Abstract
Neurodegenerative diseases (NDs), including chronic disease such as Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and multiple sclerosis, and acute diseases like traumatic brain injury and ischemic stroke are characterized by progressive degeneration, brain tissue damage and loss of neurons, accompanied by behavioral and cognitive dysfunctions. So far, there are no complete cures for NDs; thus, early and timely diagnoses are essential and beneficial to patients’ treatment. Magnetic resonance imaging (MRI) has become one of the advanced medical imaging techniques widely used in the clinical examination of NDs due to its non-invasive diagnostic value. In this review, research published in English in current decade from PubMed electronic database on the use of MRI to detect specific biomarkers of NDs was collected, summarized, and discussed, which provides valuable suggestions for the early diagnosis, prevention, and treatment of NDs in the clinic.




Similar content being viewed by others
Data Availability
Not applicable.
References
Zugaza JL (2021) Cell signaling in neurodegeneration. Int J Mol Sci 22(16):8978
Capizzi A, Woo J, Verduzco-Gutierrez M (2020) Traumatic brain injury: an overview of epidemiology, pathophysiology, and medical management. Med Clin North Am 104:213–238
Tao T, Liu M, Chen M, Luo Y, Wang C, Xu T, Jiang Y, Guo Y, et al (2020) Natural medicine in neuroprotection for ischemic stroke: challenges and prospective. Pharmacol Ther 216:107695
Group GBDNDC (2017) Global, regional, and national burden of neurological disorders during 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol 16:877–897
Erkkinen MG, Kim MO, Geschwind MD (2018) Clinical neurology and epidemiology of the major neurodegenerative diseases.Cold Spring Harb Perspect Biol 10(4):a033118
Guo J, Lee VJNM (2014) Cell-to-cell transmission of pathogenic proteins in neurodegenerative diseases. Nat Med 20:130–138
Castillo X, Castro-Obregón S, Gutiérrez-Becker B, Gutiérrez-Ospina G, Karalis N, Khalil A, Lopez-Noguerola J, Rodríguez L et al (2019) Re-thinking the etiological framework of neurodegeneration. Front Neurosci 13:728
Dugger BN, Dickson DW (2017) Pathology of neurodegenerative diseases. Cold Spring Harb Perspect Biol 9(7):a028035
Rankin KP, Gorno-Tempini ML, Allison SC, Stanley CM, Glenn S, Weiner MW, Miller BL (2006) Structural anatomy of empathy in neurodegenerative disease. Brain 129:2945–2956
Jordan LC, DeBaun MR, Donahue MJ (2021) Advances in neuroimaging to improve care in sickle cell disease. Lancet Neurol 20:398–408
Buzsáki G, Anastassiou CA, Koch C (2012) The origin of extracellular fields and currents–EEG, ECoG. LFP and spikes Nat Rev Neurosci 13:407–420
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, Klunk WE, Koroshetz WJ et al (2011) The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7:263–269
Chandra A, Dervenoulas G, Politis M (2019) Alzheimer’s Disease Neuroimaging I: Magnetic resonance imaging in Alzheimer’s disease and mild cognitive impairment. J Neurol 266:1293–1302
Hansson O (2021) Biomarkers for neurodegenerative diseases. Nat Med 27:954–963
Blennow K, Mattsson N, Scholl M, Hansson O, Zetterberg H (2015) Amyloid biomarkers in Alzheimer’s disease. Trends Pharmacol Sci 36:297–309
Koga S, Dickson DW (2018) Recent advances in neuropathology, biomarkers and therapeutic approach of multiple system atrophy. J Neurol Neurosurg Psychiatry 89:175–184
Gwinn K, David KK, Swanson-Fischer C, Albin R, Hillaire-Clarke CS, Sieber BA, Lungu C, Bowman FD et al (2017) Parkinson’s disease biomarkers: perspective from the NINDS Parkinson’s Disease Biomarkers Program. Biomark Med 11:451–473
Herukka SK, Simonsen AH, Andreasen N, Baldeiras I, Bjerke M, Blennow K, Engelborghs S, Frisoni GB et al (2017) Recommendations for cerebrospinal fluid Alzheimer’s disease biomarkers in the diagnostic evaluation of mild cognitive impairment. Alzheimers Dement 13:285–295
Aisen PS, Cummings J, Jack CR Jr, Morris JC, Sperling R, Frolich L, Jones RW, Dowsett SA et al (2017) On the path to 2025: understanding the Alzheimer’s disease continuum. Alzheimers Res Ther 9:60
Lesko LJ, Atkinson AJ Jr (2001) Use of biomarkers and surrogate endpoints in drug development and regulatory decision making: criteria, validation, strategies. Annu Rev Pharmacol Toxicol 41:347–366
Scheltens P, Blennow K, Breteler MM, de Strooper B, Frisoni GB, Salloway S, Van der Flier WM (2016) Alzheimer’s disease. Lancet 388:505–517
Simren J, Leuzy A, Karikari TK, Hye A, Benedet AL, Lantero-Rodriguez J, Mattsson-Carlgren N, Scholl M et al (2021) The diagnostic and prognostic capabilities of plasma biomarkers in Alzheimer’s disease. Alzheimers Dement 17(7):1145–1156
Cummings JL, Morstorf T, Zhong K (2014) Alzheimer’s disease drug-development pipeline: few candidates, frequent failures. Alzheimers Res Ther 6:37
Schirinzi T, Di Lorenzo F, Sancesario GM, Di Lazzaro G, Ponzo V, Pisani A, Mercuri NB, Koch G et al (2018) Amyloid-mediated cholinergic dysfunction in motor impairment related to Alzheimer’s disease. J Alzheimers Dis 64:525–532
Villa C, Lavitrano M, Salvatore E, Combi R (2020) Molecular and imaging biomarkers in Alzheimer’s disease: a focus on recent insights. J Pers Med 10(3):61
Nho K, Kueider-Paisley A, MahmoudianDehkordi S, Arnold M, Risacher SL, Louie G, Blach C, Baillie R et al (2019) Altered bile acid profile in mild cognitive impairment and Alzheimer’s disease: relationship to neuroimaging and CSF biomarkers. Alzheimers Dement 15:232–244
Luo J, Agboola F, Grant E, Masters CL, Albert MS, Johnson SC, McDade EM, Voglein J et al (2020) Sequence of Alzheimer disease biomarker changes in cognitively normal adults: a cross-sectional study. Neurology 95:e3104–e3116
