Abstract
Purpose
In this study, we investigated whether metabolic dysfunction in women with Polycystic ovarian syndrome (PCOS) induces granulosa cell (GC) stress and activates in the endoplamatic reticulum and the mitochondria (UPRer and UPRmt, respectively).
Methods
Women who were diagnosed with PCOS (based on the Rotterdam criteria), were divided into two groups, PCOS with insulin resistance (PCOS-IR; n = 20) and PCOS with no insulin resistance (PCOS-nIR; n = 20), and compared to healthy oocyte donors (CONT; n = 20). Insulin resistance (IR) was assessed on the results of homeostasis model assessment (HOMA) that determines IR using the concentration of fasting plasma glucose and fasting insuline. Expression of UPRer genes (i.e., IRE1, ATF4, ATF6, XBP1, BIP, and CHOP), and UPRmt genes (i.e., HSP60, HSP10, CLPP, and HSP40) was assessed in cumulus GCs by qRT-PCR.
Results
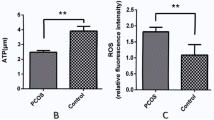
We found that several genes involved in UPRer and UPRmt were overexpressed in the GCs of PCOS-IR and PCOS-nIR compared to CONT. IRE1, ATF4 and XBP1, that are activated by ER stress, were significantly overexpressed in PCOS-IR compared to CONT. BIP and CHOP were overexpressed in PCOS groups compared to CONT. HSP10 and HSP40 were upregulated in PCOS-IR and PCOS-nIR groups compared to the CONT. HSP60 and CLPP showed no statistical different expression in PCOS-IR and PCOS-nIR compared to CONT group.
Conclusion
Our findings suggest that the GCs of women with PCOS (with or without IR) are metabolically distressed and upregulate UPRer and UPRmt genes. Our study contributes to the understanding of the molecular mechanisms underlying the pathological changes that occur in the follicular microenvironment of women with PCOS.



Similar content being viewed by others
Data availability
All the data are included in the paper.
References
M. Cozzolino, E. Seli, Mitochondrial function in women with polycystic ovary syndrome. Curr. Opin. Obstet. Gynecol. 32, 205–12. (2020)
D.A. Dumesic, S.E. Oberfield, E. Stener-Victorin, J.C. Marshall, J.S. Laven, R.S. Legro, Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr. Rev. 36, 487–525 (2015)
J.K. Zawadski, A. Dunaif. Diagnostic Criteria for Polycystic Ovary Syndrome: Towards a Rational Approach. In: Dunaif, A., Givens, J.R. and Haseltine, F., Eds., Polycystic Ovary Syndrome, Blackwell Scientific, Boston, 377–384 (1992)
R. Azziz, E. Carmina, D. Dewailly, E. Diamanti-Kandarakis, H.F. Escobar-Morreale, W. Futterweit et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil. Steril. 91, 456–488 (2009)
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group, Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 81, 19–25 (2004)
F.J. Broekmans, E.A. Knauff, O. Valkenburg, J.S. Laven, M.J. Eijkemans, B.C. Fauser, PCOS according to the Rotterdam consensus criteria: Change in prevalence among WHO-II anovulation and association with metabolic factors. BJOG 113, 1210–1217 (2006)
J. Adams, Z. Liu, Y.A. Ren, W.S. Wun, W. Zhou, S. Kenigsberg et al. Enhanced Inflammatory Transcriptome in the Granulosa Cells of Women With Polycystic Ovarian Syndrome. J. Clin. Endocrinol. Metab. 101, 3459–3468 (2016)
N.B. Karuputhula, R. Chattopadhyay, B. Chakravarty, K. Chaudhury, Oxidative status in granulosa cells of infertile women undergoing IVF. Syst. Biol. Reprod. Med. 59, 91–98 (2013)
J. Schmidt, B. Weijdegard, A.L. Mikkelsen, S. Lindenberg, L. Nilsson, M. Brannstrom, Differential expression of inflammation-related genes in the ovarian stroma and granulosa cells of PCOS women. Mol. Hum. Reprod. 20, 49–58 (2014)
M. Cozzolino, A. Vitagliano, L. Pellegrini, M. Chiurazzi, A. Andriasani, G. Ambrosini, N. Garrido, Therapy with probiotics and synbiotics for polycystic ovarian syndrome: a systematic review and meta-analysis. Eur. J. Nutr. 59(7), 2841–2856 (2020).
