Abstract
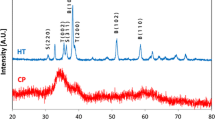
The thermal oxidation of CuS powder was examined under flow of nitrogen and dry air using thermogravimetry/differential thermal analysis. After 473 K, the oxidation of CuS occurred as a weight loss and accompanied with two overlapped exothermic peaks. The melting temperature delayed due to the formation of oxide and sulphate on the surface of the particles. X-ray diffraction revealed that the as-prepared thin films are mixed phases of hexagonal CuS, orthorhombic Cu2S and orthorhombic CuSO4. After annealing at 573 or 773 K, the films oxidized and new orthorhombic Cu8O phase appeared, and its intensity became dominant upon increasing the temperature and time. Nanoparticles morphology was observed for as-prepared films and upon annealing the nanoparticle became more rounded and bigger. The transmittance of the as-prepared films was almost zero over the entire measured range and it increased with increase in the annealing temperature and time, whereas the reflectance decreased. Both refractive and extinction coefficient values decreased with increase in annealing temperature and annealing time, while the bandgap virtually increased. The resistivity of the as-prepared film remained nearly constant until 543 K. Above 543 K the resistivity increased sharply. Negative and positive temperature coefficients in resistivity phenomena were explored in the annealed films and they were strongly dependent on both annealing temperature and time.















Similar content being viewed by others
References
Mohamed S H, El-Hagary M and Emam-Ismail M 2010 J. Phys. D: Appl. Phys. 43 075401
Zhang Y, Chong X, Sun H, Kedir M M, Kim K-J, Ohodnicki P R et al 2020 J. Mater. Chem. C 8 3069
Masar M, Urbanek M, Urbanek P, Machovska Z, Maslik J, Yadav R S et al 2019 Mater. Chem. Phys. 237 121823
Jeyabanu K, Devendran P, Manikandan A, Packiaraj R, Ramesh K and Nallamuthu N 2019 Physica B 573 92
Heidari G, Rabani M and Ramezanzadeh B 2017 Int. J. Hydrog. Energy 42 9545
Poudel K, Gautam M, Jin S G, Choi H-G, Yong C S and Kim J O 2019 Int. J. Pharm. 562 135
Sabah F A, Ahmed N M, Hassan Z and Rasheed H S 2016 Procedia Chem. 19 15
Diliegros-Godines C J, Lombardero-Juarez D I, Machorro-Mejía R, Silva González R and Pal M 2019 Opt. Mater. 91 147
Rastogi A C, Salkalachen S and Bhide V G 1978 Thin Solid Films 52 1
Mohamed S H, Hadia N M A, Awad M A and Hafez M I 2019 Appl. Phys. A 125 587
Sahoo A K, Mohanta P and Bhattacharyya A S 2015 IOP Conf. Series: Mater. Sci. Eng. 73 012123
Ramya M and Ganesan S 2010 Int. J. Pure Appl. Phys. 6 243
Dunn J G and Muzenda C 2001 Thermochimica 369 117
Dunn J G, Ginting A R and O’Connor B 1994 J. Therm. Anal. 41 671
Dixit S G, Mahadeshwar A R and Haram S K 1998 Colloids Surf. A: Physicochem. Eng. Asp. 133 69
Suja R, Geetha D and Ramesh P 2013 Int. J. Sci. Eng. Res. 4 1
Ross S D 1974 Sulfates and other oxy-anions of Group VI in V C Farmer (ed) The infrared spectra of minerals (London: Mineralogy Society) p 423
Boey H T, Tan W L, Abu Bakar N H H, Abu Bakar M and Ismail J 2007 J. Phys. Sci. 18 87
Selvi S S T, Linet J M and Sagadevan S 2018 J. Exp. Nanosci. 13 130
Arun K J, Batra A K, Krishna A, Bhat K, Aggarwal M D and Joseph Francis P J 2015 Am. J. Mater. Sci. 5 36
Debbichi L, Marco de Lucas M C, Pierson J F and Kruger P 2012 J. Phys. Chem. C 116 10232
Sathish, Rafi S M, Shaik H, Madhavi P, Kosuri Y R, Abdul Sattar S et al 2019 Mater. Sci. Semicond. Process. 96 127
Bagul S V, Chavhan S D and Sharma R 2007 J. Phys. Chem. Solids 68 1623
Grozdanov I and Najdoski M 1995 J. Solid State Chem. 114 469
Bennouna A and Ameziane E L 1991 Sol. Energy Mater. 22 201
Christy R S and Kumaran J T T 2014 J. Non-Oxide Glass. 6 13
Abazine K, Anakiou H, El Hasnaoui M, Graca M P F, Fonseca M A, Costa L C et al 2016 J. Compos. Mater. 50 3283
Kirstein K, Reichmann K, Preis W and Mitsche S 2011 J. Eur. Ceram. Soc. 31 2339
Drofenik M, Lisjak D and Zajc I 1997 J. Am. Ceram. Soc. 80 1741
Chowdhury U, Goswami S, Bhattacharya D, Midya A, Mandal P, Das P et al 2013 J. Appl. Phys. 114 194104
Alqahtani M S, Hadia N M A and Mohamed S H 2018 Optik 173 101
Sorokin G P and Paradenko A P 1966 Sov. Phys. J. 9 59
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohamed, S.H., Awad, M.A., Hafez, M.I. et al. Change in properties upon thermal treatment of copper sulphide powder and thin films. Bull Mater Sci 44, 81 (2021). https://doi.org/10.1007/s12034-021-02379-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-021-02379-5