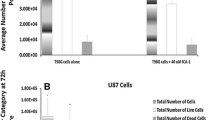
Abstract
Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) has been considered to be one of the most promising candidates in research on treatments for cancer, because it induces apoptosis in a wide variety of cancer cells but not in most normal human cell types. However, many cells including glioblastoma (GBM) cells are resistant to TRAIL-induced apoptosis, which limits the potential application of TRAIL in cancer therapy. Icaritin, a hydrolytic product of icariin from Epimedium Genus, has been identified as a potential therapeutic and preventive agent in renal cell carcinoma and breast cancer. In this study, we investigated whether Icaritin treatment could modulate TRAIL-induced apoptosis in GBM. The effect of icaritin on TRAIL sensitivity was assessed in human GBM U87 and U373 cells. The underlying regulatory cascades were approached by biochemical and pharmacological strategies. We found that nontoxic concentration of icaritin alone had no significant effect on the level of apoptosis, but a combination treatment of TRAIL and icaritin caused a significantly more profound apoptosis. The sensitization was accompanied by c-FLIP down-regulation and inhibition of NF-κB activity. Studies have further demonstrated that silencing NF-κB alone was sufficient to down-regulate c-FLIP expression and sensitized both tested cells to TRAIL-induced apoptosis. These data suggest that icaritin sensitizes TRAIL-induced tumor cell apoptosis via suppression of NF-κB-dependent c-FLIP expression, providing in vitro evidence supporting the notion that icaritin is a potential sensitizer of TRAIL in anticancer therapy against human GBM.




Similar content being viewed by others
References
Fuller, G. N. (2008). The WHO Classification of Tumours of the Central Nervous System, 4th edition. Archives of Pathology and Laboratory Medicine, 132, 906.
Ohgaki, H., & Kleihues, P. (2005). Population-based studies on incidence, survival rates, and genetic alterations in astrocytic and oligodendroglial gliomas. Journal of Neuropathology and Experimental Neurology, 64, 479–489.
Krakstad, C., & Chekenya, M. (2010). Survival signalling and apoptosis resistance in glioblastomas: Opportunities for targeted therapeutics. Molecular Cancer, 9, 135.
Park, D. M., & Rich, J. N. (2009). Biology of glioma cancer stem cells. Molecules and Cells, 28, 7–12.
Stupp, R., Mason, W. P., van Den Bent, M. J., Weller, M., Fisher, B., Taphoorn, M. J., et al. (2005). Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. The New England Journal of Medicine, 352, 987–996.
Kroemer, G., Galluzzi, L., Vandenabeele, P., Abrams, J., Alnemri, E. S., Baehrecke, E. H., et al. (2009). Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death and Differentiation, 16, 3–11.
Ruano, Y., Mollejo, M., Camacho, F. I., Rodriguez de Lope, A., Fiano, C., Ribalta, T., et al. (2008). Identification of survival-related genes of the phosphatidylinositol 3′-kinase signaling pathway in glioblastoma multiforme. Cancer, 112, 1575–1584.
Strik, H., Deininger, M., Streffer, J., Grote, E., Wickboldt, J., Dichgans, J., et al. (1999). BCL-2 family protein expression in initial and recurrent glioblastomas: modulation by radiochemotherapy. Journal of Neurology, Neurosurgery and Psychiatry, 67, 763–768.
Stegh, A. H., Kim, H., Bachoo, R. M., Forloney, K. L., Zhang, J., Schulze, H., et al. (2007). Bcl2L12 inhibits post-mitochondrial apoptosis signaling in glioblastoma. Genes & Development, 21, 98–111.
Singh, S. K., Clarke, I. D., Terasaki, M., Bonn, V. E., Hawkins, C., Squire, J., & Dirks, P. B. (2003). Identification of a cancer stem cell in human brain tumors. Cancer Research, 63, 5821–5828.
Singh, S. K., Hawkins, C., Clarke, I. D., Squire, J. A., Bayani, J., Hide, T., et al. (2004). Identification of human brain tumour initiating cells. Nature, 432, 396–401.
