Abstract
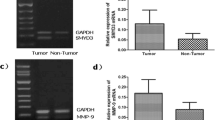
This study aimed to investigate mRNA and protein expressions of SET and MYND domain-containing protein 3 (SMYD3), STAT3, and phosphorylated STAT3 (pSTAT3) in gastric cancer (GC). This study was also conducted to explore the correlations between these proteins and biological behaviors of GC. SMYD3, STAT3, and pSTAT3 expressions were detected in GC tissues and adjacent non-tumor tissues by semiquantitative/quantitative reverse transcription polymerase chain reaction and Western blot analysis. SMYD3, STAT3, and pSTAT3 expressions in tissue sections were evaluated by immunohistochemistry. Staining results were compared with clinicopathological characteristics and the outcome of patients. The mRNA expression levels of SMYD3 or STAT3 and the protein expression levels of SMYD3, STAT3, or pSTAT3 in GC tissues were significantly higher than those in adjacent non-tumor tissues. Lymph node metastasis was identified as an independently relative factor for SMYD3 expression; the degree of differentiation and serosal invasion were identified as the independently relative factors for pSTAT3 expression in GC tissues. SMYD3 expression and STAT3 or pSTAT3 expressions in GC tissues were significantly and positively correlated. Multivariate analysis results demonstrated that primary tumor location, lymph node metastasis, SMYD3 expression, and pSTAT3 expression were independent prognostic indicators of GC. pSTAT3 expression was an optimal prognostic predictor of patients, as identified by Cox regression with Akaike’s information criterion value calculation. High SMYD3 and pSTAT3 expressions may indicate poor prognosis of patients with GC.




Similar content being viewed by others

References
Yuan Y. A survey and evaluation of population-based screening for gastric cancer. Cancer Biol Med. 2013;10:72–80.
Lin Y, Ueda J, Kikuchi S, et al. Comparative epidemiology of gastric cancer between Japan and China. World J Gastroenterol. 2011;17:4421–8.
Abe N, Watanabe T, Suzuki K, et al. Risk factors predictive of lymph node metastasis in depressed early gastric cancer. Am J Surg. 2002;183:168–72.
Hamamoto R, Furukawa Y, Morita M, et al. SMYD3 encodes a histone methyltransferase involved in the proliferation of cancer cells. Nat Cell Biol. 2004;6:731–40.
Kim H, Heo K, Kim JH, et al. Requirement of histone methyltransferase SMYD3 for estrogen receptor-mediated transcription. J Biol Chem. 2009;284:19867–77.
Chen LB, Xu JY, Yang Z, et al. Silencing SMYD3 in hepatoma demethylates RIZI promoter induces apoptosis and inhibits cell proliferation and migration. World J Gastroenterol. 2007;13:5718–24.
He C, Xu J, Zhang J, et al. High expression of trimethylated histone H3 lysine 4 is associated with poor prognosis in hepatocellular carcinoma. Hum Pathol. 2012;43:1425–35.
Luo XG, Xi T, Guo S, et al. Effects of SMYD3 overexpression on transformation, serum dependence, and apoptosis sensitivity in NIH3T3 cells. IUBMB Life. 2009;61:679–84.
Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798–809.
Buettner R, Mora LB, Jove R. Activated STAT signaling in human tumors provides novel molecular targets for therapeutic intervention. Clin Cancer Res. 2002;8:945–54.
Turkson J, Jove R. STAT proteins: novel molecular targets for cancer drug discovery. Oncogene. 2000;19:6613–26.
Deng JY, Sun D, Liu XY, et al. STAT-3 correlates with lymph node metastasis and cell survival in gastric cancer. World J Gastroentrol. 2010;16:5380–7.
Kim DY, Cha ST, Ahn DH, et al. STAT3 expression in gastric cancer indicates a poor prognosis. J Gastroenterol Hepatol. 2009;24:646–51.
Dolled-Filhart M, Camp RL, Kowalski DP, et al. Tissue microarray analysis of signal transducers and activators of transcription 3 (Stat 3) and phospho-Stat3 (Tyr705) in node-negative breast cancer shows nuclear localization is associated with a better prognosis. Clin Cancer Res. 2003;9:594–600.
Deng J, Liang H, Ying G, et al. Methylation of CpG sites in RNF180 DNA promoter prediction poor survival of gastric cancer. Oncotarget. 2014;5:3173–83.
Hamamoto R, Silva FP, Tsuge M, et al. Enhanced SMYD3 expression is essential for the growth of breast cancer cells. Cancer Sci. 2006;97:113–8.
Wang SZ, Luo XG, Shen J, et al. Knockdown of SMYD3 by RNA interference inhibits cervical carcinoma cells growth and invasion in vitro. BMB Rep. 2008;41:294–9.
Groner B. Determinants of the extent and duration of STAT3 signaling. Jak-Stat. 2012;1:211–5.
Shah NG, Trivedi TI, Tankshali RA, et al. Stat3 expression in oral squamous cell carcinoma: association with clinicopathological parameters and survival. Int J Biol Mark. 2006;21:175–83.
Gong W, Wang L, Yao JC, et al. Expression of activated signal transducer and activator of transcription 3 predicts expression of vascular endothelial growth factor and angiogenic phenotype of human gastric cancer. Clin Cancer Res. 2005;11:1386–93.
Siveen KS, Sikka S, Surana R, et al. Targeting the STAT3 signaling pathway in cancer: role of synthetic and natural inhibitors. Biochim Biophys Acta. 2014;1845:136–54.
Gao F, Lv Y, Zhu Y, et al. Correlation of epigenetic aberrance with STAT3 signaling pathway in gastric carcinogenesis. Dig Dis Sci. 2012;57:2055–62.
Choi JH, Ahn MJ, Park CK, et al. Phospho-Stat3 expression and correlation with VEGF, p53, and Bcl-2 in gastric carcinoma using tissue microarray. APMIS. 2006;114:619–25.
Deng J, Liang H, Zhang R, et al. STAT3 is associated with lymph node metastasis in gastric cancer. Tumor Biol. 2013;34:2791–800.
Acknowledgments
This work was supported by National Natural Science Foundation of China (No. 31470816).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Deng Jingyu is the Joint First Author.
Rights and permissions
About this article
Cite this article
Liu, Y., Deng, J., Luo, X. et al. Overexpression of SMYD3 was associated with increased STAT3 activation in gastric cancer. Med Oncol 32, 404 (2015). https://doi.org/10.1007/s12032-014-0404-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0404-y