Abstract
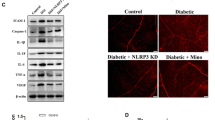
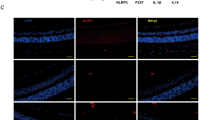
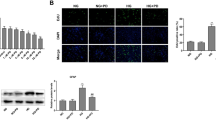
Oxidative stress and neuroinflammation contribute significantly to the development and progression of diabetic retinopathy. Fenofibrate has received great attention as it benefits diabetic patients by reducing retinal laser requirement. Nuclear factor erythroid-2-related factor 2 (Nrf2) is a master regulator of anti-oxidative defense. Activation of nucleotide binding domain, leucine-rich repeat-containing receptor (NLR), pyrin domain-containing 3 (NLRP3) inflammasome plays a pivotal role in neuroinflammation. The purpose of this study is to determine whether fenofibrate protects retinas from oxidative damage and neuroinflammation via modulating the Nrf2 pathway and blocking NLRP3 inflammasome activation during diabetes. Diabetes is induced by intraperitoneal injection of streptozotocin in mice. Fenofibrate was given to mice in rodent chow. Upregulation of Nrf2 and NLRP3 inflammasome, enhanced ROS formation, and increased leukostasis and vascular leakage were observed in diabetic mouse retinas. Notably, Nrf2 and Caspase-1 were mainly colocalized with glutamine synthetase, one of the Mȕller cell markers. Fenofibrate further increased the expression of Nrf2 and its target gene NQO-1 and HO-1 and reduced ROS formation in diabetic retinas. In addition, retinal expression of NLRP3, Caspase-1 p20, IL-1β p17, and ICAM-1 were dramatically increased in vehicle-treated diabetic mice, which were abolished by fenofibrate intervention. Moreover, fenofibrate treatment also attenuated diabetes-induced retinal leukostasis and vascular leakage in mice. Taken together, fenofibrate attenuates oxidative stress and neuroinflammation in diabetic retinas, which is at least partially through modulating Nrf2 expression and NLRP3 inflammasome activation.





Similar content being viewed by others
References
Keech AC, Mitchell P, Summanen PA, O’Day J, Davis TM, Moffitt MS, Taskinen MR, Simes RJ, Tse D, Williamson E, Merrifield A, Laatikainen LT, d’Emden MC, Crimet DC, O’Connell RL, Colman PG, Investigators FS (2007) Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): a randomised controlled trial. Lancet 370:1687–1697. https://doi.org/10.1016/S0140-6736(07)61607-9
Group AS, Group AES, Chew EY, Ambrosius WT, Davis MD, Danis RP, Gangaputra S, Greven CM, Hubbard L, Esser BA, Lovato JF, Perdue LH, Goff DC Jr., Cushman WC, Ginsberg HN, Elam MB, Genuth S, Gerstein HC, Schubart U, Fine LJ (2010) Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 363:233–244. https://doi.org/10.1056/NEJMoa1001288
Noonan JE, Jenkins AJ, Ma JX, Keech AC, Wang JJ, Lamoureux EL (2013) An update on the molecular actions of fenofibrate and its clinical effects on diabetic retinopathy and other microvascular end points in patients with diabetes. Diabetes 62:3968–3975. https://doi.org/10.2337/db13-0800
Brownlee M (2005) The pathobiology of diabetic complications: a unifying mechanism. Diabetes 54:1615–1625
Cheng Y, Zhang J, Guo W, Li F, Sun W, Chen J, Zhang C, Lu X, Tan Y, Feng W, Fu Y, Liu GC, Xu Z, Cai L (2016) Up-regulation of Nrf2 is involved in FGF21-mediated fenofibrate protection against type 1 diabetic nephropathy. Free Radic Biol Med 93:94–109. https://doi.org/10.1016/j.freeradbiomed.2016.02.002
Ibarra-Lara L, Hong E, Soria-Castro E, Torres-Narvaez JC, Perez-Severiano F, Del Valle-Mondragon L, Cervantes-Perez LG, Ramirez-Ortega M, Pastelin-Hernandez GS, Sanchez-Mendoza A (2012) Clofibrate PPARalpha activation reduces oxidative stress and improves ultrastructure and ventricular hemodynamics in no-flow myocardial ischemia. J Cardiovasc Pharmacol 60:323–334. https://doi.org/10.1097/FJC.0b013e31826216ed
Ruiz S, Pergola PE, Zager RA, Vaziri ND (2013) Targeting the transcription factor Nrf2 to ameliorate oxidative stress and inflammation in chronic kidney disease. Kidney Int 83:1029–1041. https://doi.org/10.1038/ki.2012.439
