Abstract
Lewis acidity of SbCl3 and BiCl3 has been investigated in the solid as well as in solution state. Two β-ketoimine ligands, [O=C(Me)]CH[C(Me)NHAr] (L1, Ar=2,6-diisopropylphenyl) and [(CH2)2{N(H)C(Me)CHC(Me)=O}2] (L2) are used to prepare the complexes [(L1)(THF)SbCl3]2 (1), [(L1)(THF)BiCl3]2 (2), [L2SbCl3]2 (3) and [(L2)3(BiCl3)2] (4), which are characterized by multinuclear NMR spectroscopy, elemental analysis and single-crystal X-ray diffraction experiments. Solid-state structural analysis of 1–4 and solution studies on SbCl3 and BiCl3 using the Gutmann-Beckett method reveal the higher Lewis acidity of BiCl3 compared to SbCl3.
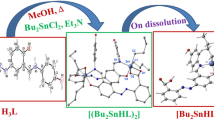
Graphic abstract
BiCl3 is more Lewis acidic than SbCl3 as evidenced by Gutmann-Beckett analysis and investigation into the solid-state structures of β-ketoimine complexes by single-crystal X-ray diffraction experiments.





Similar content being viewed by others
References
(a) Ollevier T 2013 New trends in bismuth-catalyzed synthetic transformations Org. Biomol. Chem. 11 2740; (b) Bothwell J M, Scott W, Krabbe S W and Mohan R S 2011 Applications of bismuth(III) compounds in organic synthesis Chem. Soc. Rev. 40 4649
Norman N C 1994 Coordination chemistry of antimony and bismuth: Lewis acidity, σ ∗ -orbitals and coordination geometry Phosph. Sulfur Silicon Relat. Elem. 87 167
Kannan R, Kumar S, Andrews A P, Jemmis E D and Venugopal A 2017 Consequence of Ligand bite angle on bismuth Lewis acidity Inorg. Chem. 56 9391
Balasubramaniam S, Kumar S, Andrews A P, Varghese B, Jemmis E D and Venugopal A 2019 A dicationic bismuth(III) Lewis acid: catalytic hydrosilylation of olefins Eur. J. Inorg. Chem. 3265
Balasubramaniam S, Varghese B and Venugopal A 2019 Bismuth(III) dication trapped on a chlorobismuthate J. Coord. Chem. 72 1417
(a) Chitnis S S, Vos K A, Burford N, McDonald R and Ferguson M J 2016 Distinction between coordination and phosphine ligand oxidation: interactions of di- and triphosphines with Pn3+ (Pn = P, As, Sb, Bi) Chem. Commun. 52 685; (b) Chitnis S S, Robertson A P M, Burford N, Patrick B O, McDonald R and Ferguson M J 2015 Bipyridine complexes of E3+ (E = P, As, Sb, Bi): strong Lewis acids, sources of E(OTf)3 and synthons for EI and EV cations Chem. Sci. 6 6545; (c) Robertson A P M, Burford N, Gray P A and Ferguson R 2014 Interpnictogen cations: exploring new vistas in coordination chemistry Angew. Chem. Int. Ed. 53 6050; (d) Chitnis S S, Burford N, Decken A and Ferguson M J 2013 Coordination complexes of bismuth triflates with tetrahydrofuran and diphosphine ligands Inorg. Chem. 52 7242
(a) Beckett M A, Strickland G C, Holland J R and Varma K S 1996 A convenient n.m.r. method for the measurement of Lewis acidity at boron centres: correlation of reaction rates of Lewis acid initiated epoxide polymerizations with Lewis acidity Polymer 37 4629; (b) Mayer U, Gutmann V and Gerger W 1975 The acceptor number—A quantitative empirical parameter for the electrophilic properties of solvents Monatsh. Chem. 106 1235
Everett Jr G W and Holm R H 1965 The synthesis and proton resonance study of the solution equilibria of bis(β-ketoamino) nickel(II) complexes J. Am. Chem. Soc. 87 2117
Gogoi S, Bhuyan R and Barua N C 2005 Iodine-catalyzed conversion of β-dicarbonyl compounds into β-enaminones within a minute under solvent-free conditions Synth. Commun. 35 2811
(a) Altomare A, Cascarano G, Giacovazzo C and Guagliardi A 1994 J. Appl. Crystallogr. 27 435; (b) Sheldrick G M 2008 A short history of SHELX Acta Cryst. A64 112
Johnson J A, Ashokan A, Chalana A K, Andrews A P and Venugopal A 2017 Neutral and cationic β-ketoiminato bismuth complexes Z. Anorg. Allg. Chem. 643 607
Cordero B, Gomez V, Platero-prats A E, Rev M, Echeverr J, Cremades E, Barrag F and Alverez S 2008 Covalent radii revisited Dalton Trans. 2832
Adamczyk-wozniak A, Jakubczyk M, Sporzyński A and Żukowska G 2011 Quantitative determination of the Lewis acidity of phenylboronic catechol esters—Promising anion receptors for polymer electrolytes Inorg. Chem. Commun. 14 1753
Sivaev I B and Bregadze V I 2014 C–H Bond activation of arenes by [8,8′-μ-I-3,3′-Co(1,2-C2B9H10)2] in the presence of sterically hindered Lewis bases Coord. Chem. Rev. 270–271 75
(a) Tsao F A, Waked A E, Cao L, Hofmann J, Liu L, Grimme S and Stephan D W 2016 S(VI) Lewis acids: fluorosulfoxonium cations Chem. Commun. 52 12418; (b) Großekappenberg H, Reißmann M, Schmidtmann M and Müller T 2015 Quantitative assessment of the Lewis acidity of silylium ions Organometallics 34 4952; (c) Pan B and Gabbaï F P 2014 [Sb(C6F5)4][B(C6F5)4]: An air stable, Lewis acidic stibonium salt that activates strong element-fluorine bonds J. Am. Chem. Soc. 136 9564; (d) Caputo C B, Hounjet L J, Dobrovetsky R and Stephan D W 2013 Lewis acidity of organofluorophosphonium salts: hydrodefluorination by a saturated acceptor Science 341 1374
Acknowledgements
We thank the Council of Scientific and Industrial Research (Grant Number: 01(2777)/14/EMR-II) for the generous funding. Alex P. Andrews is thanked for X-ray diffraction experiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue on 150 years of the Periodic Table
Rights and permissions
About this article
Cite this article
Johnson, J.A., Venugopal, A. Probing the Lewis acidity of heavier pnictogen trichlorides. J Chem Sci 131, 114 (2019). https://doi.org/10.1007/s12039-019-1706-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-019-1706-6