Abstract
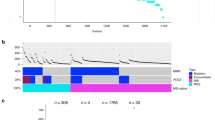
Mutation frequencies of common genetic alterations in colorectal cancer have been in the spotlight for many years. This study highlights few rare somatic mutations, which possess the attributes of a potential CRC biomarker yet are often neglected. Next-generation sequencing was performed over 112 tumor samples to detect genetic alterations in 31 rare genes in colorectal cancer. Mutations were detected in 26/31 (83.9 %) uncommon genes, which together contributed toward 149 gene mutations in 67/112 (59.8 %) colorectal cancer patients. The most frequent mutations include KDR (19.6 %), PTEN (17 %), FBXW7 (10.7 %), SMAD4 (10.7 %), VHL (8 %), KIT (8 %), MET (7.1 %), ATM (6.3 %), CTNNB1 (4.5 %) and CDKN2A (4.5 %). RB1, ERBB4 and ERBB2 mutations were persistent in 3.6 % patients. GNAS, FGFR2 and FGFR3 mutations were persistent in 1.8 % patients. Ten genes (EGFR, NOTCH1, SMARCB1, ABL1, STK11, SMO, RET, GNAQ, CSF1R and FLT3) were found mutated in 0.9 % patients. Lastly, no mutations were observed in AKT, HRAS, MAP2K1, PDGFR and JAK2. Significant associations were observed between VHL with tumor site, ERBB4 and SMARCB1 with tumor invasion, CTNNB1 with lack of lymph node involvement and CTNNB1, FGFR2 and FGFR3 with TNM stage. Significantly coinciding mutation pairs include PTEN and SMAD4, PTEN and KDR, EGFR and RET, EGFR and RB1, FBXW7 and CTNNB1, KDR and FGFR2, FLT3 and CTNNB1, RET and RB1, ATM and SMAD4, ATM and CDKN2A, ERBB4 and SMARCB1. This study elucidates few potential colorectal cancer biomarkers, specifically KDR, PTEN, FBXW7 and SMAD4, which are found mutated in more than 10 % patients.




Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359–86. doi:10.1002/ijc.29210.
Kalia M. Biomarkers for personalized oncology: recent advances and future challenges. Metabolism. 2015;64(3):S16–21.
Network Cancer Genome Atlas. Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487(7407):330–7.
Takahashi Y, Kitadai Y, Bucana CD, Cleary KR, Ellis LM. Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res. 1995;55(18):3964–8.
Loaiza-Bonilla A, Jensen C, Shroff S, Furth E, Bonilla-Reyes PA, Deik AF, et al. KDR mutation as a novel predictive biomarker of exceptional response to regorafenib in metastatic colorectal cancer. Cureus. 2016;8(2):e478. doi:10.7759/cureus.478.
Dong G, Guo X, Fu X, Wan S, Zhou F, Myers RE, et al. Potentially functional genetic variants in KDR gene as prognostic markers in patients with resected colorectal cancer. Cancer Sci. 2012;103(3):561–8.
Lee SY, Haq F, Kim D, Jun C, Jo HJ, Ahn SM, Lee WS. Comparative genomic analysis of primary and synchronous metastatic colorectal cancers. PLoS One. 2014;9(3):e90459.
Mockus SM, Potter CS, Stafford GA, Ananda G, Hinerfeld D, Tsongalis GJ. Targeting KDR mutations in lung adenocarcinoma. Cancer Res. 2015;75(15 Supplement):73–73.
Silva IP, Salhi A, Giles KM, Vogelsang M, Han SW, Ismaili N, et al. Identification of a novel pathogenic germline KDR variant in melanoma. Clin Cancer Res. 2016;22(10):2377–85.
Sammarco I, Capurso G, Coppola L, Bonifazi AP, Cassetta S, Delle Fave G, et al. Expression of the proto-oncogene c-KIT in normal and tumor tissues from colorectal carcinoma patients. Int J Colorectal Dis. 2004;19(6):545–53.
