Abstract
Purpose
Recent guidelines on adrenal incidentalomas suggested in patients with an indeterminate adrenal mass and no significant hormone excess that follow up with a repeat noncontrast CT or MRI after 6–12 months may be an option.
Methods
We report the case of a 32-year-old woman who presented with a 2.9 × 1.9 cm left adrenal incidentaloma that was stable in size for 4 years. Ten years later the left adrenal mass was a stage IV adrenocortical carcinoma (ACC).
Results
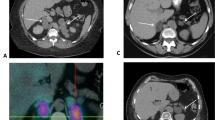
In 2006, a 32-year-old French Canadian woman was referred to endocrinology for a left 2.9 × 1.9 cm incidentally discovered adrenal mass (31 HU). She had normal hormonal investigation. The patient was followed with adrenal imaging and hormonal investigation yearly for 4 years and the lesion stayed stable in size over the 4 years. Ten years later, in 2016, the patient presented with renal colic. Urological CT unexpectedly revealed that the left adrenal mass was now measuring 9 × 8.2 cm and 2 new hepatic lesions were found. Biochemical workup demonstrated hypercorticism and hyperandrogenemia: plasma cortisol after 1 mg overnight DST of 476 nmol/L and DHEA-S of 14.0 μmol/L (N 0.9–6.5). Twenty-four hour urine steroid profiling was consistent with an adrenocortical carcinoma (ACC) co-secreting cortisol, androgens and glucocorticoid precursors. The diagnosis of ACC with hepatic ACC metastases was confirmed at histology. Following genetic analysis, germline heterozygous variant of uncertain significance (VUS) was identified in the exon 16 of the APC gene (c.2414G > A, p.Arg805Gln). Immunohistochemical staining’s of the ACC was positive for IGF-2 and cytoplasmic/nuclear β-catenin staining.
Conclusions
This case illustrates that (1) small adrenal incidentaloma stable in size may evolve to ACC and (2) better genetic characterization of these patients may eventually give clues on this unusual evolution.



Similar content being viewed by others
Abbreviations
- ACC:
-
adrenocortical carcinoma
- DST:
-
dexamethasone suppression test
- HBP:
-
high blood pressure
- CT:
-
computed tomography
- HU:
-
Hounsfield unit
- LCMS:
-
liquid chromatography and mass spectrometry
- MRI:
-
magnetic resonance imaging
- PET:
-
positron emission tomography
- N:
-
normal
References
M. Fassnacht, W. Arlt, I. Bancos, H. Dralle, J. Newell-Price, A. Sahdev, A. Tabarin, M. Terzolo, S. Tsagarakis, O.M. Dekkers, Management of adrenal incidentalomas: European Society of Endocrinology Clinical Practice Guideline in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 175(2), G1–G34 (2016). https://doi.org/10.1530/EJE-16-0467
J.M. Hines, I. Bancos, C. Bancos, R.D. Singh, A.V. Avula, W.F. Young, S.K. Grebe, R.J. Singh, High-resolution, accurate-mass (HRAM) mass spectrometry urine steroid profiling in the diagnosis of adrenal disorders. Clin. Chem. 63(12), 1824–1835 (2017). https://doi.org/10.1373/clinchem.2017.271106
J.M. Schwarz, D.N. Cooper, M. Schuelke, D. Seelow, MutationTaster2: mutation prediction for the deep-sequencing age. Nat. Methods 11(4), 361–362 (2014). https://doi.org/10.1038/nmeth.2890
D. Salgado, J.P. Desvignes, G. Rai, A. Blanchard, M. Miltgen, A. Pinard, N. Levy, G. Collod-Beroud, C. Beroud, UMD-predictor: a high-throughput sequencing compliant system for pathogenicity prediction of any human cDNA substitution. Hum. Mutat. 37(5), 439–446 (2016). https://doi.org/10.1002/humu.22965
