Abstract
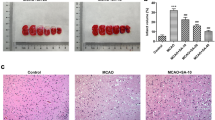
Previous studies have demonstrated that increased O-linked N-acetylglucosamine (O-GlcNAc) level could promote cell survival following environmental stresses. This study aimed to explore the role of O-GlcNAc transferase (OGT) during cerebral ischemia/reperfusion (I/R) injury. The mouse model with cerebral I/R injury was induced by middle cerebral artery occlusion/reperfusion (MCAO/R). The expression of ogt in brain tissues was detected by qRT-PCR, Western blot, and immunohistochemistry (IHC) staining assay. Neurological deficit was evaluated using a modified scoring system. The infarct volume was assessed by TTC staining assay. Neuronal apoptosis in brain tissues was evaluated by TUNEL staining assay. The level of cleaved caspase-3 in brain tissues was detected by Western blot and IHC staining assay. The expression of critical proteins involved in mitochondrial fission, including OPA1, Mfn1, and Mfn2, as well as Mff and Drp1 was detected by Western blot and IHC, respectively. The expression of ogt during cerebral I/R injury was significantly upregulated. Ogt knockdown significantly increased neurological score and infarct volume in I/R-induced mice. Meanwhile, ogt knockdown significantly enhanced neuronal apoptosis and cleaved caspase-3 level in brain tissues of I/R-induced mice. In addition, ogt knockdown markedly decreased serine 637 phosphorylation level of mitochondrial fission protein dynamin-related protein 1 (Drp1) and promoted Drp1 translocation from the cytosol to the mitochondria. Moreover, the specific Drp1 inhibitor mdivi-1 effectively attenuated ogt knockdown-induced brain injury of I/R-stimulated mice in vivo. Our study revealed that OGT protects against cerebral I/R injury by inhibiting the function of Drp1 in mice, suggesting that ogt may be a potential therapeutic target for cerebral I/R injury.







Similar content being viewed by others
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Aquino-Gil, M., Pierce, A., Perez-Cervera, Y., Zenteno, E., & Lefebvre, T. (2017). OGT: A short overview of an enzyme standing out from usual glycosyltransferases. Biochemical Society Transactions, 45(2), 365–370. https://doi.org/10.1042/bst20160404
Benakis, C., Brea, D., Caballero, S., Faraco, G., Moore, J., Murphy, M., et al. (2016). Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ T cells. Nature Medicine, 22(5), 516–523. https://doi.org/10.1038/nm.4068
Bertholet, A. M., Delerue, T., Millet, A. M., Moulis, M. F., David, C., Daloyau, M., et al. (2016). Mitochondrial fusion/fission dynamics in neurodegeneration and neuronal plasticity. Neurobiology of Diseases, 90, 3–19. https://doi.org/10.1016/j.nbd.2015.10.011
Brentnall, M., Rodriguez-Menocal, L., De Guevara, R. L., Cepero, E., & Boise, L. H. (2013). Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biology, 14, 32. https://doi.org/10.1186/1471-2121-14-32
Cereghetti, G. M., Stangherlin, A., Martins de Brito, O., Chang, C. R., Blackstone, C., Bernardi, P., et al. (2008). Dephosphorylation by calcineurin regulates translocation of Drp1 to mitochondria. Proceedings of the National Academy of Sciences United States of America, 105(41), 15803–15808. https://doi.org/10.1073/pnas.0808249105
Champattanachai, V., Marchase, R. B., & Chatham, J. C. (2007). Glucosamine protects neonatal cardiomyocytes from ischemia-reperfusion injury via increased protein-associated O-GlcNAc. American Journal of Physiology, Cell Physiology, 292(1), C178-187. https://doi.org/10.1152/ajpcell.00162.2006
Champattanachai, V., Marchase, R. B., & Chatham, J. C. (2008). Glucosamine protects neonatal cardiomyocytes from ischemia-reperfusion injury via increased protein O-GlcNAc and increased mitochondrial Bcl-2. American Journal of Physiology, Cell Physiology, 294(6), C1509-1520. https://doi.org/10.1152/ajpcell.00456.2007