Mouton-Liger F, Dumurgier J, Cognat E, Hourregue C, Zetterberg H, Vanderstichele H, Vanmechelen E, Bouaziz-Amar E et al (2020) CSF levels of the BACE1 substrate NRG1 correlate with cognition in Alzheimer’s disease. Alzheimers Res Ther 12:88
Zou K, Abdullah M, Michikawa M (2020) Current biomarkers for Alzheimer’s disease: from CSF to blood. J Pers Med 10(3):85
Milà-Alomà M, Brinkmalm A, Ashton NJ, Kvartsberg H, Shekari M, Operto G, Salvadó G, Falcon C et al (2021) CSF Synaptic biomarkers in the preclinical stage of Alzheimer disease and their association with MRI and PET: a cross-sectional study. Neurology 97(21):e2065–e2078
Zhao Q, Chen X, Zhou Y (2016) Quantitative multimodal multiparametric imaging in Alzheimer’s disease. Brain Inform 3:29–37
Ottoy J, Niemantsverdriet E, Verhaeghe J, De Roeck E, Struyfs H, Somers C, Wyffels L, Ceyssens S et al (2019) Association of short-term cognitive decline and MCI-to-AD dementia conversion with CSF, MRI, amyloid- and (18)F-FDG-PET imaging. Neuroimage Clin 22:101771
Abu-Rumeileh S, Mometto N, Bartoletti-Stella A, Polischi B, Oppi F, Poda R, Stanzani-Maserati M, Cortelli P, Liguori R, Capellari S, Parchi P (2018) Cerebrospinal fluid biomarkers in patients with frontotemporal dementia spectrum: a single-center study. J Alzheimers Dis 66:551–563
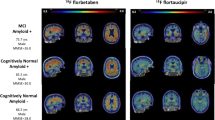
Okafor M, Nye JA, Shokouhi M, Shaw LM, Goldstein F, Hajjar I (2020) 18F-flortaucipir PET associations with cerebrospinal fluid, cognition, and neuroimaging in mild cognitive impairment due to Alzheimer’s disease. J Alzheimers Dis 74:589–601
Jang H, Park J, Woo S, Kim S, Kim HJ, Na DL, Lockhart SN, Kim Y et al (2019) Prediction of fast decline in amyloid positive mild cognitive impairment patients using multimodal biomarkers. Neuroimage Clin 24:101941
SanMartín CD, Paula-Lima AC, Hidalgo C, Núñez MT (2012) Sub-lethal levels of amyloid β-peptide oligomers decrease non-transferrin-bound iron uptake and do not potentiate iron toxicity in primary hippocampal neurons. Biometals 25:805–813
Telling ND, Everett J, Collingwood JF, Dobson J, van der Laan G, Gallagher JJ, Wang J, Hitchcock AP (2017) Iron biochemistry is correlated with amyloid plaque morphology in an established mouse model of Alzheimer’s disease. Cell Chem Biol 24:1205-1215.e1203
Acosta-Cabronero J, Williams GB, Cardenas-Blanco A, Arnold RJ, Lupson V, Nestor PJ (2013) In vivo quantitative susceptibility mapping (QSM) in Alzheimer’s disease. PLoS ONE 8:e81093
Du L, Zhao Z, Cui A, Zhu Y, Zhang L, Liu J, Shi S, Fu C et al (2018) Increased iron deposition on brain quantitative susceptibility mapping correlates with decreased cognitive function in Alzheimer’s disease. ACS Chem Neurosci 9:1849–1857
Li D, Liu Y, Zeng X, Xiong Z, Yao Y, Liang D, Qu H, Xiang H et al (2020) Quantitative study of the changes in cerebral blood flow and iron deposition during progression of Alzheimer’s disease. J Alzheimers Dis 78:439–452
Zhu WZ, Zhong WD, Wang W, Zhan CJ, Wang CY, Qi JP, Wang JZ, Lei T (2009) Quantitative MR phase-corrected imaging to investigate increased brain iron deposition of patients with Alzheimer disease. Radiology 253:497–504
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
Damulina A, Pirpamer L, Soellradl M, Sackl M, Tinauer C, Hofer E, Enzinger C, Gesierich B et al (2020) Cross-sectional and longitudinal assessment of brain iron level in Alzheimer disease using 3-T MRI. Radiology 296:619–626
Liu Y, Nguyen M, Robert A, Meunier B (2019) Metal ions in Alzheimer’s disease: a key role or not? Acc Chem Res 52:2026–2035
Benson G, Hildebrandt A, Lange C, Schwarz C, Kobe T, Sommer W, Floel A, Wirth M (2018) Functional connectivity in cognitive control networks mitigates the impact of white matter lesions in the elderly. Alzheimers Res Ther 10:109
Gaubert M, Lange C, Garnier-Crussard A, Kobe T, Bougacha S, Gonneaud J, de Flores R, Tomadesso C et al (2021) Topographic patterns of white matter hyperintensities are associated with multimodal neuroimaging biomarkers of Alzheimer’s disease. Alzheimers Res Ther 13:29
Lao PJ, Gutierrez J, Keator D, Rizvi B, Banerjee A, Igwe KC, Laing KK, Sathishkumar M et al (2020) Alzheimer-related cerebrovascular disease in Down syndrome. Ann Neurol 88:1165–1177
Walsh P, Sudre CH, Fiford CM, Ryan NS, Lashley T, Frost C, Barnes J (2020) CSF amyloid is a consistent predictor of white matter hyperintensities across the disease course from aging to Alzheimer’s disease. Neurobiol Aging 91:5–14
Ranjbar S, Velgos SN, Dueck AC, Geda YE, Mitchell JR (2019) Brain MR radiomics to differentiate cognitive disorders. J Neuropsychiatry Clin Neurosci 31:210–219
Freeman LR, Haley-Zitlin V, Stevens C, Granholm AC (2011) Diet-induced effects on neuronal and glial elements in the middle-aged rat hippocampus. Nutr Neurosci 14:32–44
Bulgart HR, Neczypor EW, Wold LE, Mackos AR (2020) Microbial involvement in Alzheimer disease development and progression. Mol Neurodegener 15:42
Lim YY, Baker JE, Bruns L Jr, Mills A, Fowler C, Fripp J, Rainey-Smith SR, Ames D et al (2020) Association of deficits in short-term learning and Aβ and hippocampal volume in cognitively normal adults. Neurology 95:e2577–e2585
Andriuta D, Moullart V, Schraen S, Devendeville A, Meyer ME, Godefroy O (2018) Inferior parietal cortex hypoperfusion is the most specific imaging marker for AD patients with positive CSF biomarker assays in a memory clinic in France. Alzheimer Dis Assoc Disord 32:89–93
