Abstract
Purpose
Many cell signaling pathways essential for normal stem cell development are involved in cancer initiation and progression. In the present study, motivated by a possible contribution of reprogramming process in induction of cancer, we compared the expression level of main genes involved in iPS generation, i.e., miR-302, miR-145, SOX2, c-MYC, and P21, in a series of tumor and non-tumor tissues of stomach.
Methods
A total number of 34 tumors and their matched non-tumor (as control) gastric surgical specimens were obtained. The expression of the candidate genes was evaluated by using real-time PCR and immunohistochemistry (IHC) techniques.
Results
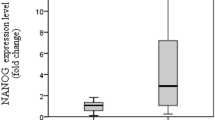
Our data revealed a significant downregulation of miR-302b, P21, and miR-145 genes in intestinal and SOX2 gene in diffuse type of tumor samples. SOX2, but not the other genes, showed a significant downregulation in both proximal (cardia and fundus) and distal (body and antrum) sites of stomach. Based on receiver-operating characteristic (ROC) analyses, the highest total area under the curve (AUC) was found for SOX2 (AUC = 82 %, P < 0.001). Interestingly, all tumor samples revealed a negative signal for c-MYC expression, while non-tumor samples represented an intense cytoplasmic staining.
Conclusions
Despite the fact that some hESC-specific genes are upregulated in tumors, our data revealed a significant downregulation of all candidate genes, except for c-MYC, in tumor samples of stomach. Moreover, ROC data demonstrated that SOX2 gene expression index is a better potential biomarker of gastric cancer, compared to other tested genes. SOX2 expression has a good sensitivity and specificity to discriminate correctly between tumor/non-tumor and also high/low grades of tumor malignancy. It seems downregulation of miR-302b, miR-145, and P21 could contribute to gastric tumor initiation and progression.





Similar content being viewed by others
References
Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12(3):354–62.
Cho RW, Clarke MF. Recent advances in cancer stem cells. Curr Opin Genet Dev. 2008;18(1):48–53.
Ben-David U, Benvenisty N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat Rev Cancer. 2011;11(4):268–77.
Lee AS et al. Tumorigenicity as a clinical hurdle for pluripotent stem cell therapies. Nat Med. 2013;19(8):998–1004.
Chen PS, Su JL, Hung MC. Dysregulation of microRNAs in cancer. J Biomed Sci. 2012;19:90.
Barroso-del Jesus A, Lucena-Aguilar G, Menendez P. The miR-302-367 cluster as a potential stemness regulator in ESCs. Cell Cycle. 2009;8(3):394–8.
Barroso-delJesus A et al. Embryonic stem cell-specific miR302-367 cluster: human gene structure and functional characterization of its core promoter. Mol Cell Biol. 2008;28(21):6609–19.
Card DA et al. Oct4/Sox2-regulated miR-302 targets cyclin D1 in human embryonic stem cells. Mol Cell Biol. 2008;28(20):6426–38.
Xu N et al. MicroRNA-145 regulates OCT4, SOX2, and KLF4 and represses pluripotency in human embryonic stem cells. Cell. 2009;137(4):647–58.
Gao P et al. The molecular mechanism of microRNA-145 to suppress invasion-metastasis cascade in gastric cancer. Oncogene. 2013;32(4):491–501.
Xing AY et al. Deregulated expression of miR-145 in manifold human cancer cells. Exp Mol Pathol. 2013;95(1):91–7.
Sachdeva M et al. p53 represses c-Myc through induction of the tumor suppressor miR-145. Proc Natl Acad Sci U S A. 2009;106(9):3207–12.
Abbas T, Dutta A. p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer. 2009;9(6):400–14.
Hong H et al. Suppression of induced pluripotent stem cell generation by the p53-p21 pathway. Nature. 2009;460(7259):1132–5.
Aloni-Grinstein R, et al. The barrier to cancer stem cell formation. FEBS Lett. 2014; 53.
Kawamura T et al. Linking the p53 tumour suppressor pathway to somatic cell reprogramming. Nature. 2009;460(7259):1140–4.
Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. An Attempt at a Histo-Clinical Classification. Acta Pathol Microbiol Scand. 1965;64:31–49.
Sugano H, Nakamura K, Kato Y. Pathological studies of human gastric cancer. Acta Pathol Jpn. 1982;32 Suppl 2:329–47.
Pfaffl M. Relative quantification. In: Dorak MT, editor. Real-time PCR, vol. 63. New York: Taylor and Francis Group; 2006. p. 63–82.
Li XL et al. Expression of the SRY-related HMG box protein SOX2 in human gastric carcinoma. Int J Oncol. 2004;24(2):257–63.
Otsubo T et al. SOX2 is frequently downregulated in gastric cancers and inhibits cell growth through cell-cycle arrest and apoptosis. Br J Cancer. 2008;98(4):824–31.