J. Bendl, J. Stourac, O. Salanda, A. Pavelka, E.D. Wieben, J. Zendulka, J. Brezovsky, J. Damborsky, PredictSNP: robust and accurate consensus classifier for prediction of disease-related mutations. PLoS Comput. Biol. 10(1), e1003440 (2014). https://doi.org/10.1371/journal.pcbi.1003440
Y. Choi, A.P. Chan, PROVEAN web server: a tool to predict the functional effect of amino acid substitutions and indels. Bioinformatics 31(16), 2745–2747 (2015). https://doi.org/10.1093/bioinformatics/btv195
P.Y. Cheah, Y.H. Wong, P.K. Koh, C. Loi, M.H. Chew, C.L. Tang, A novel indel in exon 9 of APC upregulates a ‘skip exon 9’ isoform and causes very severe familial adenomatous polyposis. Eur. J. Hum. Genet. 22(6), 833–836 (2014). https://doi.org/10.1038/ejhg.2013.245
R.B. van der Luijt, H.F.A. Vasen, C.M.J. Tops, C. Breukel, R. Fodde, P.M. Khan, APC mutation in the alternatively spliced region of exon 9 associated with late onset familial adenomatous polyposis. Hum. Genet. 96, 705–710 (1995)
M.C. Curia, D.L. Esposito, G. Aceto, R. Palmirotta, S. Crognale, R. Valanzano, F. Ficari, F. Tonelli, P. Battista, R. Mariani-Costantini, A. Cama, Transcript dosage effect in familial adenomatous polyposis: model offered by two kindreds with Exon 9 APC gene mutations. Hum. Mutat. 11, 197–201 (1998)
T.M. Nogueira, R. Lirov, E.M. Caoili, A.M. Lerario, B.S. Miller, M.C. Fragoso, N.R. Dunnick, G.D. Hammer, T. Else, Radiographic characteristics of adrenal masses preceding the diagnosis of adrenocortical. Cancer Horm. Cancer 6(4), 176–181 (2015). https://doi.org/10.1007/s12672-015-0225-2
L. Ozsari, M. Kutahyalioglu, K.M. Elsayes, R.A. Vicens, K. Sircar, T. Jazaerly, S.G. Waguespack, N.L. Busaidy, M.E. Cabanillas, R. Dadu, M.I. Hu, R. Vassilopoulou-Sellin, C. Jimenez, J.E. Lee, M.A. Habra, Preexisting adrenal masses in patients with adrenocortical carcinoma: clinical and radiological factors contributing to delayed diagnosis. Endocrine 51(2), 351–359 (2016). https://doi.org/10.1007/s12020-015-0694-7
W. Arlt, M. Biehl, A.E. Taylor, S. Hahner, R. Libe, B.A. Hughes, P. Schneider, D.J. Smith, H. Stiekema, N. Krone, E. Porfiri, G. Opocher, J. Bertherat, F. Mantero, B. Allolio, M. Terzolo, P. Nightingale, C.H.L. Shackleton, X. Bertagna, M. Fassnacht, P.M. Stewart, Urine steroid metabolomics as a biomarker tool for detecting malignancy in adrenal tumors. JCEM 96(12), 3775–3784 (2011). https://doi.org/10.1210/jc.2011-1565
L.I. Velikanova, Z.R. Shafigullina, A.A. Lisitsin, N.V. Vorokhobina, K. Grigoryan, E.A. Kukhianidze, E.G. Strelnikova, N.S. Krivokhizhina, L.M. Krasnov, E.A. Fedorov, I.V. Sablin, A.L. Moskvin, E.A. Bessonova, Different types of urinary steroid profiling obtained by high-performance liquid chromatography and gas chromatography-mass spectrometry in patients with adrenocortical carcinoma. Horm. Cancer 7, 327–335 (2016). https://doi.org/10.1007/s12672-016-0267-0
S. Schweitzer, M. Kunz, M. Kurlbaum, M. Kroiss, J. Vey, S. Kendl, T. Deutschbein, S. Hahner, M. Fassnacht, T. Dandekar, Plasma steroid metabolome profiling for the diagnosis of adrenocortical carcinoma. Eur. J. Endocrinol. 180(2), 117–125 (2019)
S. Petersenn, P.A. Richter, T. Broemel, C.O. Ritter, T. Deutschbein, F.U. Beil, B. Allolio, M. Fassnacht, German ACC Study Group, Computed tomography criteria for discrimination of adrenal adenomas and adrenocortical carcinomas: analysis of the German ACC registry. Eur. J. Endocrinol. 172(4), 415–422 (2015). https://doi.org/10.1530/EJE-14-0916
J. Dinnes, I. Bancos, L. Ferrante di Ruffano, V. Chortis, C. Davenport, S. Bayliss, A. Sahdev, P. Guest, M. Fassnacht, J.J. Deeks, W. Arlt, Management of endocrine disease: imaging for the diagnosis of malignancy in incidentally discovered adrenal masses: a systematic review and meta-analysis. Eur. J. Endocrinol. 175(2), R51–R64 (2016). https://doi.org/10.1530/EJE-16-0461
