Abstract
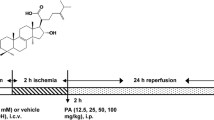
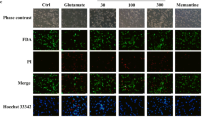
Ischemic stroke is one of the most dangerous acute diseases which causes death or deformity. Apoptosis has been shown to play an important role in the development and pathogenesis of cerebral ischemia–reperfusion injury (I/R injury), but the related mechanism is unclear. Levo-tetrahydropalmatine (l-THP), a bioactive ingredient extracted from the Chinese herb Corydalis, can penetrate the blood–brain barrier and exert various pharmacological effects on neural tissues. The present study examined the neuroprotective effect of l-THP on neuronal apoptosis induced by cerebral I/R injury. Results showed that pretreatment with l-THP (12.5, 25, and 50 mg/kg) improved neurological outcomes and reduced infarct volume and cerebral edema in comparison with the brains of the middle cerebral artery occlusion (MCAO) group. These findings provided evidence for the neuroprotective effects of l-THP against cerebral I/R injury. Furthermore, administration of l-THP enhanced the expression of Bcl-2 and attenuated the content of Bax, cleaved caspase-3, and PARP. l-THP could improve the reduction of NeuN-positive cells induced by I/R injury. These results suggested that l-THP could inhibit neuroapoptosis in cerebral ischemic rats. c-Abl was discovered as the critical protein responsible for neurocyte apoptosis; however, few data have been published on the relation between ischemic stroke and the expression of c-Abl. We found that both c-Abl expression and neuronal apoptosis were significantly increased in the MCAO group, while pretreatment with l-THP could ameliorate this effect. Therefore, we deduced that reduced c-Abl overexpression may play a role in the anti-apoptosis effect of l-THP after cerebral I/R injury. Thus, l-THP may provide a potential therapeutic approach for the treatment of ischemic stroke.

ᅟ






Similar content being viewed by others
Abbreviations
- I/R:
-
Ischemia/reperfusion
- l-THP:
-
Levo-tetrahydropalmatine
- MCAO:
-
Middle cerebral artery occlusion
- TTC:
-
Triphenyltetrazolium chloride
References
Andresen L, Theodorou K, Grünewald S, Czech-Zechmeister B, Könnecke B, Lühder F, Trendelenburg G (2016) Evaluation of the therapeutic potential of anti-TLR4-antibody MTS510 in experimental stroke and significance of different routes of application. PLoS One 11:e0148428. https://doi.org/10.1371/journal.pone.0148428
Brahmachari S, Ge P, Lee SH, Kim D, Karuppagounder SS, Kumar M, Mao X, Shin JH, Lee Y, Pletnikova O, Troncoso JC, Dawson VL, Dawson TM, Ko HS (2016) Activation of tyrosine kinase c-Abl contributes to α-synuclein-induced neurodegeneration. J Clin Investig 126:2970–2988. https://doi.org/10.1172/JCI85456
Broughton BRS, Reutens DC, Sobey CG (2009) Apoptotic mechanisms after cerebral ischemia. Stroke 40:e331–e339
Chung KS, Han G, Kim BK, Kim HM, Yang JS, Ahn J, Lee K, Song KB, Won M (2013) A novel antitumor piperazine alkyl compound causes apoptosis by inducing RhoB expression via ROS-mediated c-Abl/p38 MAPK signaling. Cancer Chemother Pharmacol 72:1315–1324. https://doi.org/10.1007/s00280-013-2310-y
Dawson TM, Dawson VL (2017) Mitochondrial mechanisms of neuronal cell death: potential therapeutics. Annu Rev Pharmacol Toxicol 57:437–454. https://doi.org/10.1146/annurev-pharmtox-010716-105001
Han Y, Zhang W, Tang Y, Bai W, Yang F, Xie L, Li X, Zhou S, Pan S, Chen Q, Ferro A, Ji Y (2012) L-tetrahydropalmatine, an active component of corydalis yanhusuo W.T. Wang, protects against myocardial ischaemia-reperfusion injury in rats. PLoS One 7:e38627. https://doi.org/10.1371/journal.pone.0038627
Hantschel O, Superti-Furga G (2004) Regulation of the c-Abl and Bcr-Abl tyrosine kinases. Nat Rev Mol Cell Biol 5:33–44
Healey JS, Oldgren J, Ezekowitz M, Zhu J, Pais P, Wang J, Commerford P, Jansky P, Avezum A, Sigamani A (2016) Occurrence of death and stroke in patients in 47 countries 1 year after presenting with atrial fibrillation: a cohort study. Lancet 388:1161–1169
Ito Y, Pandey P, Mishra NC, Kumar S, Narula N, Kharbanda S, Saxena S, Kufe D (2001) Targeting of the c-Abl tyrosine kinase to mitochondria in endoplasmic reticulum stress-induced apoptosis. Mol Cell Biol 21:6233–6242. https://doi.org/10.1128/MCB.21.18.6233
Jung JE, Kim GS, Chen H, Maier CM, Narasimhan P, Song YS, Niizuma K, Katsu M, Okami N, Yoshioka H, Sakata H, Goeders CE, Chan PH (2010) Reperfusion and neurovascular dysfunction in stroke: from basic mechanisms to potential strategies for neuroprotection. Mol Neurobiol 41:172–179