Burggren AC, Mahmood Z, Harrison TM, Siddarth P, Miller KJ, Small GW, Merrill DA, Bookheimer SY (2017) Hippocampal thinning linked to longer TOMM40 poly-T variant lengths in the absence of the APOE ε4 variant. Alzheimers Dement 13:739–748
Sørensen L, Igel C, Liv Hansen N, Osler M, Lauritzen M, Rostrup E, Nielsen M (2016) Early detection of Alzheimer’s disease using MRI hippocampal texture. Hum Brain Mapp 37:1148–1161
Sørensen L, Igel C, Pai A, Balas I, Anker C, Lillholm M, Nielsen M (2017) Differential diagnosis of mild cognitive impairment and Alzheimer’s disease using structural MRI cortical thickness, hippocampal shape, hippocampal texture, and volumetry. Neuroimage Clin 13:470–482
Urbanowitsch N, Gorenc L, Herold CJ, Schroder J (2013) Autobiographical memory: a clinical perspective. Front Behav Neurosci 7:194
Thomann PA, Seidl U, Brinkmann J, Hirjak D, Traeger T, Wolf RC, Essig M, Schroder J (2012) Hippocampal morphology and autobiographic memory in mild cognitive impairment and Alzheimer’s disease. Curr Alzheimer Res 9:507–515
Hirjak D, Wolf RC, Remmele B, Seidl U, Thomann AK, Kubera KM, Schroder J, Maier-Hein KH et al (2017) Hippocampal formation alterations differently contribute to autobiographic memory deficits in mild cognitive impairment and Alzheimer’s disease. Hippocampus 27:702–715
Gertje EC, Pluta J, Das S, Mancuso L, Kliot D, Yushkevich P, Wolk D (2016) Clinical Application of automatic segmentation of medial temporal lobe subregions in prodromal and dementia-level Alzheimer’s disease. J Alzheimers Dis 54:1027–1037
Kim GW, Kim BC, Park KS, Jeong GW (2020) A pilot study of brain morphometry following donepezil treatment in mild cognitive impairment: volume changes of cortical/subcortical regions and hippocampal subfields. Sci Rep 10:10912
Buckley RF (2021) Recent advances in imaging of preclinical, sporadic, and autosomal dominant Alzheimer’s disease. Neurotherapeutics 18:709–727
Caunca MR, Del Brutto V, Gardener H, Shah N, Dequatre-Ponchelle N, Cheung YK, Elkind MS, Brown TR et al (2016) Cerebral microbleeds, vascular risk factors, and magnetic resonance imaging markers: the Northern Manhattan study. J Am Heart Assoc 5(9):e003477
Choi BR, Lee SR, Han JS, Woo SK, Kim KM, Choi DH, Kwon KJ, Han SH et al (2011) Synergistic memory impairment through the interaction of chronic cerebral hypoperfusion and amlyloid toxicity in a rat model. Stroke 42:2595–2604
Saridin FN, Hilal S, Villaraza SG, Reilhac A, Gyanwali B, Tanaka T, Stephenson MC, Ng SL et al (2020) Brain amyloid β, cerebral small vessel disease, and cognition: a memory clinic study. Neurology 95:e2845–e2853
Fazekas F, Kleinert R, Roob G, Kleinert G, Kapeller P, Schmidt R, Hartung HP (1999) Histopathologic analysis of foci of signal loss on gradient-echo T2*-weighted MR images in patients with spontaneous intracerebral hemorrhage: evidence of microangiopathy-related microbleeds. AJNR Am J Neuroradiol 20:637–642
Tikka S, Baumann M, Siitonen M, Pasanen P, Pöyhönen M, Myllykangas L, Viitanen M, Fukutake T et al (2014) CADASIL and CARASIL. Brain Pathol 24:525–544
Tsai HH, Pasi M, Tsai LK, Chen YF, Chen YW, Tang SC, Gurol ME, Yen RF et al (2020) Superficial cerebellar microbleeds and cerebral amyloid angiopathy: a magnetic resonance imaging/positron emission tomography study. Stroke 51:202–208
Attems J (2005) Sporadic cerebral amyloid angiopathy: pathology, clinical implications, and possible pathomechanisms. Acta Neuropathol 110:345–359
Enzinger C, Barkhof F, Ciccarelli O, Filippi M, Kappos L, Rocca MA, Ropele S, Rovira À et al (2015) Nonconventional MRI and microstructural cerebral changes in multiple sclerosis. Nat Rev Neurol 11:676–686
Nandigam RN, Viswanathan A, Delgado P, Skehan ME, Smith EE, Rosand J, Greenberg SM, Dickerson BC (2009) MR imaging detection of cerebral microbleeds: effect of susceptibility-weighted imaging, section thickness, and field strength. AJNR Am J Neuroradiol 30:338–343
Goos JD, van der Flier WM, Knol DL, Pouwels PJ, Scheltens P, Barkhof F, Wattjes MP (2011) Clinical relevance of improved microbleed detection by susceptibility-weighted magnetic resonance imaging. Stroke 42:1894–1900
Basselerie H, Bracoud L, Zeestraten E, Bouguen E, Kiyasova V, Pueyo M, Cognard C, Dumas H et al (2017) Incident cerebral microbleeds detected by susceptibility weight-imaging help to identify patients with mild cognitive impairment progressing to Alzheimer’s disease. J Alzheimers Dis 60:253–262
Masson C, Leys D, Buée L (2000) Cerebral amyloid angiopathies. Presse Med 29:1717–1722
Sparacia G, Agnello F, La Tona G, Iaia A, Midiri F, Sparacia B (2017) Assessment of cerebral microbleeds by susceptibility-weighted imaging in Alzheimer’s disease patients: a neuroimaging biomarker of the disease. Neuroradiol J 30:330–335
Zlokovic BV (2011) Neurovascular pathways to neurodegeneration in Alzheimer’s disease and other disorders. Nat Rev Neurosci 12:723–738
Csiszar A, Tarantini S, Fülöp GA, Kiss T, Valcarcel-Ares MN, Galvan V, Ungvari Z, Yabluchanskiy A (2017) Hypertension impairs neurovascular coupling and promotes microvascular injury: role in exacerbation of Alzheimer’s disease. Geroscience 39:359–372
Pettigrew C, Soldan A, Zhu Y, Cai Q, Wang MC, Moghekar A, Miller MI, Singh B et al (2020) Cognitive reserve and rate of change in Alzheimer’s and cerebrovascular disease biomarkers among cognitively normal individuals. Neurobiol Aging 88:33–41
Fabiani M, Gordon BA, Maclin EL, Pearson MA, Brumback-Peltz CR, Low KA, McAuley E, Sutton BP et al (2014) Neurovascular coupling in normal aging: a combined optical. ERP and fMRI study Neuroimage 85(Pt 1):592–607
Van Skike CE, Lin AL, Roberts Burbank R, Halloran JJ, Hernandez SF, Cuvillier J, Soto VY, Hussong SA et al (2020) mTOR drives cerebrovascular, synaptic, and cognitive dysfunction in normative aging. Aging Cell 19:e13057