El-Serafi MM, Bahnassy AA, Ali NM, Eid SM, Kamel MM, Abdel-Hamid NA, Zekri ARN. The prognostic value of c-Kit, K-ras codon 12, and p53 codon 72 mutations in Egyptian patients with stage II colorectal cancer. Cancer. 2010;116(21):4954–64.
Zenali M. Retrospective review of MET gene mutations. Oncoscience. 2015;2(5):533.
Malapelle U, Pisapia P, Sgariglia R, Vigliar E, Biglietto M, Carlomagno C, et al. Less frequently mutated genes in colorectal cancer: evidences from next-generation sequencing of 653 routine cases. J Clin Pathol. 2016;69(9):767–71.
Lee JW, Soung YH, Seo SH, Kim SY, Park CH, Wang YP, et al. Somatic mutations of ERBB2 kinase domain in gastric, colorectal, and breast carcinomas. Clin Cancer Res. 2006;12(1):57–61.
Soung YH, Lee JW, Kim SY, Wang YP, Jo KH, Moon SW, et al. Somatic mutations of the ERBB4 kinase domain in human cancers. Int J Cancer. 2006;118(6):1426–9.
Williams CS, Bernard JK, Beckler MD, Almohazey D, Washington MK, Smith JJ, Frey MR. ERBB4 is over-expressed in human colon cancer and enhances cellular transformation. Carcinogenesis. 2015;36(7):710–8.
Matsuda Y, Ishiwata T, Yamahatsu K, Kawahara K, Hagio M, Peng WX, et al. Overexpressed fibroblast growth factor receptor 2 in the invasive front of colorectal cancer: a potential therapeutic target in colorectal cancer. Cancer Lett. 2011;309(2):209–19.
Jang JH, Shin KH, Park JG. Mutations in fibroblast growth factor receptor 2 and fibroblast growth factor receptor 3 genes associated with human gastric and colorectal cancers. Cancer Res. 2001;61(9):3541–3.
Luo Y, Tsuchiya KD, Park DI, Fausel R, Kanngurn S, Welcsh P, et al. RET is a potential tumor suppressor gene in colorectal cancer. Oncogene. 2013;32(16):2037–47.
Chen W, Drakos E, Grammatikakis I, Schlette EJ, Li J, Leventaki V, et al. mTOR signaling is activated by FLT3 kinase and promotes survival of FLT3-mutated acute myeloid leukemia cells. Mol Cancer. 2010;9(1):1.
Lian QF, Zhan XJ. Identification of differential gene expressions in colorectal cancer and polyp by cDNA microarray. World J Gastroenterol. 2012;18(6):570–5.
Lan YT, Jen-Kou L, Lin CH, et al. Mutations in the RAS and PI3 K pathways are associated with metastatic location in colorectal cancers. J Surg Oncol. 2015;111:905–10.
Day FL, Jorissen RN, Lipton L, et al. PIK3CA and PTEN gene and exon mutation-specific clinicopathologic and molecular associations in colorectal cancer. Clin Cancer Res. 2013;19:3285–96.
Berg M, Danielsen SA, Ahlquist T, Merok MA, et al. DNA sequence profiles of the colorectal cancer critical gene set KRAS-BRAF-PIK3CA-PTEN-TP53 related to age at disease onset. PLoS ONE. 2010;5(11):e13978.
Salmena L, Carracedo A, Pandolfi PP. Tenets of PTEN tumor suppression. Cell. 2008;133(3):403–14.
Rychahou PG, Jackson LN, Silva SR, et al. Targeted molecular therapy of the PI3 K pathway: therapeutic significance of PI3 K subunit targeting in colorectal carcinoma. Ann Surg. 2006;243(6):833–44.
Colakoglu T, Yildirim S, Kayaselcuk F, et al. Clinicopathological significance of PTEN loss and the phosphoinositide 3-kinase/Akt pathway in sporadic colorectal neoplasms: is PTEN loss predictor of local recurrence? Am J Surg. 2008;195(6):719–25.
Avizienyte E, Roth S, Loukola A, Hemminki A, Lothe RA, Stenwig AE, et al. Somatic mutations in LKB1 are rare in sporadic colorectal and testicular tumors. Cancer Res. 1998;58(10):2087–90.