J. Kataoka, I. Larsson, S. Bjorkman, B. Eliasson, J. Schmidt, E. Stener-Victorin, Prevalence of polycystic ovary syndrome in women with severe obesity - Effects of a structured weight loss programme. Clin. Endocrinol. (Oxf.) 91, 750–758 (2019)
S. Sam, Obesity and Polycystic Ovary Syndrome. Obes Manag. 3(2), 69–73 (2007)
B.O. Yildiz, E.S. Knochenhauer, R. Azziz, Impact of obesity on the risk for polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 93, 162–168 (2008)
N. Takahashi, M. Harada, Y. Hirota, E. Nose, J.M. Azhary, H. Koike et al. Activation of Endoplasmic Reticulum Stress in Granulosa Cells from Patients with Polycystic Ovary Syndrome Contributes to Ovarian Fibrosis. Sci. Rep. 7(10824), 017–11252-7 (2017)
J.M.K. Azhary, M. Harada, N. Takahashi, E. Nose, C. Kunitomi, H. Koike et al. Endoplasmic Reticulum Stress Activated by Androgen Enhances Apoptosis of Granulosa Cells via Induction of Death Receptor 5 in PCOS. Endocrinology 160, 119–132 (2019)
P. Walter, D. Ron, The unfolded protein response: from stress pathway to homeostatic regulation. Science 334, 1081–1086 (2011)
S.Z. Hasnain, R. Lourie, I. Das, A.C. Chen, M.A. McGuckin, The interplay between endoplasmic reticulum stress and inflammation. Immunol. Cell Biol. 90, 260–70. (2012)
J. Grootjans, A. Kaser, R.J. Kaufman, R.S. Blumberg, The unfolded protein response in immunity and inflammation. Nat. Rev. Immunol. 16, 469–484 (2016)
D.T. Rutkowski, R.J. Kaufman, A trip to the ER: coping with stress. Trends Cell Biol. 14, 20–28 (2004)
D. Ron, P. Walter, Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 8, 519–529 (2007)
M. Schroder, R.J. Kaufman, The mammalian unfolded protein response. Annu Rev. Biochem 74, 739–789 (2005)
C. Xu, B. Bailly-Maitre, J.C. Reed, Endoplasmic reticulum stress: cell life and death decisions. J. Clin. Investig. 115, 2656–2664 (2005)
Y.C. Chae, A. Angelin, S. Lisanti, A.V. Kossenkov, K.D. Speicher, H. Wang et al. Landscape of the mitochondrial Hsp90 metabolome in tumours. Nat. Commun. 4, 2139 (2013)
J.E. Aldridge, T. Horibe, N.J. Hoogenraad, Discovery of genes activated by the mitochondrial unfolded protein response (mtUPR) and cognate promoter elements. PLoS ONE 2, e874 (2007)
A.J. Caplan, A.K. Mandal, M.A. Theodoraki, Molecular chaperones and protein kinase quality control. Trends Cell Biol. 17, 87–92 (2007)
T. Wang, E. Babayev, Z. Jiang, G. Li, M. Zhang, E. Esencan, et al. Mitochondrial unfolded protein response gene Clpp is required to maintain ovarian follicular reserve during aging, for oocyte competence, and development of pre-implantation embryos. Aging Cell. 17(4), e12784 (2018)
Z. Huang, D. Wells, The human oocyte and cumulus cells relationship: new insights from the cumulus cell transcriptome. Mol. Hum. Reprod. 16, 715–725 (2010)
A. Uyar, S. Torrealday, E. Seli, Cumulus and granulosa cell markers of oocyte and embryo quality. Fertil. Steril. 99, 979–97. (2013)
M. Harada, E. Nose, N. Takahashi, Y. Hirota, T. Hirata, O. Yoshino et al. Evidence of the activation of unfolded protein response in granulosa and cumulus cells during follicular growth and maturation. Gynecol. Endocrinol. 31, 783–787 (2015)
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group, Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 81, 19–25 (2004)
D.R. Matthews, J.P. Hosker, A.S. Rudenski, B.A. Naylor, D.F. Treacher, R.C. Turner, Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28, 412–419 (1985)
M. Cozzolino, M. Cruz, A. Patel, J. Patel, A. Pacheco, J.A. Garcia-Velasco, Serum and follicular fluid Stem Cell Factor assay in IVF poor responder and normal responder patients: a predictive biomarker of oocyte retrieval. Arch. Gynecol. Obstet. 300, 447–54. (2019)
R. Azziz, How polycystic ovary syndrome came into its own. F. S Sci. 2, 2–10 (2021)
L. Harborne, R. Fleming, H. Lyall, J. Norman, N. Sattar, Descriptive review of the evidence for the use of metformin in polycystic ovary syndrome. Lancet 361, 1894–1901 (2003)
A.S. Lee, Glucose-regulated proteins in cancer: molecular mechanisms and therapeutic potential. Nat. Rev. Cancer 14(4), 263–76. (2004)
P. Walter, D. Ron, The unfolded protein response: from stress pathway to homeostatic regulation. Science 334, 1081–1086 (2011)
C.M. Haynes, K. Petrova, C. Benedetti, Y. Yang, D. Ron, ClpP mediates activation of a mitochondrial unfolded protein response in C. elegans. Dev. Cell 13, 467–480 (2007)
T. Nakagawa, H. Zhu, N. Morishima, E. Li, J. Xu, B.A. Yankner et al. Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-beta. Nature 403, 98–103 (2000)
J. Jin, Y. Ma, X. Tong, W. Yang, Y. Dai, Y. Pan et al. Metformin inhibits testosterone-induced endoplasmic reticulum stress in ovarian granulosa cells via inactivation of p38 MAPK. Hum. Reprod. 35, 1145–58. (2020)
M. Harada, E. Nose, N. Takahashi, Y. Hirota, T. Hirata, O. Yoshino et al. Evidence of the activation of unfolded protein response in granulosa and cumulus cells during follicular growth and maturation. Gynecol. Endocrinol. 31, 783–787 (2015)
H.J. Park, J.Y. Park, J.W. Kim, S.G. Yang, J.M. Jung, M.J. Kim, et al. Melatonin improves the meiotic maturation of porcine oocytes by reducing endoplasmic reticulum stress during in vitro maturation. J. Pineal. Res. 2018;64:https://doi.org/10.1111/jpi.12458. Epub 2017 Dec 4.
