Abstract
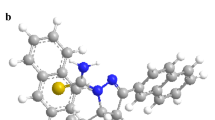
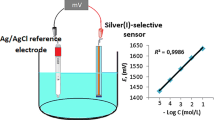
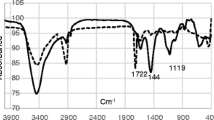
In this study, 5,10,15,20-tetrakis(p-chlorophenyl)porphyrin has been synthesized and used as an ionophore for the preparation of a poly(vinyl chloride) (PVC) membrane sensor for aluminium(III) ions. The optimum composition of the best performing membrane contained ionophore, bis(2-ethylhexyl) adipate, PVC and potassium tetrakis(p-chlorophenyl) borate in the ratio of 4.0:63.0:32.0:1.0 (mg). The prepared PVC membrane morphology has been analysed by scanning electron microscopy. The developed aluminium(III)-selective sensor works in a wide linear concentration range of 1.0 × 10–5 to 1.0 × 10–1 mol l–1 and, the detection limit of this sensor is 2.81 × 10−6 mol l–1. The sensor displays near-Nernstian slope of 25.0 ± 2.7 mV per decade for Al3+ ions. The aluminium(III)-selective sensor has a wide working pH range of 5.0–10.0. The sensor shows good reusability, long-term stability and a fast response time of less than 5 s. In addition, the sensor shows good selectivity for Al3+ ions over different cations. This aluminium(III)-selective sensor was successfully used as an indicator electrode in the potentiometric titration of Al3+ ions with EDTA. The developed sensor was successfully applied to the direct determination of Al3+ in different water samples.












Similar content being viewed by others
References
Ma Y-H, Yuan R, Chai Y-Q and Liu X-L 2010 Mater. Sci. Eng. C 30 209
Ahmed M J, Hoque M R, Khan A S M and Bhattacharjee S C 2010 Eurasian. J. Anal. Chem. 5 1
Silvestre A L P, Milani M I, Rossini E L, Pezza L and Pezza R P 2018 Spectrochim. Acta A Mol. Biomol. Spectrosc. 204 432
Arvand M and Asadollahzadeh S A 2008 Talanta 75 1046
Gürdere M B, Özbek O and Ceylan M 2016 Synt. Comm. 46 322
Bera R T, Sahoo S K, Mittal S K and Ashok Kumar S K 2010 Int. J. Electrochem. Sci. 5 29
Kumar R S and Ashok Kumar S K 2019 Inorg. Chem. Commun. 106 165
Flaten T P 2001 Brain Res. 55 187
Yari A, Darvishi L and Shamsipur M 2006 Anal. Chim. Acta 555 329
Harigaya K, Kuwahar Y and Nishi H 2008 Chem. Pharm. Bull. 56 475
Frankowski M, Zioła-Frankowska A, Kurzyca I, Novotný K, Vaculoviˇc T, Kanický, et al 2011 Environ. Monit. Assess. 182 71
Krishnan S S, Gillespie K A and Crapper D R 1972 Anal. Chem. 44 1469
Bradley C and Leung F Y 1994 Clin. Chem. 40 431
Zanjanchi M A, Noei H and Moghimi M 2006 Talanta 70 933
Zuziak J, Reczyński W, Baś B and Jakubowska M 2018 Anal. Biochem. 558 69
Işıldak Ö, Deligönül N and Özbek O 2019 Turk. J. Chem. 43 1149
Isildak Ö, Özbek O and Gürdere M B 2020 J. Anal. Test. 4 273
Isildak Ö, Özbek O and Yigit K M 2020 Bulg. Chem. Commun. 52 448
Özbek O, Isildak Ö and Isildak I 2021 Biochem. Eng. J. 176 108181
Isildak O and Özbek O 2021 Crit. Rev. Anal. Chem. 51 218
Özbek O, Isildak Ö, Gürdere M B and Cetin A 2021 Org. Commun. 14 228
Temelli B and Unaleroglu C 2009 Tetrahedron 65 2043
Zhao L, Zhao Y, Li R, Wu D, Xu R, Li S et al 2020 Chemosphere 238 24552
Huang D, Li X, Chen M, Chen F, Wan Z, Rui R et al 2019 J. Electroanal. Chem. 841 101
Özbek O, Isildak Ö and Berkel C 2020 J. Incl. Phenom. Macrocycl. Chem. 98 1
Isildak O and Özbek O 2020 J. Chem. Sci. 132 29
Adler A D, Longo F R, Finarelli J D, Goldmacher J, Assour J and Korsakoff L 1967 J. Org. Chem. 32 476
Liu F, Duan L, Wang Y-L, Zhang Q and Wang J-Y 2009 Synt. Comm. 39 3990
Isildak O, Özbek O and Yigit K M 2021 Int. J. Environ. Anal. Chem. 101 2035
Topcu C 2016 Talanta 161 623
Buck R P and Lindner E 1994 Pure Appl. Chem. 66 2527
Özbek O and Isildak Ö 2021 Int. J. Environ. Anal. Chem. https://doi.org/10.1080/03067319.2021.1877283
Umezawa Y, Bühlmann P, Umezawa K, Tohda K and Amemiya A S 2000 Pure Appl. Chem. 72 1851
Gupta V K, Jain A K, Singh L P and Khurana U 1997 Anal. Chim. Acta 355 33
Gupta V K, Jain A K and Kumar P 2006 Electrochim. Acta 52 736
Mousavi M F, Arvand-Barmchi M and Zanjanchi M A 2001 Electroanal. 13 1125
Abbaspour A, Esmaeilbeig A R, Jarrahpour A A, Khajeh B and Kia R 2002 Talanta 58 397
Saleh M B, Hassan S S M, Abdel Gaber A A and Abdel Kream N A 2001 Anal. Chim. Acta 434 247
Soleimani M and Afshar M G 2014 Russ. J. Electrochem. 50 554
Özbek O and Isildak Ö 2022 ChemistrySelect 7 e202103988
Acknowledgements
We thank the Tokat Gaziosmanpasa University Scientific Research Projects Commission (Project Number 2019/43) for the financial support. In addition, we would like to thank Research Assistant Caglar Berkel and M.Sc. student Alper Cetin for their contributions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Özbek, O., Isildak, Ö. Use of 5,10,15,20-tetrakis(p-chlorophenyl)porphyrin as sensor material: potentiometric determination of aluminium(III) ions. Bull Mater Sci 45, 114 (2022). https://doi.org/10.1007/s12034-022-02696-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-022-02696-3