I. Belmihoub, S. Silvera, M. Sibony, B. Dousset, P. Legmann, X. Bertagna, J. Bertherat, G. Assie, From benign adrenal incidentaloma to adrenocortical carcinoma: an exceptional random event. Eur. J. Endocrinol. 176(6), K15–K19 (2017). https://doi.org/10.1530/EJE-17-0037
S. Gaujoux, S. Pinson, A.P. Gimenez-Roqueplo, L. Amar, B. Ragazzon, P. Launay, T. Meatchi, R. Libe, X. Bertagna, A. Audebourg, J. Zucman-Rossi, F. Tissier, J. Bertherat, Inactivation of the APC gene is constant in adrenocortical tumors from patients with familial adenomatous polyposis but not frequent in sporadic adrenocortical cancers. Clin. Cancer Res. 16(21), 5133–5141 (2010). https://doi.org/10.1158/1078-0432.CCR-10-1497
G. Assie, E. Letouzé, M. Fassnacht, A. Jouinot, W. Luscap, O. Barreau, H. Omeiri, S. Rodriguez, K. Perlemoine, F. René-Corail, N. Elarouci, S. Sbiera, M. Kroiss, B. Allolio, J. Waldmann, M. Quinkler, M. Mannelli, F. Mantero, T. Papathomas, R. De Krijger, A. Tabarin, V. Kerlan, E. Baudin, F. Tissier, B. Dousset, L. Groussin, L. Amar, E. Clauser, X. Bertagna, B. Ragazzon, F. Beuschlein, R. Libe, A. Reyniès de, J. Bertherat, Integrated genomic characterization of adrenocortical carcinoma. Nat. Genet. 46(6), 607–612 (2014). https://doi.org/10.1038/ng.2953
J.S. Shiroky, J.P. Lerner-Ellis, A. Govindarajan, D.R. Urbach, K.M. Devon, Characteristics of adrenal masses in familial adenomatous polyposis. Dis. Colon Rectum 61, 679–685 (2018). https://doi.org/10.1097/DCR.0000000000001008
S. Gaujoux, S. Grabar, M. Fassnacht, B. Ragazzon, P. Launay, R. Libe, I. Chokri, A. Audebourg, B. Royer, S. Sbiera, M.C. Vacher-Lavenu, B. Dousset, X. Bertagna, B. Allolio, J. Bertherat, F. Tissier, Beta-catenin activation is associated with specific clinical and pathologic characteristics and a poor outcome in adrenocortical carcinoma. Clin. Cancer Res. 17(2), 328–336 (2011). https://doi.org/10.1158/1078-0432.CCR-10-2006
R. Maharjan, S. Backman, T. Åkerström, P. Hellman, P. Björklund, Comprehensive analysis of CTNNB1 in adrenocortical carcinomas: Identi cation of novel mutations and correlation to survival. Sci. Rep. 8, 1–10 (2018). https://doi.org/10.1038/s41598-018-26799-2
Funding
This research was supported in part by a salary grant to I. Bourdeau from Fonds de Recherche du Québec-Santé (FRQ-S) and the generous financial support from Fondation McAbbie for the laboratory work.
Author information
Authors and Affiliations
Contributions
We thank the patient who kindly consents that we report her case. We thank Dr. Serge Nolet for technical assistance for leukocyte DNA. N.G. and I. Bourdeau contributed to literature search and writing of the manuscript. I. Bourdeau designed the study. I. Bancos provided the metabolomic profile. G.C. and K.C. contributed to technical laboratory work. I.B and all other co-authors contributed towards patient care and finalizing the draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The authors confirm that written informed consent was obtained from the patient for publication of the submitted article and the accompanying images.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gagnon, N., Boily, P., Alguire, C. et al. Small adrenal incidentaloma becoming an aggressive adrenocortical carcinoma in a patient carrying a germline APC variant. Endocrine 68, 203–209 (2020). https://doi.org/10.1007/s12020-020-02209-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02209-4