Hamilton R, Patel S, Lee EB, Jackson EM, Lopinto J, Arnold SE, Clark CM, Basil A et al (2010) Lack of shunt response in suspected idiopathic normal pressure hydrocephalus with Alzheimer disease pathology. Ann Neurol 68:535–540
Reeves BC, Karimy JK, Kundishora AJ, Mestre H, Cerci HM, Matouk C, Alper SL, Lundgaard I et al (2020) Glymphatic system impairment in Alzheimer’s disease and idiopathic normal pressure hydrocephalus. Trends Mol Med 26:285–295
Chao LL, Buckley ST, Kornak J, Schuff N, Madison C, Yaffe K, Miller BL, Kramer JH et al (2010) ASL perfusion MRI predicts cognitive decline and conversion from MCI to dementia. Alzheimer Dis Assoc Disord 24:19–27
Azuma S, Kazui H, Kanemoto H, Suzuki Y, Sato S, Suehiro T, Matsumoto T, Yoshiyama K et al (2019) Cerebral blood flow and Alzheimer’s disease-related biomarkers in cerebrospinal fluid in idiopathic normal pressure hydrocephalus. Psychogeriatrics 19:527–538
Stern Y, Arenaza-Urquijo EM, Bartrés-Faz D, Belleville S, Cantilon M, Chetelat G, Ewers M, Franzmeier N et al (2020) Whitepaper: defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimers Dement 16:1305–1311
Debette S, Seshadri S, Beiser A, Au R, Himali JJ, Palumbo C, Wolf PA, DeCarli C (2011) Midlife vascular risk factor exposure accelerates structural brain aging and cognitive decline. Neurology 77:461–468
Ritchie K, Ritchie CW, Yaffe K, Skoog I, Scarmeas N (2015) Is late-onset Alzheimer’s disease really a disease of midlife? Alzheimers Dement (N Y) 1:122–130
Neth BJ, Graff-Radford J, Mielke MM, Przybelski SA, Lesnick TG, Schwarz CG, Reid RI, Senjem ML et al (2019) Relationship Between risk factors and brain reserve in late middle age: implications for cognitive aging. Front Aging Neurosci 11:355
Stern Y, Barulli D (2019) Cognitive reserve. Handb Clin Neurol 167:181–190
Soloveva MV, Jamadar SD, Poudel G, Georgiou-Karistianis N (2018) A critical review of brain and cognitive reserve in Huntington’s disease. Neurosci Biobehav Rev 88:155–169
McKenzie C, Bucks RS, Weinborn M, Bourgeat P, Salvado O, Gavett BE (2020) Cognitive reserve predicts future executive function decline in older adults with Alzheimer’s disease pathology but not age-associated pathology. Neurobiol Aging 88:119–127
Frenzel S, Wittfeld K, Habes M, Klinger-Konig J, Bulow R, Volzke H, Grabe HJ (2019) A biomarker for Alzheimer’s disease based on patterns of regional brain atrophy. Front Psychiatry 10:953
Tolosa E, Garrido A, Scholz SW, Poewe W (2021) Challenges in the diagnosis of Parkinson’s disease. Lancet Neurol 20:385–397
Gao L, Wang W, Wang X, Yang F, Xie L, Shen J, Brimble MA, Xiao Q et al (2021) Fluorescent probes for bioimaging of potential biomarkers in Parkinson’s disease. Chem Soc Rev 50:1219–1250
Shahnawaz M, Mukherjee A, Pritzkow S, Mendez N, Rabadia P, Liu X, Hu B, Schmeichel A et al (2020) Discriminating α-synuclein strains in Parkinson’s disease and multiple system atrophy. Nature 578:273–277
Parnetti L, Cicognola C, Eusebi P, Chiasserini D (2016) Value of cerebrospinal fluid α-synuclein species as biomarker in Parkinson’s diagnosis and prognosis. Biomark Med 10:35–49
Bindas AJ, Kulkarni S, Koppes RA, Koppes AN (2021) Parkinson’s disease and the gut: models of an emerging relationship. Acta Biomater 132:325–344
Balestrino R, Schapira AHV (2020) Parkinson disease. Eur J Neurol 27:27–42
Schapira AHV, Chaudhuri KR, Jenner P (2017) Non-motor features of Parkinson disease. Nat Rev Neurosci 18:435–450
Compta Y, Parkkinen L, Kempster P, Selikhova M, Lashley T, Holton JL, Lees AJ, Revesz T (2014) The significance of α-synuclein, amyloid-β and tau pathologies in Parkinson’s disease progression and related dementia. Neurodegener Dis 13:154–156
Chen CH, Lee BC, Lin CH (2020) Integrated Plasma and neuroimaging biomarkers associated with motor and cognition severity in Parkinson’s disease. J Parkinsons Dis 10:77–88
Halliday G, Lees A, Stern M (2011) Milestones in Parkinson’s disease–clinical and pathologic features. Mov Disord 26:1015–1021
Campbell MC, Jackson JJ, Koller JM, Snyder AZ, Kotzbauer PT, Perlmutter JS (2020) Proteinopathy and longitudinal changes in functional connectivity networks in Parkinson disease. Neurology 94:e718–e728
Korat Š, Bidesi NSR, Bonanno F, Di Nanni A, Hoàng ANN, Herfert K, Maurer A, Battisti UM et al (2021) Alpha-synuclein PET tracer development-an overview about current efforts. Pharmaceuticals (Basel) 14(9):847
Cheng HC, Ulane CM, Burke RE (2010) Clinical progression in Parkinson disease and the neurobiology of axons. Ann Neurol 67:715–725
Blair JC, Barrett MJ, Patrie J, Flanigan JL, Sperling SA, Elias WJ, Druzgal TJ (2019) Brain MRI reveals ascending atrophy in Parkinson’s disease across severity. Front Neurol 10:1329
Guo JL, Lee VMY (2014) Cell-to-cell transmission of pathogenic proteins in neurodegenerative diseases. Nat Med 20:130–138
Vasconcellos LF, Pereira JS, Adachi M, Greca D, Cruz M, Malak AL, Charchat-Fichman H (2018) Volumetric brain analysis as a predictor of a worse cognitive outcome in Parkinson’s disease. J Psychiatr Res 102:254–260
Wang Q, Liu Y, Zhou J (2015) Neuroinflammation in Parkinson’s disease and its potential as therapeutic target. Transl Neurodegener 4:19
Truelove-Hill M, Erus G, Bashyam V, Varol E, Sako C, Gur RC, Gur RE, Koutsouleris N et al (2020) A multidimensional neural maturation index reveals reproducible developmental patterns in children and adolescents. J Neurosci 40:1265–1275
