Abstract
Osteosarcoma, usually originating in the stroma, is the most common primary bone cancer in adolescents, and its prognosis is poor. Surgery, adjuvant and neoadjuvant chemotherapy and radiation therapy are not satisfactory at the present time. Therefore, it is critical to develop novel therapeutic strategies to improve the quality of life and long-term survival rate of osteosarcoma patients. In this study, we discovered that zoledronic acid (ZOL) dramatically increased cell death in osteosarcoma cells, and this cytotoxicity was greatly reversed by liproxstatin-1 (a ferroptosis inhibitor). ZOL also had an obvious effect on lipid peroxidation and reactive oxygen species (ROS), which suggested that ZOL most certainly induces ferroptosis in osteosarcoma cells. In addition, we further found that ZOL increases cytochrome P450 oxidoreductase (POR) expression dose dependently in osteosarcoma cell lines. Knockdown of POR attenuated ZOL-induced cytotoxicity and attenuated the effect of ferroptosis in osteosarcoma cells, which indicated that POR plays an important role in ferroptosis. Moreover, we also found that ZOL inhibits osteosarcoma growth in vivo. Our findings suggest that ZOL induces ferroptosis by upregulating POR expression to increase ROS levels and upregulate lipid peroxidation levels in osteosarcoma cells. POR may be used as a therapeutic target to inhibit osteosarcoma.






Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ZOL:
-
Zoledronic acid
- ROS:
-
Reactive oxygen species
- DEGs:
-
Differentially expressed genes
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- GO:
-
Gene Ontology
- RIPA:
-
Radioimmunoprecipitation assay
- FITC:
-
Fluorescein isothiocyanate
References
Zhao J, Zhao Y, Ma X, Zhang B, Feng H. Targeting ferroptosis in osteosarcoma. J Bone Oncol. 2021;30:100380.
Kong H, Yu W, Chen Z, Li H, Ye G, Hong J, Xie Z, Chen K, Wu Y, Shen H. CCR9 initiates epithelial–mesenchymal transition by activating Wnt/β-catenin pathways to promote osteosarcoma metastasis. Cancer Cell Int. 2021;21:648.
Isakoff MS, Bielack SS, Meltzer P, Gorlick R. Osteosarcoma: current treatment and a collaborative pathway to success. J Clin Oncol. 2015;33:3029–35.
Lu KH, Lu EW, Lin CW, Yang JS, Yang SF. New insights into molecular and cellular mechanisms of zoledronate in human osteosarcoma. Pharmacol Ther. 2020;214:107611.
Lilienthal I, Herold N. Targeting molecular mechanisms underlying treatment efficacy and resistance in osteosarcoma: a review of current and future strategies. Int J Mol Sci. 2020;21:6885.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS, Morrison B, Stockwell BR. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72.
Li D, Li Y. The interaction between ferroptosis and lipid metabolism in cancer. Signal Transduct Target Ther. 2020;5:108.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Friedmann Angeli JP, Krysko DV, Conrad M. Ferroptosis at the crossroads of cancer-acquired drug resistance and immune evasion. Nat Rev Cancer. 2019;19:405–14.
Hassannia B, Vandenabeele P, Vanden Berghe T. Targeting ferroptosis to iron out cancer. Cancer Cell. 2019;35:830–49.
Yu M, Gai C, Li Z, Ding D, Zheng J, Zhang W, Lv S, Li W. Targeted exosome-encapsulated erastin induced ferroptosis in triple negative breast cancer cells. Cancer Sci. 2019;110:3173–82.
Sun Y, Berleth N, Wu W, Schlütermann D, Deitersen J, Stuhldreier F, Berning L, Friedrich A, Akgün S, Mendiburo MJ, Wesselborg S, Conrad M, Berndt C, Stork B. Fin56-induced ferroptosis is supported by autophagy-mediated GPX4 degradation and functions synergistically with mTOR inhibition to kill bladder cancer cells. Cell Death Dis. 2021;12:1028.