Eramo, A., Ricci-Vitiani, L., Zeuner, A., Pallini, R., Lotti, F., Sette, G., et al. (2006). Chemotherapy resistance of glioblastoma stem cells. Cell Death and Differentiation, 13, 1238–1241.
Lathia, J. D., Gallagher, J., Myers, J. T., Li, M., Vasanji, A., McLendon, R. E., et al. (2011). Direct in vivo evidence for tumor propagation by glioblastoma cancer stem cells. PLoS ONE, 6, e24807.
Ashkenazi, A., Pai, R. C., Fong, S., Leung, S., Lawrence, D. A., Marsters, S. A., et al. (1999). Safety and antitumor activity of recombinant soluble Apo2 ligand. The Journal of Clinical Investigation, 104, 155–162.
Di Pietro, R., & Zauli, G. (2004). Emerging non-apoptotic functions of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)/Apo2L. Journal of Cellular Physiology, 201, 331–340.
Pan, G., O’Rourke, K., Chinnaiyan, A. M., Gentz, R., Ebner, R., Ni, J., & Dixit, V. M. (1997). The receptor for the cytotoxic ligand TRAIL. Science, 276, 111–113.
LeBlanc, H. N., & Ashkenazi, A. (2003). Apo2L/TRAIL and its death and decoy receptors. Cell Death and Differentiation, 10, 66–75.
Kuijlen, J. M., Bremer, E., Mooij, J. J., den Dunnen, W. F., & Helfrich, W. (2010). Review: On TRAIL for malignant glioma therapy? Neuropathology and Applied Neurobiology, 36, 168–182.
Zhu, D. Y., & Lou, Y. J. (2005). Inducible effects of icariin, icaritin, and desmethylicaritin on directional differentiation of embryonic stem cells into cardiomyocytes in vitro. Acta Pharmacologica Sinica, 26, 477–485.
Zhang, G., Qin, L., Sheng, H., Wang, X. L., Wang, Y. X., Yeung, D. K., et al. (2009). A novel semisynthesized small molecule icaritin reduces incidence of steroid-associated osteonecrosis with inhibition of both thrombosis and lipid-deposition in a dose-dependent manner. Bone, 44, 345–356.
Wang, Z., Zhang, X., Wang, H., Qi, L., & Lou, Y. (2007). Neuroprotective effects of icaritin against beta amyloid-induced neurotoxicity in primary cultured rat neuronal cells via estrogen-dependent pathway. Neuroscience, 145, 911–922.
Wang, Z., Wang, H., Wu, J., Zhu, D., Zhang, X., Ou, L., et al. (2009). Enhanced co-expression of beta-tubulin III and choline acetyltransferase in neurons from mouse embryonic stem cells promoted by icaritin in an estrogen receptor-independent manner. Chemico-Biological Interactions, 179, 375–385.
Huang, J., Yuan, L., Wang, X., Zhang, T. L., & Wang, K. (2007). Icaritin and its glycosides enhance osteoblastic, but suppress osteoclastic, differentiation and activity in vitro. Life Sciences, 81, 832–840.
Tong, J. S., Zhang, Q. H., Huang, X., Fu, X. Q., Qi, S. T., Wang, Y. P., et al. (2011). Icaritin causes sustained ERK1/2 activation and induces apoptosis in human endometrial cancer cells. PLoS ONE, 6, e16781.
Zheng, Q., Liu, W. W., Li, B., Chen, H. J., Zhu, W. S., Yang, G. X., et al. (2014). Anticancer effect of icaritin on human lung cancer cells through inducing S phase cell cycle arrest and apoptosis. Journal of Huazhong University of Science and Technology. Medical sciences = Hua zhong ke ji da xue xue bao. Yi xue Ying De wen ban = Huazhong keji daxue xuebao. Yixue Yingdewen ban, 34, 497–503.
