Abstract
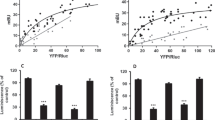
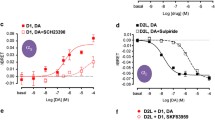
The two most common polymorphisms of the human DRD4 gene encode a dopamine D4 receptor (D4R) with four or seven repeats of a proline-rich sequence of 16 amino acids (D4.4R or D4.7R). Although the seven-repeat polymorphism has been repeatedly associated with attention-deficit hyperactivity disorder and substance use disorders, the differential functional properties between D4.4R and D4.7R remained enigmatic until recent electrophysiological and optogenetic-microdialysis experiments indicated a gain of function of D4.7R. Since no clear differences in the biochemical properties of individual D4.4R and D4.7R have been reported, it was previously suggested that those differences emerge upon heteromerization with dopamine D2 receptor (D2R), which co-localizes with D4R in the brain. However, contrary to a gain of function, experiments in mammalian transfected cells suggested that heteromerization with D2R results in lower MAPK signaling by D4.7R as compared to D4.4R. In the present study, we readdressed the question of functional differences of D4.4R and D4.7R forming homomers or heteromers with the short isoform of D2R (D2SR), using a functional bioluminescence resonance energy transfer (BRET) assay that allows the measurement of ligand-induced changes in the interaction between G protein-coupled receptors (GPCRs) forming homomers or heteromers with their cognate G protein. Significant functional and pharmacological differences between D4.4R and D4.7R were only evident upon heteromerization with the short isoform of D2R (D2SR). The most dramatic finding was a significant increase and decrease in the constitutive activity of D2S upon heteromerization with D4.7R and D4.4R, respectively, providing the first clear mechanism for a functional difference between both products of polymorphic variants and for a gain of function of the D4.7R.






Similar content being viewed by others
References
LaHoste GJ, Swanson JM, Wigal SB, Glabe C, Wigal T, King N, Kennedy JL (1996) Dopamine D4 receptor gene polymorphism is associated with attention deficit hyperactivity disorder. Mol Psychiatry 1:121–124
Chang FM, Kidd JR, Livak KJ, Pakstis AJ, Kidd KK (1996) The world-wide distribution of allele frequencies at the human dopamine D4 receptor locus. Hum Genet 98:91–101
Wang E, Ding YC, Flodman P, Kidd JR, Kidd KK, Grady DL, Ryder OA, Spence MA (2004) The genetic architecture of selection at the human dopamine receptor D4 (DRD4) gene locus. Am J Hum Genet 74:931–944
Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, Sklar P (2005) Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry 57:1313–1323
Li D, Sham PC, Owen MJ, He L (2006) Meta-analysis shows significant association between dopamine system genes and attention deficit hyperactivity disorder (ADHD). Hum Mol Genet 15:2276–2284
Gizer IR, Ficks C, Waldman ID (2009) Candidate gene studies of ADHD: a meta-analytic review. Hum Genet 126:51–90
Belcher AM, Volkow ND, Moeller FG, Ferré S (2014) Personality traits and vulnerability or resilience to substance use disorders. Trends Cogn Sci 18:211–217
Zhong P, Liu W, Yan Z (2016) Aberrant regulation of synchronous network activity by the attention-deficit/hyperactivity disorder-associated human dopamine D4 receptor variant D4.7 in the prefrontal cortex. J Physiol 594:135–147
Bonaventura J, Quiroz C, Cai NS, Rubinstein M, Tanda G, Ferré S (2017) Key role of the dopamine D(4) receptor in the modulation of corticostriatal glutamatergic neurotransmission. Sci Adv 3:e1601631
González S, Rangel-Barajas C, Peper M, Lorenzo R, Moreno E, Ciruela F, Borycz J, Ortiz J et al (2012) Dopamine D4 receptor, but not the ADHD-associated D4.7 variant, forms functional heteromers with the dopamine D2S receptor in the brain. Mol Psychiatry 17:650–662
Asghari V, Sanyal S, Buchwaldt S, Paterson A, Jovanovic V, Van Tol HH (1995) Modulation of intracellular cyclic AMP levels by different human dopamine D4 receptor variants. J Neurochem 65:1157–1165
Sánchez-Soto M, Bonifazi A, Cai N-S, Ellenberger MP, Newman AH, Ferré S, Yano H (2016) Evidence for noncanonical neurotransmitter activation: norepinephrine as a dopamine D2-like receptor agonist. Mol Pharmacol 89:457–466
Yepes G, Guitart X, Rea W, Richard PA, Earley CJ, Quiroz C, Ferré S (2017) Targeting hypersensitive corticostriatal terminals in restless legs syndrome. Ann Neurol 82:951–960
Borroto-Escuela DO, Van Craenenbroeck K, Romero-Fernandez W, Guidolin D, Woods AS, Rivera A, Haegeman G, Agnati LF et al (2011) Dopamine D2 and D4 receptor heteromerization and its allosteric receptor-receptor interactions. Biochem Biophys Res Commun 404:928–934
Urizar E, Yano H, Kolster R, Galés C, Lambert N, Javitch JA (2011) CODA-RET reveals functional selectivity as a result of GPCR heteromerization. Nat Chem Biol 7:624–630
Guitart X, Navarro G, Moreno E, Yano H, Cai NS, Sánchez-Soto M, Kumar-Barodia S, Naidu YT et al (2014) Functional selectivity of allosteric interactions within G protein-coupled receptor oligomers: the dopamine D1-D3 receptor heterotetramer. Mol Pharmacol 86:417–429
Ferré S, Casadó V, Devi LA, Filizola M, Jockers R, Lohse MJ, Milligan G, Pin J-P et al (2014) G protein-coupled receptor oligomerization revisited: functional and pharmacological perspectives. Pharmacol Rev 66:413–434
Patel S, Freedman S, Chapman KL, Emms F, Fletcher AE, Knowles M, Marwood R, Mcallister G et al (1997) Biological profile of L-745,870, a selective antagonist with high affinity for the dopamine D4 receptor. J Pharmacol Exp Ther 283:636–647
Nilsson CL, Ekman A, Hellstrand M, Eriksson E (1996) Inverse agonism at dopamine D2 receptors. Haloperidol-induced prolactin release from GH4C1 cells transfected with the human D2 receptor is antagonized by R(−)-n-propylnorapomorphine, raclopride, and phenoxybenzamine. Neuropsychopharmacology 15:53–61
Acknowledgements
We thank Dr. J. A. Javitch (Columbia University, New York) for kindly providing some of the DNA constructs (D2SR-nRLuc, D2SR-cRLuc, and Gαi1-YFP).
Funding
This work is supported by the intramural funds of the National Institute on Drug Abuse, a grant from the Spanish “Ministerio de Economía y Competitividad,” and the European Regional Development Funds of the European Union (SAF2014-54840-R).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sánchez-Soto, M., Yano, H., Cai, NS. et al. Revisiting the Functional Role of Dopamine D4 Receptor Gene Polymorphisms: Heteromerization-Dependent Gain of Function of the D4.7 Receptor Variant. Mol Neurobiol 56, 4778–4785 (2019). https://doi.org/10.1007/s12035-018-1413-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1413-1