Hayashima K, Kimura I, Katoh H. Role of ferritinophagy in cystine deprivation-induced cell death in glioblastoma cells. Biochem Biophys Res Commun. 2021;539:56–63.
Luo Y, Gao X, Zou L, Lei M, Feng J, Hu Z. Bavachin induces ferroptosis through the STAT3/P53/SLC7A11 axis in Osteosarcoma cells. Oxid Med Cell Longev. 2021;2021:1783485.
Lin H, Chen X, Zhang C, Yang T, Deng Z, Song Y, Huang L, Li F, Li Q, Lin S, Jin D. EF24 induces ferroptosis in osteosarcoma cells through HMOX1. Biomed Pharmacother. 2021;136:111202.
Chen M, Jiang Y, Sun Y. KDM4A-mediated histone demethylation of SLC7A11 inhibits cell ferroptosis in osteosarcoma. Biochem Biophys Res Commun. 2021;550:77–83.
Xu Z, Chen L, Wang C, Zhang L, Xu W. MicroRNA-1287-5p promotes ferroptosis of osteosarcoma cells through inhibiting GPX4. Free Radic Res. 2022;55:1119–29.
Shi Y, Gong M, Deng Z, Liu H, Chang Y, Yang Z, Cai L. Tirapazamine suppress osteosarcoma cells in part through SLC7A11 mediated ferroptosis. Biochem Biophys Res Commun. 2021;567:118–24.
Liu Q, Wang K. The induction of ferroptosis by impairing STAT3/Nrf2/GPx4 signaling enhances the sensitivity of osteosarcoma cells to cisplatin. Cell Biol Int. 2019;43:1245–56.
Koppula P, Zhuang L, Gan B. Cytochrome P450 reductase (POR) as a ferroptosis fuel. Protein Cell. 2021;12:675–9.
Zou Y, Li H, Graham ET, Deik AA, Eaton JK, Wang W, Sandoval-Gomez G, Clish CB, Doench JG, Schreiber SL. Cytochrome P450 oxidoreductase contributes to phospholipid peroxidation in ferroptosis. Nat Chem Biol. 2020;16:302–9.
Yan B, Ai Y, Sun Q, Ma Y, Cao Y, Wang J, Zhang Z, Wang X. Membrane damage during ferroptosis is caused by oxidation of phospholipids catalyzed by the oxidoreductases POR and CYB5R1. Mol Cell. 2021;81:355-69.e310.
Guengerich FP, Waterman MR, Egli M. Recent structural insights into cytochrome P450 function. Trends Pharmacol Sci. 2016;37:625–40.
Russell RG, Rogers MJ. Bisphosphonates: from the laboratory to the clinic and back again. Bone. 1999;25:97–106.
Van Poznak CH, Temin S, Yee GC, Janjan NA, Barlow WE, Biermann JS, Bosserman LD, Geoghegan C, Hillner BE, Theriault RL, Zuckerman DS, Von Roenn JH. American society of clinical oncology executive summary of the clinical practice guideline update on the role of bone-modifying agents in metastatic breast cancer. J Clin Oncol. 2011;29:1221–7.
Ensrud KE. Bisphosphonates for postmenopausal osteoporosis. JAMA. 2021;325:96.
Clemons MJ, Dranitsaris G, Ooi WS, Yogendran G, Sukovic T, Wong BY, Verma S, Pritchard KI, Trudeau M, Cole DE. Phase II trial evaluating the palliative benefit of second-line zoledronic acid in breast cancer patients with either a skeletal-related event or progressive bone metastases despite first-line bisphosphonate therapy. J Clin Oncol. 2006;24:4895–900.
Rosen LS, Gordon D, Kaminski M, Howell A, Belch A, Mackey J, Apffelstaedt J, Hussein MA, Coleman RE, Reitsma DJ, Chen BL, Seaman JJ. Long-term efficacy and safety of zoledronic acid compared with pamidronate disodium in the treatment of skeletal complications in patients with advanced multiple myeloma or breast carcinoma: a randomized, double-blind, multicenter, comparative trial. Cancer. 2003;98:1735–44.