Racine AM, Brickhouse M, Wolk DA, Dickerson BC (2018) Alzheimer’s Disease Neuroimaging I: The personalized Alzheimer’s disease cortical thickness index predicts likely pathology and clinical progression in mild cognitive impairment. Alzheimers Dement (Amst) 10:301–310
Kang SW, Jeon S, Yoo HS, Chung SJ, Lee PH, Sohn YH, Yun M, Evans AC et al (2019) Effects of Lewy body disease and Alzheimer disease on brain atrophy and cognitive dysfunction. Neurology 92:e2015–e2026
Balazova Z, Novakova M, Minsterova A, Rektorova I (2019) Structural and functional magnetic resonance imaging of dementia with lewy bodies. Int Rev Neurobiol 144:95–141
Ye R, Touroutoglou A, Brickhouse M, Katz S, Growdon JH, Johnson KA, Dickerson BC, Gomperts SN (2020) Topography of cortical thinning in the Lewy body diseases. Neuroimage Clin 26:102196
Licker V, Turck N, Kovari E, Burkhardt K, Cote M, Surini-Demiri M, Lobrinus JA, Sanchez JC et al (2014) Proteomic analysis of human substantia nigra identifies novel candidates involved in Parkinson’s disease pathogenesis. Proteomics 14:784–794
Kumari S, Goyal V, Kumaran SS, Dwivedi SN, Srivastava A, Jagannathan NR (2020) Quantitative metabolomics of saliva using proton NMR spectroscopy in patients with Parkinson’s disease and healthy controls. Neurol Sci 41:1201–1210
(2021) Corrigendum to: Spatiotemporal changes in substantia nigra neuromelanin content in Parkinson’s disease. Brain 144:e24
Sung YH, Noh Y, Kim EY (2021) Early-stage Parkinson’s disease: abnormal nigrosome 1 and 2 revealed by a voxelwise analysis of neuromelanin-sensitive MRI. Hum Brain Mapp 42:2823–2832
Pavese N (2019) Is neuromelanin the imaging biomarker for the early diagnosis of Parkinson’s disease that we were looking for? Parkinsonism Relat Disord 58:1–2
Carballo-Carbajal I, Laguna A, Romero-Giménez J, Cuadros T, Bové J, Martinez-Vicente M, Parent A, Gonzalez-Sepulveda M et al (2019) Brain tyrosinase overexpression implicates age-dependent neuromelanin production in Parkinson’s disease pathogenesis. Nat Commun 10:973
Oxtoby NP, Leyland LA, Aksman LM, Thomas GEC, Bunting EL, Wijeratne PA, Young AL, Zarkali A et al (2021) Sequence of clinical and neurodegeneration events in Parkinson’s disease progression. Brain 144:975–988
Pavese N, Tai YF (2018) Nigrosome imaging and neuromelanin sensitive MRI in diagnostic evaluation of Parkinsonism. Mov Disord Clin Pract 5:131–140
Singh S, Nimse SB, Mathew DE, Dhimmar A, Sahastrabudhe H, Gajjar A, Ghadge VA, Kumar P et al (2021) Microbial melanin: recent advances in biosynthesis, extraction, characterization, and applications. Biotechnol Adv 53:107773
Bose A, Petsko GA, Eliezer D (2018) Parkinson’s disease and melanoma: co-occurrence and mechanisms. J Parkinsons Dis 8:385–398
Seo H, Clark HA (2020) Gadolinium-based MRI contrast agent for the detection of tyrosinase. Analyst 145:1169–1173
Hain EG, Sparenberg M, Rasińska J, Klein C, Akyüz L, Steiner B (2018) Indomethacin promotes survival of new neurons in the adult murine hippocampus accompanied by anti-inflammatory effects following MPTP-induced dopamine depletion. J Neuroinflammation 15:162
Piao YS, Lian TH, Hu Y, Zuo LJ, Guo P, Yu SY, Liu L, Jin Z et al (2017) Restless legs syndrome in Parkinson disease: clinical characteristics, abnormal iron metabolism and altered neurotransmitters. Sci Rep 7:10547
Kumar H, Lim HW, More SV, Kim BW, Koppula S, Kim IS, Choi DK (2012) The role of free radicals in the aging brain and Parkinson’s disease: convergence and parallelism. Int J Mol Sci 13:10478–10504
Lima IV, Bastos LF, Limborço-Filho M, Fiebich BL, de Oliveira AC (2012) Role of prostaglandins in neuroinflammatory and neurodegenerative diseases. Mediators Inflamm 2012:946813
Langley J, Huddleston DE, Sedlacik J, Boelmans K, Hu XP (2017) Parkinson’s disease-related increase of T2*-weighted hypointensity in substantia nigra pars compacta. Mov Disord 32:441–449
Barber TR, Griffanti L, Bradley KM, McGowan DR, Lo C, Mackay CE, Hu MT, Klein JC (2020) Nigrosome 1 imaging in REM sleep behavior disorder and its association with dopaminergic decline. Ann Clin Transl Neurol 7:26–35
Sheng H, Zhao B, Ge Y (2019) Blood perfusion and cellular microstructural changes associated with iron deposition in multiple sclerosis lesions. Front Neurol 10:747
Bergsland N, Zivadinov R, Schweser F, Hagemeier J, Lichter D, Guttuso T Jr (2019) Ventral posterior substantia nigra iron increases over 3 years in Parkinson’s disease. Mov Disord 34:1006–1013
Lotfipour AK, Wharton S, Schwarz ST, Gontu V, Schäfer A, Peters AM, Bowtell RW, Auer DP et al (2012) High resolution magnetic susceptibility mapping of the substantia nigra in Parkinson’s disease. J Magn Reson Imaging 35:48–55
An H, Zeng X, Niu T, Li G, Yang J, Zheng L, Zhou W, Liu H et al (2018) Quantifying iron deposition within the substantia nigra of Parkinson’s disease by quantitative susceptibility mapping. J Neurol Sci 386:46–52
Alberico SL, Cassell MD, Narayanan NS (2015) The vulnerable ventral tegmental area in Parkinson’s disease. Basal Ganglia 5:51–55
Porter E, Roussakis AA, Lao-Kaim NP, Piccini P (2020) Multimodal dopamine transporter (DAT) imaging and magnetic resonance imaging (MRI) to characterise early Parkinson’s disease. Parkinsonism Relat Disord 79:26–33
de la Fuente-Fernández R (2012) Role of DaTSCAN and clinical diagnosis in Parkinson disease. Neurology 78:696–701
Yoo SW, Oh YS, Hwang EJ, Ryu DW, Lee KS, Lyoo CH, Kim JS (2019) “Depressed” caudate and ventral striatum dopamine transporter availability in de novo Depressed Parkinson’s disease. Neurobiol Dis 132:104563
Oh YS, Kim JS, Hwang EJ, Lyoo CH (2018) Striatal dopamine uptake and olfactory dysfunction in patients with early Parkinson’s disease. Parkinsonism Relat Disord 56:47–51