Chen WJ, Wu C, Xu Z, Kuse Y, Hara H, Duh EJ (2017) Nrf2 protects photoreceptor cells from photo-oxidative stress induced by blue light. Exp Eye Res 154:151–158. https://doi.org/10.1016/j.exer.2016.12.001
Ogura Y, Sutterwala FS, Flavell RA (2006) The inflammasome: first line of the immune response to cell stress. Cell 126:659–662. https://doi.org/10.1016/j.cell.2006.08.002
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J (2010) Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol 11:136–140. https://doi.org/10.1038/ni.1831
Loukovaara S, Piippo N, Kinnunen K, Hytti M, Kaarniranta K, Kauppinen A (2017) NLRP3 inflammasome activation is associated with proliferative diabetic retinopathy. Acta Ophthalmol. https://doi.org/10.1111/aos.13427
Deng Y, Han X, Yao Z, Sun Y, Yu J, Cai J, Ren G, Jiang G, Han F (2017) PPARalpha agonist stimulated angiogenesis by improving endothelial precursor cell function via a NLRP3 inflammasome pathway. Cell Physiol Biochem 42:2255–2266. https://doi.org/10.1159/000479999
Zhong Y, Li J, Chen Y, Wang JJ, Ratan R, Zhang SX (2012) Activation of endoplasmic reticulum stress by hyperglycemia is essential for Muller cell-derived inflammatory cytokine production in diabetes. Diabetes 61:492–504. https://doi.org/10.2337/db11-0315
Cheng R, Ding L, He X, Takahashi Y, Ma JX (2016) Interaction of PPARalpha with the canonic Wnt pathway in the regulation of renal fibrosis. Diabetes 65:3730–3743. https://doi.org/10.2337/db16-0426
Li J, Wang JJ, Yu Q, Chen K, Mahadev K, Zhang SX (2010) Inhibition of reactive oxygen species by Lovastatin downregulates vascular endothelial growth factor expression and ameliorates blood-retinal barrier breakdown in db/db mice: role of NADPH oxidase 4. Diabetes 59:1528–1538. https://doi.org/10.2337/db09-1057
Akiyama G, Azuchi Y, Guo X, Noro T, Kimura A, Harada C, Namekata K, Harada T (2017) Edaravone prevents retinal degeneration in adult mice following optic nerve injury. Invest Ophthalmol Vis Sci 58:4908–4914. https://doi.org/10.1167/iovs.17-22250
Li J, Wang JJ, Zhang SX (2011) Preconditioning with endoplasmic reticulum stress mitigates retinal endothelial inflammation via activation of X-box binding protein 1. J Biol Chem 286:4912–4921. https://doi.org/10.1074/jbc.M110.199729
Lewis GP, Fisher SK (2003) Up-regulation of glial fibrillary acidic protein in response to retinal injury: its potential role in glial remodeling and a comparison to vimentin expression. Int Rev Cytol 230:263–290
Du Y, Veenstra A, Palczewski K, Kern TS (2013) Photoreceptor cells are major contributors to diabetes-induced oxidative stress and local inflammation in the retina. Proc Natl Acad Sci USA 110:16586–16591. https://doi.org/10.1073/pnas.1314575110
No Authors (1987) Indications for photocoagulation treatment of diabetic retinopathy: Diabetic Retinopathy Study Report no. 14. The Diabetic Retinopathy Study Research Group. Int Ophthalmol Clin 27:239–253
No Authors (1991) Early photocoagulation for diabetic retinopathy. ETDRS report number 9. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 98:766–785
Wei Y, Gong J, Xu Z, Duh EJ (2016) Nrf2 promotes reparative angiogenesis through regulation of NADPH oxidase-2 in oxygen-induced retinopathy. Free Radic Biol Med 99:234–243. https://doi.org/10.1016/j.freeradbiomed.2016.08.013
Wei Y, Gong J, Xu Z, Thimmulappa RK, Mitchell KL, Welsbie DS, Biswal S, Duh EJ (2015) Nrf2 in ischemic neurons promotes retinal vascular regeneration through regulation of semaphorin 6A. Proc Natl Acad Sci USA 112:E6927–E6936. https://doi.org/10.1073/pnas.1512683112
Xu Z, Wei Y, Gong J, Cho H, Park JK, Sung ER, Huang H, Wu L, Eberhart C, Handa JT, Du Y, Kern TS, Thimmulappa R, Barber AJ, Biswal S, Duh EJ (2014) NRF2 plays a protective role in diabetic retinopathy in mice. Diabetologia 57:204–213. https://doi.org/10.1007/s00125-013-3093-8
Inoue Y, Shimazawa M, Noda Y, Nagano R, Otsuka T, Kuse Y, Nakano Y, Tsuruma K, Nakagami Y, Hara H (2017) RS9, a novel Nrf2 activator, attenuates light-induced death of cells of photoreceptor cells and Muller glia cells. J Neurochem 141:750–765. https://doi.org/10.1111/jnc.14029