Zhang X et al. SOX2 in gastric carcinoma, but not Hath1, is related to patients’ clinicopathological features and prognosis. J Gastrointest Surg. 2010;14(8):1220–6.
Bornschein J et al. Dysregulation of CDX1, CDX2 and SOX2 in patients with gastric cancer also affects the non-malignant mucosa. J Clin Pathol. 2013;66(9):819–22.
Matsuoka J et al. Role of the stemness factors sox2, oct3/4, and nanog in gastric carcinoma. J Surg Res. 2012;174(1):130–5.
Hutz K, et al. The stem cell factor SOX2 regulates the tumorigenic potential in human gastric cancer cells. Carcinogenesis. 2014.
Cai N, Wang YD, Zheng PS. The microRNA-302-367 cluster suppresses the proliferation of cervical carcinoma cells through the novel target AKT1. RNA. 2013;19(1):85–95.
Lin SL, Chang DC, Ying SY, Leu D, Wu DT. MicroRNA miR-302 inhibits the tumorigenecity of human pluripotent stem cells by coordinate suppression of the CDK2 and CDK4/6 cell cycle pathways. Cancer Res. 2010;70(22):9473--82.
Wang L et al. MicroRNA-302b suppresses cell proliferation by targeting EGFR in human hepatocellular carcinoma SMMC-7721 cells. BMC Cancer. 2013;13:448.
Yan GJ et al. MicroRNA miR-302 inhibits the tumorigenicity of endometrial cancer cells by suppression of Cyclin D1 and CDK1. Cancer Lett. 2014;345(1):39–47.
Tabrizi M, Khalili M, Vasei M, Nouraei N, Mansour Samaei N, Khavanin A, Khajehei M, Mowla SJ. Evaluating the miR-302b and miR-145 Expression in Formalin-Fixed Paraffin-Embedded Samples of Esophageal Squamous Cell Carcinoma. Arch Iran Med. 2015 Mar;18(3):173–8.
Zhang L et al. The impact of C-MYC gene expression on gastric cancer cell. Mol Cell Biochem. 2010;344(1–2):125–35.
Geisler JP et al. Nuclear and cytoplasmic c-myc staining in endometrial carcinoma and their relationship to survival. Int J Gynecol Cancer. 2004;14(1):133–7.
Acknowledgments
We are grateful to Dr. Forouzandeh Fereidooni, the previous head of the Iran Tumor Bank, Dr. Zahra Hosseini, Dr. Fatemeh Kamali, and Mr. Ahmad Joulaie for supplying clinical samples and providing patients’ clinicopathological information. All clinical samples were provided by Iran National Tumor Bank (which is funded by the Cancer Institute of Tehran University, for cancer research). We also offer special appreciation to Prof. Reza Malekzadeh (Tehran University of Medical Sciences) for worthwhile advisors and Mrs. Rozita Edalat, Mehdi Parniyan and Vahid Kia (Pasteur Institute of Iran) for providing technical advice concerning Real-Time PCR.
Conflict of Interest
The authors have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
Comparative gene expression in different sites of stomach. Histograms show the median value of relative gene expression in tumor and non-tumor samples in proximal (Cardia and Fudus) and distal (Body and Antrum) sites of stomach. A significant down-regulation in both sites was exclusively observed for SOX2 gene, whereas miR-302b, P21 and miR-145 showed significant differences only in the proximal site of stomach (p-value <0.01 based on Wilcoxon test). * According to Dunn-Bonferroni correction method a threshold value of 0.01 was considered as statistically significant. (JPEG 91.2 kb)
Online Resource 2
Correlation between the expressions of candidate genes. The correlation coefficients of all genes were analyzed by using the Spearman’s rho test. According to this test, miR-302b showed a significant correlation with miR-145. (*) and (**) signs indicate that the correlation is significant at the 0.05 and 0.001 levels (2-tailed) respectively. (GIF 10.2 kb)
Online Resource 3
Immunocytochemistry for SOX2 protein expression in NT2 cell line. Activity of SOX2 primary antibody was confirmed by immunocytochemistry on human embryonic carcinoma cell line NT2, as a positive control. Antibody was detected by using a horseradish peroxidase (HRP) conjugated secondary antibody and diaminobenzidine (DAB) method that produces a brown color. Cells that not probed with primary antibody were considered as negative controls (A). As it is evident in panel B, NT2 showed an intense nuclear signal for SOX2 expression. (GIF 43.6 kb)
Rights and permissions
About this article
Cite this article
Khalili, M., Vasei, M., Khalili, D. et al. Downregulation of the Genes Involved in Reprogramming (SOX2, c-MYC, miR-302, miR-145, and P21) in Gastric Adenocarcinoma. J Gastrointest Canc 46, 251–258 (2015). https://doi.org/10.1007/s12029-015-9695-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-015-9695-2