Ross CA, Aylward EH, Wild EJ, Langbehn DR, Long JD, Warner JH, Scahill RI, Leavitt BR et al (2014) Huntington disease: natural history, biomarkers and prospects for therapeutics. Nat Rev Neurol 10:204–216
Doan NT, van den Bogaard SJ, Dumas EM, Webb AG, van Buchem MA, Roos RA, van der Grond J, Reiber JH et al (2014) Texture analysis of ultrahigh field T2*-weighted MR images of the brain: application to Huntington’s disease. J Magn Reson Imaging 39:633–640
Xu W, Ocak U, Gao L, Tu S, Lenahan CJ, Zhang J, Shao A (2021) Selective autophagy as a therapeutic target for neurological diseases. Cell Mol Life Sci 78:1369–1392
Byrne LM, Rodrigues FB, Johnson EB, Wijeratne PA, De Vita E, Alexander DC, Palermo G, Czech C et al (2018) Evaluation of mutant huntingtin and neurofilament proteins as potential markers in Huntington’s disease. Sci Transl Med 10(458):eaat7108
Dixon SJ, Stockwell BR (2014) The role of iron and reactive oxygen species in cell death. Nat Chem Biol 10:9–17
Chen L, Hua J, Ross CA, Cai S, van Zijl PCM, Li X (2019) Altered brain iron content and deposition rate in Huntington’s disease as indicated by quantitative susceptibility MRI. J Neurosci Res 97:467–479
Boada FE, LaVerde G, Jungreis C, Nemoto E, Tanase C, Hancu I (2005) Loss of cell ion homeostasis and cell viability in the brain: what sodium MRI can tell us. Curr Top Dev Biol 70:77–101
Reetz K, Romanzetti S, Dogan I, Saß C, Werner CJ, Schiefer J, Schulz JB, Shah NJ (2012) Increased brain tissue sodium concentration in Huntington’s disease - a sodium imaging study at 4 T. Neuroimage 63:517–524
Odish OFF, Reijntjes R, van den Bogaard SJA, Roos RAC, Leemans A (2018) Progressive microstructural changes of the occipital cortex in Huntington’s disease. Brain Imaging Behav 12:1786–1794
Nanetti L, Contarino VE, Castaldo A, Sarro L, Bachoud-Levi AC, Giavazzi M, Frittoli S, Ciammola A et al (2018) Cortical thickness, stance control, and arithmetic skill: an exploratory study in premanifest Huntington disease. Parkinsonism Relat Disord 51:17–23
Wouters E, Grajchen E, Jorissen W, Dierckx T, Wetzels S, Loix M, Tulleners MP, Staels B et al (2020) Altered PPARγ expression promotes myelin-induced foam cell formation in macrophages in multiple sclerosis. Int J Mol Sci 21(23):9329
Martinez B, Peplow PV (2020) MicroRNAs in blood and cerebrospinal fluid as diagnostic biomarkers of multiple sclerosis and to monitor disease progression. Neural Regen Res 15:606–619
Paul A, Comabella M, Gandhi R (2019) Biomarkers in multiple sclerosis. Cold Spring Harb Perspect Med 9(3):a029058
Elliott C, Belachew S, Wolinsky JS, Hauser SL, Kappos L, Barkhof F, Bernasconi C, Fecker J et al (2019) Chronic white matter lesion activity predicts clinical progression in primary progressive multiple sclerosis. Brain 142:2787–2799
Lichvarova L, Blum W, Schwaller B, Szabolcsi V (2019) Parvalbumin expression in oligodendrocyte-like CG4 cells causes a reduction in mitochondrial volume, attenuation in reactive oxygen species production and a decrease in cell processes’ length and branching. Sci Rep 9:10603
O’Muircheartaigh J, Vavasour I, Ljungberg E, Li DKB, Rauscher A, Levesque V, Garren H, Clayton D et al (2019) Quantitative neuroimaging measures of myelin in the healthy brain and in multiple sclerosis. Hum Brain Mapp 40:2104–2116
Gozt A, Licari M, Halstrom A, Milbourn H, Lydiard S, Black A, Arendts G, Macdonald S et al (2020) Towards the development of an integrative, evidence-based suite of indicators for the prediction of outcome following mild traumatic brain injury: results from a pilot study. Brain Sci 10(1):23
Huang L, Obenaus A, Hamer M, Zhang JH (2016) Neuroprotective effect of hyperbaric oxygen therapy in a juvenile rat model of repetitive mild traumatic brain injury. Med Gas Res 6:187–193
Eve DJ, Steele MR, Sanberg PR, Borlongan CV (2016) Hyperbaric oxygen therapy as a potential treatment for post-traumatic stress disorder associated with traumatic brain injury. Neuropsychiatr Dis Treat 12:2689–2705
Rohling ML, Binder LM, Demakis GJ, Larrabee GJ, Ploetz DM, Langhinrichsen-Rohling J (2011) A meta-analysis of neuropsychological outcome after mild traumatic brain injury: re-analyses and reconsiderations of Binder, et al 2011 1997), Frencham et al. (2005), and Pertab et al. (2009). Clin Neuropsychol 25:608–623
Ruslin M, Wolff J, Yusuf HY, Arifin MZ, Boffano P, Forouzanfar T (2019) Use of neuron-specific enolase to predict mild brain injury in motorcycle crash patients with maxillofacial fractures: a pilot study. Chin J Traumatol 22:47–50
Katz DI, Cohen SI, Alexander MP (2015) Mild traumatic brain injury. Handb Clin Neurol 127:131–156
Kulbe JR, Geddes JW (2016) Current status of fluid biomarkers in mild traumatic brain injury. Exp Neurol 275(Pt 3):334–352
Chu Z, Wilde EA, Hunter JV, McCauley SR, Bigler ED, Troyanskaya M, Yallampalli R, Chia JM et al (2010) Voxel-based analysis of diffusion tensor imaging in mild traumatic brain injury in adolescents. AJNR Am J Neuroradiol 31:340–346
Pandit AS, Expert P, Lambiotte R, Bonnelle V, Leech R, Turkheimer FE, Sharp DJ (2013) Traumatic brain injury impairs small-world topology. Neurology 80:1826–1833
Rubinov M, Sporns O (2010) Complex network measures of brain connectivity: uses and interpretations. Neuroimage 52:1059–1069
Aoki Y, Inokuchi R, Gunshin M, Yahagi N, Suwa H (2012) Diffusion tensor imaging studies of mild traumatic brain injury: a meta-analysis. J Neurol Neurosurg Psychiatry 83:870–876
Bayly PV, Clayton EH, Genin GM (2012) Quantitative imaging methods for the development and validation of brain biomechanics models. Annu Rev Biomed Eng 14:369–396