Chatila, T., Anderson, K. A., Ho, N., & Means, A. R. (1996). A unique phosphorylation-dependent mechanism for the activation of Ca2+/calmodulin-dependent protein kinase type IV/GR. Journal of Biological Chemistry, 271(35), 21542–21548. https://doi.org/10.1074/jbc.271.35.21542
Chen, H., & Chan, D. C. (2004). Mitochondrial dynamics in mammals. Current Topics in Developmental Biology, 59, 119–144. https://doi.org/10.1016/s0070-2153(04)59005-1
Cheng, C. Y., Lin, J. G., Tang, N. Y., Kao, S. T., & Hsieh, C. L. (2014). Electroacupuncture-like stimulation at the Baihui (GV20) and Dazhui (GV14) acupoints protects rats against subacute-phase cerebral ischemia-reperfusion injuries by reducing S100B-mediated neurotoxicity. PLoS ONE, 9(3), e91426. https://doi.org/10.1371/journal.pone.0091426
Cheng, X., Hou, Z., Sun, J., Huang, Y., Wang, L., Zhou, Z., et al. (2017). Protective effects of Tongxinluo on cerebral ischemia/reperfusion injury related to Connexin 43/Calpain II/Bax/Caspase-3 pathway in rat. Journal of Ethnopharmacology, 198, 148–157. https://doi.org/10.1016/j.jep.2017.01.004
Choudhury, G. R., & Ding, S. (2016). Reactive astrocytes and therapeutic potential in focal ischemic stroke. Neurobiology of Diseases, 85, 234–244. https://doi.org/10.1016/j.nbd.2015.05.003
Dias, W. B., Cheung, W. D., Wang, Z., & Hart, G. W. (2009). Regulation of calcium/calmodulin-dependent kinase IV by O-GlcNAc modification. Journal of Biological Chemistry, 284(32), 21327–21337. https://doi.org/10.1074/jbc.M109.007310
Duan, J., Cui, J., Zheng, H., Xi, M., & Guo, C. (2019). Aralia taibaiensis protects against I/R-induced brain cell injury through the Akt/SIRT1/FOXO3a pathway. Oxidative Medicine and Cellular Longevity, 2019, 7609765. https://doi.org/10.1155/2019/7609765
Fardini, Y., Dehennaut, V., Lefebvre, T., & Issad, T. (2013). O-GlcNAcylation: A new cancer hallmark? Frontiers in Endocrinology (lausanne), 4, 99. https://doi.org/10.3389/fendo.2013.00099
Feng, J., Chen, X., Guan, B., Li, C., Qiu, J., & Shen, J. (2018). Inhibition of peroxynitrite-induced mitophagy activation attenuates cerebral ischemia-reperfusion injury. Molecular Neurobiology, 55(8), 6369–6386. https://doi.org/10.1007/s12035-017-0859-x
Galkin, A. (2019). Brain ischemia/reperfusion injury and mitochondrial complex I damage. Biochemistry (moscow), 84(11), 1411–1423. https://doi.org/10.1134/s0006297919110154
Han, S. J., Jang, H. S., Noh, M. R., Kim, J., Kong, M. J., Kim, J. I., et al. (2017). Mitochondrial NADP(+)-dependent isocitrate dehydrogenase deficiency exacerbates mitochondrial and cell damage after kidney ischemia-reperfusion injury. Journal of the American Society of Nephrology, 28(4), 1200–1215. https://doi.org/10.1681/asn.2016030349
Hu, X., De Silva, T. M., Chen, J., & Faraci, F. M. (2017). Cerebral vascular disease and neurovascular injury in ischemic stroke. Circulation Research, 120(3), 449–471. https://doi.org/10.1161/circresaha.116.308427
Itkonen, H. M., Urbanucci, A., Martin, S. E., Khan, A., Mathelier, A., Thiede, B., et al. (2019). High OGT activity is essential for MYC-driven proliferation of prostate cancer cells. Theranostics, 9(8), 2183–2197. https://doi.org/10.7150/thno.30834
Ji, K., Xue, L., Cheng, J., & Bai, Y. (2016). Preconditioning of H2S inhalation protects against cerebral ischemia/reperfusion injury by induction of HSP70 through PI3K/Akt/Nrf2 pathway. Brain Research Bulletin, 121, 68–74. https://doi.org/10.1016/j.brainresbull.2015.12.007
Kalogeris, T., Baines, C. P., Krenz, M., & Korthuis, R. J. (2016). Ischemia/reperfusion. Comprehensive Physiology, 7(1), 113–170. https://doi.org/10.1002/cphy.c160006