Guo, Y., Zhang, X., Meng, J., & Wang, Z. Y. (2011). An anticancer agent icaritin induces sustained activation of the extracellular signal-regulated kinase (ERK) pathway and inhibits growth of breast cancer cells. European Journal of Pharmacology, 658, 114–122.
Li, S., Priceman, S. J., Xin, H., Zhang, W., Deng, J., Liu, Y., et al. (2013). Icaritin inhibits JAK/STAT3 signaling and growth of renal cell carcinoma. PLoS ONE, 8, e81657.
Li, Q., Huai, L., Zhang, C., Wang, C., Jia, Y., Chen, Y., et al. (2013). Icaritin induces AML cell apoptosis via the MAPK/ERK and PI3 K/AKT signal pathways. International Journal of Hematology, 97, 617–623.
Li, L., & Wang, X. M. (2008). Progress of pharmacological research on icariin. ], Zhongguo Zhong yao za zhi = Zhongguo zhongyao zazhi = China Journal of Chinese Materia Medica, 33, 2727–2732.
Jane, E. P., Premkumar, D. R., & Pollack, I. F. (2011). Bortezomib sensitizes malignant human glioma cells to TRAIL, mediated by inhibition of the NF-{kappa}B signaling pathway. Molecular Cancer Therapeutics, 10, 198–208.
Rae, C., Langa, S., Tucker, S. J., & MacEwan, D. J. (2007). Elevated NF-kappaB responses and FLIP levels in leukemic but not normal lymphocytes: Reduction by salicylate allows TNF-induced apoptosis. Proceedings of the National Academy of Sciences of the USA, 104, 12790–12795.
Karsy, M., Gelbman, M., Shah, P., Balumbu, O., Moy, F., & Arslan, E. (2012). Established and emerging variants of glioblastoma multiforme: Review of morphological and molecular features, Folia neuropathologica/Association of Polish Neuropathologists and Medical Research Centre. Polish Academy of Sciences, 50, 301–321.
Bonavia, R., Inda, M. M., Cavenee, W. K., & Furnari, F. B. (2011). Heterogeneity maintenance in glioblastoma: A social network. Cancer Research, 71, 4055–4060.
Chang, S. M., Wen, P., Cloughesy, T., Greenberg, H., Schiff, D., Conrad, C., et al. (2005). Phase II study of CCI-779 in patients with recurrent glioblastoma multiforme. Investigational New Drugs, 23, 357–361.
Rich, J. N., Reardon, D. A., Peery, T., Dowell, J. M., Quinn, J. A., Penne, K. L., et al. (2004). Phase II trial of gefitinib in recurrent glioblastoma. Journal of Clinical Oncology, 22, 133–142.
van den Bent, M. J., Brandes, A. A., Rampling, R., Kouwenhoven, M. C., Kros, J. M., Carpentier, A. F., et al. (2009). Randomized phase II trial of erlotinib versus temozolomide or carmustine in recurrent glioblastoma: EORTC brain tumor group study 26034. Journal of Clinical Oncology, 27, 1268–1274.
Roth, W., Isenmann, S., Naumann, U., Kugler, S., Bahr, M., Dichgans, J., et al. (1999). Locoregional Apo2L/TRAIL eradicates intracranial human malignant glioma xenografts in athymic mice in the absence of neurotoxicity. Biochemical and Biophysical Research Communications, 265, 479–483.
Saito, R., Bringas, J. R., Panner, A., Tamas, M., Pieper, R. O., Berger, M. S., & Bankiewicz, K. S. (2004). Convection-enhanced delivery of tumor necrosis factor-related apoptosis-inducing ligand with systemic administration of temozolomide prolongs survival in an intracranial glioblastoma xenograft model. Cancer Research, 64, 6858–6862.
Hawkins, C. J. (2004). TRAIL and malignant glioma. Vitamins and Hormones, 67, 427–452.