Hylin MJ, Orsi SA, Zhao J, Bockhorst K, Perez A, Moore AN, Dash PK (2013) Behavioral and histopathological alterations resulting from mild fluid percussion injury. J Neurotrauma 30:702–715
Koerte IK, Hufschmidt J, Muehlmann M, Tripodis Y, Stamm JM, Pasternak O, Giwerc MY, Coleman MJ et al (2016) Cavum Septi pellucidi in symptomatic former professional football players. J Neurotrauma 33:346–353
Gardner RC, Hess CP, Brus-Ramer M, Possin KL, Cohn-Sheehy BI, Kramer JH, Berger MS, Yaffe K et al (2016) Cavum septum pellucidum in retired american pro-football players. J Neurotrauma 33:157–161
Winter TC, Kennedy AM, Byrne J, Woodward PJ (2010) The cavum septi pellucidi: why is it important? J Ultrasound Med 29:427–444
Zeiler FA, Ercole A, Czosnyka M, Smielewski P, Hawryluk G, Hutchinson PJA, Menon DK, Aries M (2020) Continuous cerebrovascular reactivity monitoring in moderate/severe traumatic brain injury: a narrative review of advances in neurocritical care. Br J Anaesth S0007-0912(19)30966-3
Garrett DD, Lindenberger U, Hoge RD, Gauthier CJ (2017) Age differences in brain signal variability are robust to multiple vascular controls. Sci Rep 7:10149
Young AMH, Adams H, Donnelly J, Guilfoyle MR, Fernandes H, Garnett MR, Czosnyka M, Smielewski P et al (2017) Glycemia is related to impaired cerebrovascular autoregulation after severe pediatric traumatic brain injury: a retrospective observational study. Front Pediatr 5:205
Bazzigaluppi P, Adams C, Koletar MM, Dorr A, Pikula A, Carlen PL, Stefanovic B (2018) Oophorectomy reduces estradiol levels and long-term spontaneous neurovascular recovery in a female rat model of focal ischemic stroke. Front Mol Neurosci 11:338
Forster BB, MacKay AL, Whittall KP, Kiehl KA, Smith AM, Hare RD, Liddle PF (1998) Functional magnetic resonance imaging: the basics of blood-oxygen-level dependent (BOLD) imaging. Can Assoc Radiol J 49:320–329
Clarençon F, Bardinet É, Martinerie J, Pelbarg V, Menjot de Champfleur N, Gupta R, Tollard E, Soto-Ares G et al (2017) Lesions in deep gray nuclei after severe traumatic brain injury predict neurologic outcome. PLoS One 12:e0186641
Haber M, Amyot F, Lynch CE, Sandsmark DK, Kenney K, Werner JK, Moore C, Flesher K et al (2021) Imaging biomarkers of vascular and axonal injury are spatially distinct in chronic traumatic brain injury. J Cereb Blood Flow Metab 41:1924–1938
Zeiler FA, Donnelly J, Calviello L, Menon DK, Smielewski P, Czosnyka M (2017) Pressure autoregulation measurement techniques in adult traumatic brain injury, part i: a scoping review of intermittent/semi-intermittent methods. J Neurotrauma 34:3207–3223
Leoni RF, Oliveira IA, Pontes-Neto OM, Santos AC, Leite JP (2017) Cerebral blood flow and vasoreactivity in aging: an arterial spin labeling study. Braz J Med Biol Res 50:e5670
Hannan MA, Rahman MA, Sohag AAM, Uddin MJ, Dash R, Sikder MH, Rahman MS, Timalsina B et al (2021) Black cumin (Nigella sativa L.): a comprehensive review on phytochemistry, health benefits, molecular pharmacology, and safety. Nutrients 13(6):1784
Wolska M, Jarosz-Popek J, Junger E, Wicik Z, Porshoor T, Sharif L, Czajka P, Postula M et al (2021) Long non-coding RNAs as promising therapeutic approach in ischemic stroke: a comprehensive review. Mol Neurobiol 58:1664–1682
Zhang TY, Cai MT, Zheng Y, Lai QL, Shen CH, Qiao S, Zhang YX (2021) Anti-alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor encephalitis: a review. Front Immunol 12:652820
Zhang W, Tian T, Gong SX, Huang WQ, Zhou QY, Wang AP, Tian Y (2021) Microglia-associated neuroinflammation is a potential therapeutic target for ischemic stroke. Neural Regen Res 16:6–11
Koch S, Mueller S, Foddis M, Bienert T, von Elverfeldt D, Knab F, Farr TD, Bernard R, et al (2019) Atlas registration for edema-corrected MRI lesion volume in mouse stroke models. J Cereb Blood Flow Metab 39:313–323
Gerriets T, Stolz E, Walberer M, Müller C, Kluge A, Bachmann A, Fisher M, Kaps M, et al (2004) Noninvasive quantification of brain edema and the space-occupying effect in rat stroke models using magnetic resonance imaging. Stroke 35:566–571
Nakajo Y, Zhao Q, Enmi JI, Iida H, Takahashi JC, Kataoka H, Yamato K, Yanamoto H (2019) Early detection of cerebral infarction after focal ischemia using a new MRI indicator. Mol Neurobiol 56:658–670
Rey J, Sarntinoranont M (2018) Pulsatile flow drivers in brain parenchyma and perivascular spaces: a resistance network model study. Fluids Barriers CNS 15:20
Liang Y, Chan YL, Deng M, Chen YK, Mok V, Wang F, Ungvari GS, Chu CW, et al (2018) Enlarged perivascular spaces in the centrum semiovale are associated with poststroke depression: a 3-month prospective study. J Affect Disord 228:166–172
Brown R, Benveniste H, Black SE, Charpak S, Dichgans M, Joutel A, Nedergaard M, Smith KJ, et al (2018) Understanding the role of the perivascular space in cerebral small vessel disease. Cardiovasc Res 114:1462–1473
Nation DA, Sweeney MD, Montagne A, Sagare AP, D’Orazio LM, Pachicano M, Sepehrband F, Nelson AR, et al (2019) Blood-brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat Med 25:270–276
Sundaram S, Hughes RL, Peterson E, Müller-Oehring EM, Brontë-Stewart HM, Poston KL, Faerman A, Bhowmick C, et al (2019) Establishing a framework for neuropathological correlates and glymphatic system functioning in Parkinson’s disease. Neurosci Biobehav Rev 103:305–315
Yu N, Sinclair B, Posada LMG, Chen Z, Di Q, Lin X, Kolbe S, Hlauschek G, Kwan P et al (2022) Asymmetric distribution of enlarged perivascular spaces in centrum semiovale may be associated with epilepsy after acute ischemic stroke. CNS Neurosci Ther 28(3):343–353
