 Recycling of carbon dioxide to methanol and derived products-closing the loop Chem. Soc. Rev. 43 7995
Zhang S, Li L, Zhao S et al 2015 Construction of Interpenetrated Ruthenium Metal-Organic Frameworks as Stable Photocatalysts for CO2 Reduction Inorg. Chem. 54 8375
Zhang S, Li L, Zhao S et al 2015 Hierarchical metal–organic framework nanoflowers for effective CO2 transformation driven by visible light J. Mater. Chem. A 3 15764
Johnson B A, Agarwala H, White T A et al 2016 Judicious Ligand Design in Ruthenium Polypyridyl CO2 Reduction Catalysts to Enhance Reactivity by Steric and Electronic Effects Chem. Eur. J. 22 14870
White T, Maji S and Ott S 2014 Mechanistic insights into electrocatalytic CO2 reduction within [RuII(tpy)(NN)X]n+ architectures Dalton Trans. 43 15028
Benzimidazole Derivative - an overview | ScienceDirect Topics. https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/benzimidazole-derivative. Accessed 22 Feb 2022
Mamedov V A 2016 Recent advances in the synthesis of benzimidazol(on)es via rearrangements of quinoxalin(on)es RSC Adv. 6 42132
Vasava M S, Bhoi M N, Rathwa S K et al 2020 Benzimidazole: A Milestone in the Field of Medicinal Chemistry Mini Rev. Med. Chem. 20 532
Phillips M A 1928 CCCXVII.—The formation of 2-substituted benziminazoles J. Chem. Soc. 2393
McDevitt M R, Ru Y and Addison A W 1993 Ruthenium(II) complexes with phenanthroline-, benzimidazole-, benzothiazole-, and pyridine-derived bidentate and tridentate ligands: reactivity and spectroscopic and electrochemical characterization Transit. Met. Chem. 18 197
Mamedov V A, Saifina D F, Gubaidullin A T, et al. 2010 Acid-catalyzed rearrangement of 3-(β-2-aminostyryl)quinoxalin-2(1H)ones—a new and efficient method for the synthesis of 2-benzimidazol-2-ylquinolines Tetrahedron Lett. 51 6503
Sprintschnik G, Sprintschnik H W, Kirsch P P, Whitten D G 1977 Photochemical reactions in organized monolayer assemblies. 6. Preparation and photochemical reactivity of surfactant ruthenium(II) complexes in monolayer assemblies and at water-solid interfaces J. Am. Chem. Soc. 99 4947
Sheldrick G M 2015 SHELXT – Integrated space-group and crystal-structure determination Acta Cryst. A 71 3
Dolomanov O V, Bourhis L J, Gildea R J et al. 2009 OLEX2: a complete structure solution, refinement and analysis program J. Appl. Cryst. 42 339
Maji S, Sarkar B, Patra M et al 2008 Formation, Reactivity, and Photorelease of Metal Bound Nitrosyl in [Ru(trpy)(L)(NO)]n+ (trpy = 2,2′:6′,2′′-Terpyridine, L = 2-Phenylimidazo[4,5-f]1,10-phenanthroline) Inorg. Chem. 47 3218
De P, Sarkar B, Maji S et al 2009 Stabilization of RuNO6 and RuNO7 States in [RuII(trpy)(bik)(NO)]n+ trpy = 2,2′:6′,2″-terpyridine, bik = 2,2′-bis(1-methylimidazolyl) ketone – Formation, Reactivity, and Photorelease of Metal-Bound Nitrosyl Eur. J. Inorg. Chem. 2009 2702
Giri B, Kumbhakar S, Kalai Selvan K and Muley A 2020 Formation, reactivity, photorelease, and scavenging of NO in ruthenium nitrosyl complexes Inorg. Chim. Acta 502 119360
Reveco P, Schmehl R H, Cherry W R, et al. 1985 Cyclometalated complexes of ruthenium. 2. Spectral and electrochemical properties and x-ray structure of bis(2,2’-bipyridine)(4-nitro-2-(2-pyridyl)phenyl)ruthenium(II) Inorg. Chem. 24 4078
Aoki K, Morita M, Niwa O and Tabei H 1988 Quantitative analysis of reversible diffusion-controlled currents of redox soluble species at interdigitated array electrodes under steady-state conditions J. Electroanal. Chem. Interf. Electrochem. 256 269
Wu S-H, Shao J-Y, Dai X et al 2017 Synthesis and Characterization of Tris(bidentate) Ruthenium Complexes of Di(pyrid-2-yl)(methyl)amine Eur. J. Inorg. Chem. 2017 3064
Rousset E, Ciofini I, Marvaud V and Hanan G S 2017 Facile One-Pot Synthesis of Ruthenium(II) Quaterpyridine-Based Photosensitizers for Photocatalyzed Hydrogen Production Inorg. Chem. 56 9515
Juris A, Balzani V, Barigelletti F et al. 1988 Ru(II) polypyridine complexes: photophysics, photochemistry, eletrochemistry, and chemiluminescence Coord. Chem. Rev. 84 85
Williams A T R, Winfield S A and Miller J N 1983 Relative fluorescence quantum yields using a computer-controlled luminescence spectrometer Analyst 108 1067
Ishida H, Tobita S, Hasegawa Y et al. 2010 Recent advances in instrumentation for absolute emission quantum yield measurements Coord. Chem. Rev. 254 2449
Chen Z, Chen C, Weinberg D R et al. 2011 Electrocatalytic reduction of CO2 to CO by polypyridyl ruthenium complexes Chem. Commun. 47 12607
Johnson B A, Maji S, Agarwala H et al 2016 Activating a Low Overpotential CO2 Reduction Mechanism by a Strategic Ligand Modification on a Ruthenium Polypyridyl Catalyst Angew. Chem. Int. Ed. 55 1825
Chen Z, Kang P, Zhang M-T and Meyer T J 2013 Making syngas electrocatalytically using a polypyridyl ruthenium catalyst Chem. Commun. 50 335
Randles J E B 1948 A cathode ray polarograph. Part II.—The current-voltage curves Trans. Faraday Soc. 44 327
Comminges C, Barhdadi R, Laurent M and Troupel M 2006 Determination of Viscosity, Ionic Conductivity, and Diffusion Coefficients in Some Binary Systems: Ionic Liquids + Molecular Solvents J. Chem. Eng. Data 51 680
Ali M M, Sato H, Mizukawa T, et al. 1998 Selective formation of HCO2– and C2O42– in electrochemical reduction of CO2 catalyzed by mono- and di-nuclear ruthenium complexes Chem. Commun. 249
Begum A and Pickup P G 2007 Electrocatalysis of CO2 reduction by ruthenium benzothiazole and bithiazole complexes Electrochem. Commun. 9 2525
Acknowledgements
Financial support received from SERB, project no. ECR/2016/000382 (fellowship to S.K.), Council of Scientific and Industrial Research (CSIR) (fellowship to B.G., A.M. and K.S.) and IIT Hyderabad are gratefully acknowledged for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Supplementary Information
Supplementary data (experimental procedures and additional experimental data) associated with this article can be found at www.ias.ac.in/chemsci.
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumbhakar, S., Giri, B., Muley, A. et al. Synthesis, characterization, structural and photophysical properties of heteroleptic ruthenium complexes containing 2-(1H-benzo[d]imidazol-2-yl)quinoline ligand towards electrocatalytic CO2 reduction. J Chem Sci 134, 71 (2022). https://doi.org/10.1007/s12039-022-02063-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-022-02063-z