Kohno N, Aogi K, Minami H, Nakamura S, Asaga T, Iino Y, Watanabe T, Goessl C, Ohashi Y, Takashima S. Zoledronic acid significantly reduces skeletal complications compared with placebo in Japanese women with bone metastases from breast cancer: a randomized, placebo-controlled trial. J Clin Oncol. 2005;23:3314–21.
Oh JY, Kim EH, Lee YJ, Sai S, Lim SH, Park JW, Chung HK, Kim J, Vares G, Takahashi A, Jeong YK, Kim MS, Kong CB. Synergistic autophagy effect of miR-212-3p in zoledronic acid-treated in vitro and orthotopic in vivo models and in patient-derived osteosarcoma cells. Cancers (Basel). 2019;11:1812.
Kim EH, Kim MS, Takahashi A, Suzuki M, Vares G, Uzawa A, Fujimori A, Ohno T, Sai S. Carbon-ion beam irradiation alone or in combination with zoledronic acid effectively kills osteosarcoma cells. Cancers (Basel). 2020;12:698.
Terpos E, Raje N, Croucher P, Garcia-Sanz R, Leleu X, Pasteiner W, Wang Y, Glennane A, Canon J, Pawlyn C. Denosumab compared with zoledronic acid on PFS in multiple myeloma: exploratory results of an international phase 3 study. Blood Adv. 2021;5:725–36.
Chen Z, Kong H, Cai Z, Chen K, Wu B, Li H, Wang P, Wu Y, Shen H. Identification of MAP3K15 as a potential prognostic biomarker and correlation with immune infiltrates in osteosarcoma. Ann Transl Med. 2021;9:1179.
Shen Z, Song J, Yung BC, Zhou Z, Wu A, Chen X. Emerging strategies of cancer therapy based on ferroptosis. Adv Mater. 2018;30:e1704007.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural Science Foundation of China (81760041) and the Shenzhen Science and Technology Program (JCYJ20220530144201002).
Author information
Authors and Affiliations
Contributions
HJC: the conception and design of the study, drafting the article; YQR: analysis of data; LHN and Song Yichang: interpretation and acquisition of data; CK and CY: revising article, final approval of the version to be submitted. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
The study was approved by the Ethics Committee of The Eighth Affiliated Hospital of Sun Yat-sen University. Animal Studies was supported by The Eighth Affiliated Hospital of Sun Yat-sen University. All animal experiments were conducted in accordance with the National Institutes of Health Laboratory Animal Care and Use Guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12032_2023_1988_MOESM1_ESM.tif
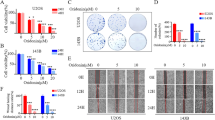
Supplementary file1 (TIF 27405 KB)—A, Colony formation capacity of MG63 and 143B cells, as assessed by a colony formation assay after 10 days. The colony formation assay showed ZOL inhibit the osteosarcoma cells. B, ZOL dose-dependently decreased the cell viability of osteosarcoma. C, Cell apoptosis were detected by flow cytometry which showed that ZOL can’ t cause apoptosis in OS cells. D, Western blot analysis showed that groups' protein expression levels appeared to be similar, which showed that ZOL can’ t induce autophagy in OS cells. E, After 2 weeks, the ZOL group and NC group which were subcutaneously injected with ZOL-treated 143B cells or normal 143B cells respectively both successfully formed xenograft tumors. (Every circle means each nude mice. Yellow means xenograft tumor has been formed, grey means xenograft tumor has not been formed) F, The change of tumor volume showed ZOL could inhibit the growth and proliferation of osteosarcoma. *P <0.05; **P <0.01
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiacong, H., Qirui, Y., Haonan, L. et al. Zoledronic acid induces ferroptosis by upregulating POR in osteosarcoma. Med Oncol 40, 141 (2023). https://doi.org/10.1007/s12032-023-01988-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-01988-w