Sun Y, Xiu C, Liu W, Tao Y, Wang J, Qu YI (2016) Grape seed proanthocyanidin extract protects the retina against early diabetic injury by activating the Nrf2 pathway. Exp Ther Med 11:1253–1258. https://doi.org/10.3892/etm.2016.3033
Park JS, Kang DH, Lee DH, Bae SH (2015) Fenofibrate activates Nrf2 through p62-dependent Keap1 degradation. Biochem Biophys Res Commun 465:542–547. https://doi.org/10.1016/j.bbrc.2015.08.056
Ding L, Cheng R, Hu Y, Takahashi Y, Jenkins AJ, Keech AC, Humphries KM, Gu X, Elliott MH, Xia X, Ma JX (2014) Peroxisome proliferator-activated receptor alpha protects capillary pericytes in the retina. Am J Pathol 184:2709–2720. https://doi.org/10.1016/j.ajpath.2014.06.021
Chen Y, Hu Y, Lin M, Jenkins AJ, Keech AC, Mott R, Lyons TJ, Ma JX (2013) Therapeutic effects of PPARalpha agonists on diabetic retinopathy in type 1 diabetes models. Diabetes 62:261–272. https://doi.org/10.2337/db11-0413
Bringmann A, Pannicke T, Grosche J, Francke M, Wiedemann P, Skatchkov SN, Osborne NN, Reichenbach A (2006) Muller cells in the healthy and diseased retina. Prog Retin Eye Res 25:397–424. https://doi.org/10.1016/j.preteyeres.2006.05.003
Sorrentino FS, Allkabes M, Salsini G, Bonifazzi C, Perri P (2016) The importance of glial cells in the homeostasis of the retinal microenvironment and their pivotal role in the course of diabetic retinopathy. Life Sci 162:54–59. https://doi.org/10.1016/j.lfs.2016.08.001
Moran EP, Wang Z, Chen J, Sapieha P, Smith LE, Ma JX (2016) Neurovascular cross talk in diabetic retinopathy: pathophysiological roles and therapeutic implications. Am J Physiol Heart Circ Physiol 311:H738–H749. https://doi.org/10.1152/ajpheart.00005.2016
Hotamisligil GS (2006) Inflammation and metabolic disorders. Nature 444:860–867. https://doi.org/10.1038/nature05485
Rampanelli E, Orso E, Ochodnicky P, Liebisch G, Bakker PJ, Claessen N, Butter LM, van den Bergh Weerman MA, Florquin S, Schmitz G, Leemans JC (2017) Metabolic injury-induced NLRP3 inflammasome activation dampens phospholipid degradation. Sci Rep 7:2861. https://doi.org/10.1038/s41598-017-01994-9
Chen W, Zhao M, Zhao S, Lu Q, Ni L, Zou C, Lu L, Xu X, Guan H, Zheng Z, Qiu Q (2017) Activation of the TXNIP/NLRP3 inflammasome pathway contributes to inflammation in diabetic retinopathy: a novel inhibitory effect of minocycline. Inflamm Res 66:157–166. https://doi.org/10.1007/s00011-016-1002-6
Yin Y, Chen F, Wang W, Wang H, Zhang X (2017) Resolvin D1 inhibits inflammatory response in STZ-induced diabetic retinopathy rats: Possible involvement of NLRP3 inflammasome and NF-kappaB signaling pathway. Mol Vis 23:242–250
Eastlake K, Banerjee PJ, Angbohang A, Charteris DG, Khaw PT, Limb GA (2016) Muller glia as an important source of cytokines and inflammatory factors present in the gliotic retina during proliferative vitreoretinopathy. Glia 64:495–506. https://doi.org/10.1002/glia.22942
Le YZ (2017) VEGF production and signaling in Muller glia are critical to modulating vascular function and neuronal integrity in diabetic retinopathy and hypoxic retinal vascular diseases. Vis Res. https://doi.org/10.1016/j.visres.2017.05.005
Funding
This study was supported by NSFC Grants 81400427, 81460163, 81741058 and 81300786; Young Talent Scholar Grant 2016KJXX-12; FRFCU Grant xjj2015015; RFDP grant 20133601120012; Research Grants from Jiangxi Education Department GJJ14094, GJJ13175; Research Grants from Jiangxi Science and Technology Department 20142BDH80005, 20142BAB215029, 20132BAB205024. Parts of this study were orally presented at annual meeting of Asia-Pacific Academy of Ophthalmology.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interests to declare.
Additional information
Qiuping Liu and Fengjun Zhang have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Liu, Q., Zhang, F., Zhang, X. et al. Fenofibrate ameliorates diabetic retinopathy by modulating Nrf2 signaling and NLRP3 inflammasome activation. Mol Cell Biochem 445, 105–115 (2018). https://doi.org/10.1007/s11010-017-3256-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-017-3256-x