L.L. Wu, D.L. Russell, R.J. Norman, R.L. Robker, Endoplasmic reticulum (ER) stress in cumulus-oocyte complexes impairs pentraxin-3 secretion, mitochondrial membrane potential (DeltaPsi m), and embryo development. Mol. Endocrinol. 26, 562–573 (2012)
L.M. Cree, E.R. Hammond, A.N. Shelling, M.C. Berg, J.C. Peek, M.P. Green, Maternal age and ovarian stimulation independently affect oocyte mtDNA copy number and cumulus cell gene expression in bovine clones. Hum. Reprod. 30, 1410–1420 (2015)
J. Liu, L.F. Luo, D.L. Wang, W.X. Wang, J.L. Zhu, Y.C. Li et al. Cadmium induces ovarian granulosa cell damage by activating PERK-eIF2alpha-ATF4 through endoplasmic reticulum stress. Biol. Reprod. 100, 292–299 (2019)
K. Vasickova, L. Moran, D. Gurin, P. Vanhara, Alleviation of endoplasmic reticulum stress by tauroursodeoxycholic acid delays senescence of mouse ovarian surface epithelium. Cell Tissue Res. 374, 643–52. (2018)
J. Lee, U. Ozcan, Unfolded protein response signaling and metabolic diseases. J. Biol. Chem. 289, 1203–1211 (2014)
S. Brenjian, A. Moini, N. Yamini, L. Kashani, M. Faridmojtahedi, M. Bahramrezaie et al. Resveratrol treatment in patients with polycystic ovary syndrome decreased pro-inflammatory and endoplasmic reticulum stress markers. Am. J. Reprod. Immunol. 83, e13186 (2020)
D. Lanneau, M. Brunet, E. Frisan, E. Solary, M. Fontenay, C. Garrido, Heat shock proteins: essential proteins for apoptosis regulation. J. Cell Mol. Med. 12, 743–761 (2008)
L. Wang, J. Tang, L. Wang, F. Tan, H. Song, J. Zhou et al. Oxidative stress in oocyte aging and female reproduction. J. Cell Physiol. 236, 7966–83. (2021)
E. Panagodimou, V. Koika, F. Markatos, A. Kaponis, G. Adonakis, N.A. Georgopoulos et al. Expression stability of ACTB, 18S, and GAPDH in human placental tissues from subjects with PCOS and controls: GAPDH expression is increased in PCOS. Hormones (Athens) 21, 329–33. (2022)
M. Cozzolino, S. Herraiz, S. Titus, L. Roberts, M. Romeu, I. Peinado, R.T. Scott, A. Pellicer, E. Seli, Transcriptomic landscape of granulosa cells and peripheral blood mononuclear cells in women with PCOS compared to young poor responders and women with normal response. Hum. Reprod. 37(6), 1274–1286 (2022)
Acknowledgements
The authors thank the participants of the study for making this work possible, as well as the medical staff that participated in sample collection and processing.
Author information
Authors and Affiliations
Contributions
M.C. take primary responsibility for the paper, collected data, made statistical analysis, and write the paper; S.H. edited the final version of the paper; Y.C., J.A.G.V., B.T., A.P., S.R., G.K., AB, recruited patients and collected data and E.S. reviewed the final version of the paper.
Funding
This study was supported by a grant from Foundation for Embryonic Competence to E.S.
Corresponding author
Ethics declarations
Conflict of interest
M.C., S.H., Y.C., J.A.G.V., B.T., A.P., S.R., G.K. A.B., A.P. declares no competing interests concerning this research. E.S. is a consultant for and receives research funding from the Foundation for Embryonic Competence.
Consent for publication
All patients were informed and gave written consent.
Ethical approval and consent to participate
All the procedures per- formed in the studies that involved human participants were made under the ethical standards of the Institutional and National Research Committee, and also with the 1964 Declaration of Helsinki as well as its later amendments, or comparable ethical standards. All study procedures were approved and conducted according to the Institutional Review Board of Hospital Universitario Puerta de Hierro, Madrid, Spain (1812-MAD-099-MC). Patients signed an informed consense.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cozzolino, M., Herraiz, S., Cakiroglu, Y. et al. Distress response in granulosa cells of women affected by PCOS with or without insulin resistance. Endocrine 79, 200–207 (2023). https://doi.org/10.1007/s12020-022-03192-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03192-8