Prior IA, Lewis PD, Mattos C. A comprehensive survey of Ras mutations in cancer. Cancer Res. 2012;72(10):2457–67.
Bai J, Gao J, Mao Z, Wang J, Li J, Li W, et al. Genetic mutations in human rectal cancers detected by targeted sequencing. J Hum Genet. 2015;60(10):589–96.
Cai ZX, Tang XD, Gao HL, Tang C, Nandakumar V, et al. APC, FBXW7, KRAS, PIK3CA, and TP53 gene mutations in human colorectal cancer tumors frequently detected by next-generation DNA sequencing. J Mol Genet Med. 2014;8. doi:10.4172/1747-0862.1000145.
Han SW, Kim HP, Shin JY, Jeong EG, Lee WC, Lee KH, et al. Targeted sequencing of cancer-related genes in colorectal cancer using next-generation sequencing. PLoS ONE. 2013;8(5):e64271.
Kuwai T, Kitadai Y, Tanaka S, Hiyama T, Tanimoto K, Chayama K. Mutation of the von Hippel-Lindau (VHL) gene in human colorectal carcinoma: association with cytoplasmic accumulation of hypoxia-inducible factor (HIF)-1α. Cancer Sci. 2004;95(2):149–53.
Thompson D, Duedal S, Kirner J, McGuffog L, Last J, Reiman A, et al. Cancer risks and mortality in heterozygous ATM mutation carriers. J. Nat. Cancer Inst. 2005;97(11):813–22.
Bougatef K, Ouerhani S, Moussa A, Kourda N, Coulet F, Colas C, et al. Prevalence of mutations in APC, CTNNB1, and BRAF in Tunisian patients with sporadic colorectal cancer. Cancer Genet Cytogenet. 2008;187(1):12–8.
Luchtenborg M, Weijenberg MP, Wark PA, Saritas AM, Roemen GM, van Muijen GN, et al. Mutations in APC, CTNNB1 and K-ras genes and expression of hMLH1 in sporadic colorectal carcinomas from the Netherlands Cohort Study. BMC Cancer. 2005;5(1):1.
Trzeciak L, Hennig E, Kolodziejski J, Nowacki M, Ostrowski J. Mutations, methylation and expression of CDKN2a/p16 gene in colorectal cancer and normal colonic mucosa. Cancer Lett. 2001;163(1):17–23.
Xing X, Cai W, Shi H, Wang Y, Li M, Jiao J, Chen M. The prognostic value of CDKN2A hypermethylation in colorectal cancer: a meta-analysis. Br J Cancer. 2013;108(12):2542–8.
Watanabe M, Sowa Y, Yogosawa M, Sakai T. Novel MEK inhibitor trametinib and other retinoblastoma gene (RB)-reactivating agents enhance efficacy of 5-fluorouracil on human colon cancer cells. Cancer Sci. 2013;104(6):687–93.
Fumagalli D, Gavin PG, Taniyama Y, Kim SI, Choi HJ, Paik S, Pogue-Geile KL. A rapid, sensitive, reproducible and cost-effective method for mutation profiling of colon cancer and metastatic lymph nodes. BMC Cancer. 2010;10:101.
Li T, Liao X, Lochhead P, Morikawa T, Yamauchi M, Nishihara R, et al. SMO expression in colorectal cancer: associations with clinical, pathological, and molecular features. Ann Surg Oncol. 2014;21(13):4164–73.
Acknowledgments
We thank the Department of Research at Sir Ganga Ram Hospital, Delhi, India, and Strand Life Sciences, Bangalore, India, and Mrs. Parul Takkar for their support. This study has been funded by Merck Serono, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in this study.
Rights and permissions
About this article
Cite this article
Jauhri, M., Bhatnagar, A., Gupta, S. et al. Targeted molecular profiling of rare genetic alterations in colorectal cancer using next-generation sequencing. Med Oncol 33, 106 (2016). https://doi.org/10.1007/s12032-016-0820-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-016-0820-2