Kang D-W, Moon J-Y, Choi J-G, Kang SY, Ryu Y, Park JB, Lee JH, Kim HW (2016) Antinociceptive profile of levo-tetrahydropalmatine in acute and chronic pain mice models: role of spinal sigma-1 receptor. Sci Rep 6. doi:https://doi.org/10.1038/srep37850
Karunakaran S, Ravindranath V (2009) Activation of p38 MAPK in the substantia nigra leads to nuclear translocation of NF-??B in MPTP-treated mice: implication in Parkinson’s disease. J Neurochem 109:1791–1799. https://doi.org/10.1111/j.1471-4159.2009.06112.x
Karuppagounder SS, Brahmachari S, Lee Y, Dawson VL, Dawson TM, Ko HS (2014) The c-Abl inhibitor, nilotinib, protects dopaminergic neurons in a preclinical animal model of Parkinson’s disease. Sci Rep 4. doi:https://doi.org/10.1038/srep04874
Kharbanda S, Pandey P, Yamauchi T, Kumar S, Kaneki M, Kumar V, Bharti A, Yuan ZM, Ghanem L, Rana A, Weichselbaum R, Johnson G, Kufe D (2000) Activation of MEK kinase 1 by the c-Abl protein tyrosine kinase in response to DNA damage. Mol Cell Biol 20:4979–4989. https://doi.org/10.1128/MCB.20.14.4979-4989.2000
Mao XW, Pan CS, Huang P, Liu YY, Wang CS, Yan L, Hu BH, Chang X, He K, Mu HN, Li Q, Sun K, Fan JY, Han JY (2015) Levo-tetrahydropalmatine attenuates mouse blood-brain barrier injury induced by focal cerebral ischemia and reperfusion: involvement of Src kinase. Sci Rep 5. doi:https://doi.org/10.1038/srep11155
Martinon F (2010) Signaling by ROS drives inflammasome activation. Eur J Immunol 40:616–619
Moskowitz MA, Lo EH, Iadecola C (2010) The science of stroke: mechanisms in search of treatments. Neuron 67:181–198
Panjarian S, Iacob RE, Chen S, Engen JR, Smithgall TE (2013) Structure and dynamic regulation of abl kinases. J Biol Chem 288:5443–5450
Schlatterer SD, Acker CM, Davies P (2011) C-Abl in neurodegenerative disease. J Mol Neurosci 45:445–452
Shafi O (2016) Inverse relationship between Alzheimer’s disease and cancer, and other factors contributing to Alzheimer’s disease: a systematic review. BMC Neurol 16:236. https://doi.org/10.1186/s12883-016-0765-2
Sommer CJ (2017) Ischemic stroke: experimental models and reality. Acta Neuropathol 133:245–261
Sun X, Majumder P, Shioya H, Wu F, Kumar S, Weichselbaum R, Kharbanda S, Kufe D (2000) Activation of the cytoplasmic c-Abl tyrosine kinase by reactive oxygen species. J Biol Chem 275:17237–17240. https://doi.org/10.1074/jbc.C000099200
Wang JB, Mantsch JR (2012) l-tetrahydropalamatine: a potential new medication for the treatment of cocaine addiction. Future Med Chem 4:177–186. https://doi.org/10.4155/fmc.11.166
Wang X, Li R, Wang X, Fu Q, Ma S (2015) Umbelliferone ameliorates cerebral ischemia-reperfusion injury via upregulating the PPAR gamma expression and suppressing TXNIP/NLRP3 inflammasome. Neurosci Lett 600:182–187. https://doi.org/10.1016/j.neulet.2015.06.016
Wang W, Li M, Wang Y, Li Q, Deng G, Wan J, Yang Q, Chen Q, Wang J (2016) GSK-3β inhibitor TWS119 attenuates rtPA-induced hemorrhagic transformation and activates the Wnt/β-catenin signaling pathway after acute ischemic stroke in rats. Mol Neurobiol 53:7028–7036. https://doi.org/10.1007/s12035-015-9607-2
Wu R, Chen H, Ma J, He Q, Huang Q, Liu Q, Li M, Yuan Z (2016) C-Abl-p38α signaling plays an important role in MPTP-induced neuronal death. Cell Death Differ 23:542–552. https://doi.org/10.1038/cdd.2015.135
Xu YD, Cui C, Sun MF et al (2017) Neuroprotective effects of loganin on MPTP-induced Parkinson’s disease mice: neurochemistry, glial reaction and autophagy studies. J Cell Biochem 118:3495–3510. https://doi.org/10.1002/jcb.26010
Yasuda S, Sugiura H, Tanaka H, Takigami S, Yamagata K (2011) p38 MAP kinase inhibitors as potential therapeutic drugs for neural diseases. Cent Nerv Syst Agents Med Chem 11:45–59. https://doi.org/10.2174/187152411794961040
Yue K, Ma B, Ru Q, Chen L, Gan Y, Wang D, Jin G, Li C (2012) The dopamine receptor antagonist levo-tetrahydropalmatine attenuates heroin self-administration and heroin-induced reinstatement in rats. Pharmacol Biochem Behav 102:1–5. https://doi.org/10.1016/j.pbb.2012.03.014
Funding
This research is supported by Chinese Nature Science Foundation (81573701), China Postdoctoral Science Foundation (2017 M611958), and Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The protocol was approved by the Animal Ethics Committee of China Pharmaceutical University and was in agreement with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Conflict of Interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Sun, R., Song, Y., Li, S. et al. Levo-tetrahydropalmatine Attenuates Neuron Apoptosis Induced by Cerebral Ischemia–Reperfusion Injury: Involvement of c-Abl Activation. J Mol Neurosci 65, 391–399 (2018). https://doi.org/10.1007/s12031-018-1063-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1063-9