Abstract
Purpose
Although adjuvant chemotherapy (AC) increases survival in early-stage gastric cancer, the effect of the time between gastrectomy and the initiation of AC on survival has not been clearly demonstrated. This study aimed to examine the effect of AC timing on survival.
Methods
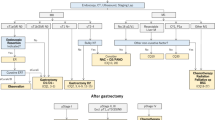
The data of patients who received AC in the postoperative period with the diagnosis of stage II and stage III gastric cancer were analyzed retrospectively. The patients were separated into two groups based on a 4-week cut-off value between the date of gastrectomy and the initiation of AC.
Results
There were 151 patients enrolled in the study. It was determined that 63 (41.7%) patients started AC in the first 4 weeks and 88 (58.3%) patients after the first 4 weeks. Patients who received AC during the first 4 weeks had a median recurrence-free survival (RFS) of 46 months, while those who received AC after 4 weeks had a median RFS of 29 months (p = 0.039). The median overall survival (OS) for patients administered AC in the first 4 weeks was 65 months, compared to 45 months for those administered AC after 4 weeks (p = 0.036). The early time interval from surgery to AC resulted as an independent prognostic factor for both OS and RFS.
Conclusion
The optimal time to start AC in patients with gastric cancer who underwent curative resection is unknown. This study reported that an interval shorter than 4 weeks was an independent prognostic risk factor for both OS and RFS.



Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article or available from the corresponding author on reasonable request.
References
Noh SH, Park SR, Yang H-K, Chung HC, Chung I-J, Kim S-W, Kim H-H, Choi J-H, Kim H-K, Yu W. Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol. 2014;15:1389–96. https://doi.org/10.1016/S1470-2045(14)70473-5.
Sasako M, Sakuramoto S, Katai H, Kinoshita T, Furukawa H, Yamaguchi T, Nashimoto A, Fujii M, Nakajima T, Ohashi Y. Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol. 2011;29:4387–93. https://doi.org/10.1200/JCO.2011.36.5908.
De Steur W, van Amelsfoort R, Hartgrink H, Putter H, Kranenbarg EM-K, van Grieken N, van Sandick J, Claassen Y, Braak J, Jansen E. Adjuvant chemotherapy is superior to chemoradiation after D2 surgery for gastric cancer in the per-protocol analysis of the randomized CRITICS trial. Ann Oncol. 2021;32:360–7. https://doi.org/10.1016/j.annonc.2020.11.004.
Bang Y-J, Van Cutsem E, Fuchs CS, Ohtsu A, Tabernero J, Ilson DH, Hyung WJ, Strong VE, Goetze TO, Yoshikawa T. KEYNOTE-585: phase III study of perioperative chemotherapy with or without pembrolizumab for gastric cancer. Future Oncol. 2019;15:943–952. https://doi.org/10.2217/fon-2018-0581
Fujitani K, Kurokawa Y, Takeno A, Endoh S, Ohmori T, Fujita J, Yamasaki M, Takiguchi S, Mori M, Doki Y. Time to initiation or duration of S-1 adjuvant chemotherapy; which really impacts on survival in stage II and III gastric cancer? Gastric Cancer. 2018;21:446–52. https://doi.org/10.1007/s10120-017-0767-9.
Qu J-L, Qu X-J, Li X, Zhang J-D, Teng Y-E, Jin B, Zhao M-F, Yu P, Liu J, Li D-Y. Early initiation of fluorouracil-based adjuvant chemotherapy improves survival in patients with resectable gastric cancer. J Buon. 2015;20:800–7.
Kang SY, Ahn MS, Song GW, Choi YW, Lee HW, Jeong SH, Park JS, Cho YK, Han SU, Sheen SS. Does the timing of adjuvant chemotherapy for gastric cancer influence patient outcome? Acta Oncol. 2015;54:1231–4. https://doi.org/10.3109/0284186X.2014.1000467.
Park HS, Jung M, Kim HS, Kim H-I, An JY, Cheong J-H, Hyung WJ, Noh SH, Kim YI, Chung HC. Proper timing of adjuvant chemotherapy affects survival in patients with stage 2 and 3 gastric cancer. Ann Surg Oncol. 2015;22:224–31. https://doi.org/10.1245/s10434-014-3949-2.
Huang S-M, Chen Y-C, Chen W-Y, Yang L-Y, Tsan D-L, Tsang N-M, Yap W-K, Tsai C-S, Leung W-M, Hong J-H. Optimal timing for postsurgical adjuvant therapy in patients with gastric cancer: a propensity score matching study. J Cancer. 2019;10:332–40. https://doi.org/10.7150/jca.27753.