Le, D. A., Wu, Y., Huang, Z., Matsushita, K., Plesnila, N., Augustinack, J. C., et al. (2002). Caspase activation and neuroprotection in caspase-3- deficient mice after in vivo cerebral ischemia and in vitro oxygen glucose deprivation. Proceedings of the National Academy of Sciences United States of America, 99(23), 15188–15193. https://doi.org/10.1073/pnas.232473399
Leech, T., Chattipakorn, N., & Chattipakorn, S. C. (2019). The beneficial roles of metformin on the brain with cerebral ischaemia/reperfusion injury. Pharmacological Research, 146, 104261. https://doi.org/10.1016/j.phrs.2019.104261
Li, X., Gong, W., Wang, H., Li, T., Attri, K. S., Lewis, R. E., et al. (2019). O-GlcNAc transferase suppresses inflammation and necroptosis by targeting receptor-interacting serine/threonine-protein kinase 3. Immunity, 50(3), 576-590.e576. https://doi.org/10.1016/j.immuni.2019.01.007
Li, Y., Wang, M., & Wang, S. (2016). Effect of inhibiting mitochondrial fission on energy metabolism in rat hippocampal neurons during ischemia/reperfusion injury. Neurological Research, 38(11), 1027–1034. https://doi.org/10.1080/01616412.2016.1215050
Liu, L., Chen, H., Jin, J., Tang, Z., Yin, P., Zhong, D., et al. (2019a). Melatonin ameliorates cerebral ischemia/reperfusion injury through SIRT3 activation. Life Sciences, 239, 117036. https://doi.org/10.1016/j.lfs.2019.117036
Liu, Z., Li, H., Su, J., Xu, S., Zhu, F., Ai, J., et al. (2019b). Numb depletion promotes Drp1-mediated mitochondrial fission and exacerbates mitochondrial fragmentation and dysfunction in acute kidney injury. Antioxidants & Redox Signaling, 30(15), 1797–1816. https://doi.org/10.1089/ars.2017.7432
Longa, E. Z., Weinstein, P. R., Carlson, S., & Cummins, R. (1989). Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke, 20(1), 84–91. https://doi.org/10.1161/01.str.20.1.84
Phipps, M. S., & Cronin, C. A. (2020). Management of acute ischemic stroke. BMJ, 368, l6983. https://doi.org/10.1136/bmj.l6983
Prabhakaran, S., Ruff, I., & Bernstein, R. A. (2015). Acute stroke intervention: A systematic review. JAMA, 313(14), 1451–1462. https://doi.org/10.1001/jama.2015.3058
Sacoman, J. L., Dagda, R. Y., Burnham-Marusich, A. R., Dagda, R. K., & Berninsone, P. M. (2017). Mitochondrial O-GlcNAc transferase (mOGT) regulates mitochondrial structure, function, and survival in HeLa cells. Journal of Biological Chemistry, 292(11), 4499–4518. https://doi.org/10.1074/jbc.M116.726752
Simon, D. N., Wriston, A., & Fan, Q. (2018). OGT (O-GlcNAc Transferase) selectively modifies multiple residues unique to lamin A. Cells, 7(5), 44. https://doi.org/10.3390/cells7050044
Snow, S. J. (2016). Stroke and t-PA–triggering new paradigms of care. The New England Journal of Medicine, 374(9), 809–811. https://doi.org/10.1056/NEJMp1514696
Sodi, V. L., Khaku, S., Krutilina, R., Schwab, L. P., Vocadlo, D. J., Seagroves, T. N., et al. (2015). mTOR/MYC axis regulates O-GlcNAc transferase expression and O-GlcNAcylation in breast cancer. Molecular Cancer Research, 13(5), 923–933. https://doi.org/10.1158/1541-7786.mcr-14-0536
Tokumitsu, H., & Soderling, T. R. (1996). Requirements for calcium and calmodulin in the calmodulin kinase activation cascade (*). Journal of Biological Chemistry, 271(10), 5617–5622. https://doi.org/10.1074/jbc.271.10.5617
Wang, L., Chen, S., Zhang, Z., Zhang, J., Mao, S., Zheng, J., et al. (2018). Suppressed OGT expression inhibits cell proliferation while inducing cell apoptosis in bladder cancer. BMC Cancer, 18(1), 1141. https://doi.org/10.1186/s12885-018-5033-y
Xu, D., Wang, W., Bian, T., Yang, W., Shao, M., & Yang, H. (2019). Increased expression of O-GlcNAc transferase (OGT) is a biomarker for poor prognosis and allows tumorigenesis and invasion in colon cancer. International Journal of Clinical and Experimental Pathology, 12(4), 1305–1314.