Shahid A, Patel VB, Morton JS, Stenson TH, Davidge ST, Oudit GY, McMurtry MS (2019) Low altitude simulation without hypoxia improves left ventricular function after myocardial infarction by reducing ventricular afterload. PLoS ONE 14:e0215814
Mandeville ET, Ayata C, Zheng Y, Mandeville JB (2017) Translational MR neuroimaging of stroke and recovery. Transl Stroke Res 8:22–32
Jin S, Han S, Stoyanova R, Ackerstaff E, Cho H (2020) Pattern recognition analysis of dynamic susceptibility contrast (DSC)-MRI curves automatically segments tissue areas with intact blood-brain barrier in a rat stroke model: a feasibility and comparison study. J Magn Reson Imaging 51:1369–1381
Huang WY, Wu G, Li JJ, Geng DY, Tan WL, Yu XR (2015) Prediction of reperfusion-associated hemorrhagic transformation using dynamic contrast-enhanced imaging in a rat stroke model. J Comput Assist Tomogr 39:787–793
To MNN, Kim HJ, Roh HG, Cho YS, Kwak JT (2020) Deep regression neural networks for collateral imaging from dynamic susceptibility contrast-enhanced magnetic resonance perfusion in acute ischemic stroke. Int J Comput Assist Radiol Surg 15:151–162
Zhu G, Federau C, Wintermark M, Chen H, Marcellus DG, Martin BW, Heit JJ (2020) Comparison of MRI IVIM and MR perfusion imaging in acute ischemic stroke due to large vessel occlusion. Int J Stroke 15:332–342
Aracki-Trenkic A, Law-Ye B, Radovanovic Z, Stojanov D, Dormont D, Pyatigorskaya N (2020) ASL perfusion in acute ischemic stroke: the value of CBF in outcome prediction. Clin Neurol Neurosurg 194:105908
Zhang SX, Yao YH, Zhang S, Zhu WJ, Tang XY, Qin YY, Zhao LY, Liu CX, Zhu WZ (2015) Comparative study of DSC-PWI and 3D-ASL in ischemic stroke patients. J Huazhong Univ Sci Technolog Med Sci 35:923–927
Tiedt S, Duering M, Barro C, Kaya AG, Boeck J, Bode FJ, Klein M, Dorn F, et al (2018) Serum neurofilament light: a biomarker of neuroaxonal injury after ischemic stroke. Neurology 91:e1338–e1347
Makris K, Haliassos A, Chondrogianni M, Tsivgoulis G (2018) Blood biomarkers in ischemic stroke: potential role and challenges in clinical practice and research. Crit Rev Clin Lab Sci 55:294–328
Al-Chalabi A, Hardiman O, Kiernan MC, Chiò A, Rix-Brooks B, van den Berg LH (2016) Amyotrophic lateral sclerosis: moving towards a new classification system. Lancet Neurol 15:1182–1194
Alexander AL, Hurley SA, Samsonov AA, Adluru N, Hosseinbor AP, Mossahebi P (2011) Tromp do PM, Zakszewski E, Field AS: Characterization of cerebral white matter properties using quantitative magnetic resonance imaging stains. Brain Connect 1:423–446
Baek SH, Park J, Kim YH, Seok HY, Oh KW, Kim HJ, Kwon YJ, Sim Y, et al (2020) Usefulness of diffusion tensor imaging findings as biomarkers for amyotrophic lateral sclerosis. Sci Rep 10:5199
Saccà F, Puorro G, Brunetti A, Capasso G, Cervo A, Cocozza S, de Leva M, Marsili A, et al (2015) A randomized controlled pilot trial of lithium in spinocerebellar ataxia type 2. J Neurol 262:149–153
Mascalchi M, Vella A (2020) Neuroimaging biomarkers in SCA2 gene carriers. Int J Mol Sci 21(3):1020
Frisoni GB, Fox NC, Jack CR Jr, Scheltens P, Thompson PM (2010) The clinical use of structural MRI in Alzheimer disease. Nat Rev Neurol 6:67–77
Lombardi G, Crescioli G, Cavedo E, Lucenteforte E, Casazza G, Bellatorre AG, Lista C, Costantino G, et al (2020) Structural magnetic resonance imaging for the early diagnosis of dementia due to Alzheimer’s disease in people with mild cognitive impairment. Cochrane Database Syst Rev 3:Cd009628
Biomarkers and surrogate endpoints (2001) preferred definitions and conceptual framework. Clin Pharmacol Ther 69:89–95
Ehrenberg AJ, Khatun A, Coomans E, Betts MJ, Capraro F, Thijssen EH, Senkevich K, Bharucha T, et al (2020) Relevance of biomarkers across different neurodegenerative diseases. Alzheimers Res Ther 12:56
Acknowledgements
The authors would like to express their gratitude to EditSprings (https://www.editsprings.cn/) for the expert linguistic services provided.
Funding
This work was supported by Grants from the National Natural Science Foundation of China (Grant No. 81903829), the Joint project of Luzhou Municipal People’s Government and Southwest Medical University, China (Grant No. 2018LZXNYD-ZK42).
Author information
Authors and Affiliations
Contributions
K-RL and LY conceived the idea. C-LY, YT, and J-MW completed the work of literature retrieval. K-RL, LY, and X-PH provided written input on scientific content of drafts. G-QH, A-GW, and LY revised the manuscript and approved the final draft for submission. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
The authors mentioned have participated in this article.
Consent for Publication
The authors approved the publication of article.
Conflicts of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. We summarized various types of biomarkers in NDs with MRI detection including pathogenic proteins, anatomical structures, body materials and pathological conditions.
2. Most biomarkers we listed are specific to different NDs which can be manifested by magnetic resonance.
3. We discussed the possibility that other biomarkers such as Aβ and p-Tau which could also be used for non-invasive examination by magnetic resonance in the future.
4. These biomarkers reveal the profound pathological features of NDs, which provide evidence for preclinical and clinical studies.
Rights and permissions
About this article
Cite this article
Li, KR., Wu, AG., Tang, Y. et al. The Key Role of Magnetic Resonance Imaging in the Detection of Neurodegenerative Diseases-Associated Biomarkers: A Review. Mol Neurobiol 59, 5935–5954 (2022). https://doi.org/10.1007/s12035-022-02944-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-02944-x