Guideline Committee of the Korean Gastric Cancer Association (KGCA), Development Working Group & Review Panel. Korean practice guideline for gastric cancer 2018: an evidence-based, multi-disciplinary approach. J Gastric Cancer. 2019;19:1–48. https://doi.org/10.5230/jgc.2019.19.e8.
Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer. 2021;24:1–21. https://doi.org/10.1007/s10120-020-01042-y
Lordick F, Carneiro F, Cascinu S, Fleitas T, Haustermans K, Piessen G, Vogel A, Smyth E: Gastric cancer,. ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33:1005–20. https://doi.org/10.1016/j.annonc.2022.07.004.
Gunduz N, Fisher B, Saffer EA. Effect of surgical removal on the growth and kinetics of residual tumor. Cancer Res. 1979;39:3861–5.
Ono I, Gunji H, Suda K, Iwatsuki K, Kaneko F. Evaluation of cytokines in donor site wound fluids. Scand J Plast Reconstr Surg Hand Surg. 1994;28:269–73. https://doi.org/10.3109/02844319409022010.
Shannon C, Ashley S, Smith I. Does timing of adjuvant chemotherapy for early breast cancer influence survival? J Clin Oncol. 2003;21:3792–7. https://doi.org/10.1200/JCO.2003.01.073.
Biagi JJ, Raphael MJ, Mackillop WJ, Kong W, King WD, Booth CM. Association between time to initiation of adjuvant chemotherapy and survival in colorectal cancer: a systematic review and meta-analysis. JAMA. 2011;305:2335–42. https://doi.org/10.1001/jama.2011.749.
Yu KD, Huang S, Zhang JX, Liu GY, Shao ZM. Association between delayed initiation of adjuvant CMF or anthracycline-based chemotherapy and survival in breast cancer: a systematic review and meta-analysis. BMC Cancer. 2013;13:240. https://doi.org/10.1186/1471-2407-13-240.
Des Guetz G, Nicolas P, Perret GY, Morere JF, Uzzan B. Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair survival? A meta-analysis. Eur J Cancer. 2010;46:1049–55. https://doi.org/10.1016/j.ejca.2010.01.020.
Greenleaf EK, Kulaylat AN, Hollenbeak CS, Almhanna K, Wong J. Timing of adjuvant chemotherapy and impact on survival for resected gastric cancer. Ann Surg Oncol. 2016;23:4203–13. https://doi.org/10.1245/s10434-016-5464-0.
Petrelli F, Zaniboni A, Ghidini A, Ghidini M, Turati L, Pizzo C, Ratti M, Libertini M, Tomasello G. Timing of adjuvant chemotherapy and survival in colorectal, gastric, and pancreatic cancer. A systematic review and meta-analysis Cancers (Basel). 2019;11:550. https://doi.org/10.3390/cancers11040550.
Ahn GT, Baek SK, Han JJ, Kim HJ, Jeong SJ, Maeng CH. Optimal time interval from surgery to adjuvant chemotherapy in gastric cancer. Oncol Lett. 2020;20:32. https://doi.org/10.3892/ol.2020.11893.
Wang S, Xu L, Wang Q, Li J, Bai B, Li Z, Wu X, Yu P, Li X, Yin J. Postoperative complications and prognosis after radical gastrectomy for gastric cancer: a systematic review and meta-analysis of observational studies. World J Surg Oncol. 2019;17:52. https://doi.org/10.1186/s12957-019-1593-9.
Chen G, Wang J, Chen K, Kang M, Zhang H, Jin X, Lin L, Chen J. Relationship between postoperative complications and the prognosis of gastric carcinoma patients who underwent surgical resection: a systematic review and meta-analysis. Cancer Control. 2021;28:10732748211011956. https://doi.org/10.1177/10732748211011955.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by [Kadriye Bir Yucel], [Osman Sutcuoglu]. The first draft of the manuscript was written by [Kadriye Bir Yucel], and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The study protocol was reviewed and approved by the joint Ethics Committee of Gazi University (date: April 4, 2022, No: 1997a2f5-d27f-4c57-a48f-a6c408376918).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent to Publish
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yücel, K.B., Sütcüoğlu, O., Yazıcı, O. et al. Retrospective Analysis of Real-Life Data Evaluating the Optimal Time Between Gastrectomy and Adjuvant Chemotherapy in Resected Gastric Cancer. J Gastrointest Canc 54, 1268–1275 (2023). https://doi.org/10.1007/s12029-023-00916-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-023-00916-5