Xu, W., Zhang, X., Wu, J. L., Fu, L., Liu, K., Liu, D., et al. (2017). O-GlcNAc transferase promotes fatty liver-associated liver cancer through inducing palmitic acid and activating endoplasmic reticulum stress. Journal of Hepatology, 67(2), 310–320. https://doi.org/10.1016/j.jhep.2017.03.017
Zachara, N. E., O’Donnell, N., Cheung, W. D., Mercer, J. J., Marth, J. D., & Hart, G. W. (2004). Dynamic O-GlcNAc modification of nucleocytoplasmic proteins in response to stress. A survival response of mammalian cells. Journal of Biological Chemistry, 279(29), 30133–30142. https://doi.org/10.1074/jbc.M403773200
Zhang, B., Li, M. D., Yin, R., Liu, Y., Yang, Y., Mitchell-Richards, K. A., et al. (2019a). O-GlcNAc transferase suppresses necroptosis and liver fibrosis. JCI Insight. https://doi.org/10.1172/jci.insight.127709
Zhang, H., Zhong, K., Lu, M., Mei, Y., Tan, E., Sun, X., et al. (2018). Neuroprotective effects of isosteviol sodium through increasing CYLD by the downregulation of miRNA-181b. Brain Research Bulletin, 140, 392–401. https://doi.org/10.1016/j.brainresbull.2018.05.015
Zhang, N., Wang, S., Li, Y., Che, L., & Zhao, Q. (2013). A selective inhibitor of Drp1, mdivi-1, acts against cerebral ischemia/reperfusion injury via an anti-apoptotic pathway in rats. Neuroscience Letters, 535, 104–109. https://doi.org/10.1016/j.neulet.2012.12.049
Zhang, Z., Parker, M. P., Graw, S., Novikova, L. V., Fedosyuk, H., Fontes, J. D., et al. (2019b). O-GlcNAc homeostasis contributes to cell fate decisions during hematopoiesis. Journal of Biological Chemistry, 294(4), 1363–1379. https://doi.org/10.1074/jbc.RA118.005993
Zhou, F., Yang, X., Zhao, H., Liu, Y., Feng, Y., An, R., et al. (2018). Down-regulation of OGT promotes cisplatin resistance by inducing autophagy in ovarian cancer. Theranostics, 8(19), 5200–5212. https://doi.org/10.7150/thno.27806
Acknowledgements
The authors would like to express our gratitude for those who have critically reviewed this manuscript and those who give us help during the experiments.
Funding
No funders.
Author information
Authors and Affiliations
Contributions
Guarantor of integrity of the entire study: JZ, LD, TH, JC. Study concepts: XL, XH. Study design: SS, HW. Definition of intellectual content: LL.
Corresponding author
Ethics declarations
Conflict of interest
The author reports no conflicts of interest in this work.
Ethical Approval
All animal experiments were approved by the Animal Committee of Hebei General Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, J., Dong, L., Huo, T. et al. O-GlcNAc Transferase (OGT) Protects Cerebral Neurons from Death During Ischemia/Reperfusion (I/R) Injury by Modulating Drp1 in Mice. Neuromol Med 24, 299–310 (2022). https://doi.org/10.1007/s12017-021-08688-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08688-6