Panner, A., Parsa, A. T., & Pieper, R. O. (2006). Use of APO2L/TRAIL with mTOR inhibitors in the treatment of glioblastoma multiforme. Expert Review of Anticancer Therapy, 6, 1313–1322.
Lee, D. H., Kim, D. W., Jung, C. H., Lee, Y. J., & Park, D. (2014). Gingerol sensitizes TRAIL-induced apoptotic cell death of glioblastoma cells. Toxicology and Applied Pharmacology, 279, 253–265.
Ashkenazi, A., & Dixit, V. M. (1998). Death receptors: Signaling and modulation. Science, 281, 1305–1308.
Irmler, M., Thome, M., Hahne, M., Schneider, P., Hofmann, K., Steiner, V., et al. (1997). Inhibition of death receptor signals by cellular FLIP. Nature, 388, 190–195.
Fulda, S. (2013). Targeting c-FLICE-like inhibitory protein (CFLAR) in cancer. Expert Opinion on Therapeutic Targets, 17, 195–201.
Al-Yacoub, N., Fecker, L. F., Mobs, M., Plotz, M., Braun, F. K., Sterry, W., & Eberle, J. (2012). Apoptosis induction by SAHA in cutaneous T cell lymphoma cells is related to downregulation of c-FLIP and enhanced TRAIL signaling. The Journal of Investigative Dermatology, 132, 2263–2274.
Tian, F., Lu, J. J., Wang, L., Li, L., Yang, J., Li, Y., et al. (2012). Expression of c-FLIP in malignant melanoma, and its relationship with the clinicopathological features of the disease. Clinical and Experimental Dermatology, 37, 259–265.
Schleich, K., Warnken, U., Fricker, N., Ozturk, S., Richter, P., Kammerer, K., et al. (2012). Stoichiometry of the CD95 death-inducing signaling complex: Experimental and modeling evidence for a death effector domain chain model. Molecular Cell, 47, 306–319.
Matsuda, F., Inoue, N., Goto, Y., Maeda, A., Cheng, Y., Sakamaki, K., & Manabe, N. (2008). cFLIP regulates death receptor-mediated apoptosis in an ovarian granulosa cell line by inhibiting procaspase-8 cleavage. The Journal of Reproduction and Development, 54, 314–320.
Murtaza, I., Saleem, M., Adhami, V. M., Hafeez, B. B., & Mukhtar, H. (2009). Suppression of cFLIP by lupeol, a dietary triterpene, is sufficient to overcome resistance to TRAIL-mediated apoptosis in chemoresistant human pancreatic cancer cells. Cancer Research, 69, 1156–1165.
Kang, Y. J., Kim, I. Y., Kim, E. H., Yoon, M. J., Kim, S. U., Kwon, T. K., & Choi, K. S. (2011). Paxilline enhances TRAIL-mediated apoptosis of glioma cells via modulation of c-FLIP, survivin and DR5. Experimental & Molecular Medicine, 43, 24–34.
Naugler, W. E., & Karin, M. (2008). NF-kappaB and cancer-identifying targets and mechanisms. Current Opinion in Genetics & Development, 18, 19–26.
Nogueira, L., Ruiz-Ontanon, P., Vazquez-Barquero, A., Moris, F., & Fernandez-Luna, J. L. (2011). The NFkappaB pathway: A therapeutic target in glioblastoma. Oncotarget, 2, 646–653.
Atkinson, G. P., Nozell, S. E., & Benveniste, E. T. (2010). NF-kappaB and STAT3 signaling in glioma: Targets for future therapies. Expert Review of Neurotherapeutics, 10, 575–586.
Kasuga, C., Ebata, T., Kayagaki, N., Yagita, H., Hishii, M., Arai, H., et al. (2004). Sensitization of human glioblastomas to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) by NF-kappaB inhibitors. Cancer Science, 95, 840–844.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hongxing Han and Bo Xu have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Han, H., Xu, B., Hou, P. et al. Icaritin Sensitizes Human Glioblastoma Cells to TRAIL-Induced Apoptosis. Cell Biochem Biophys 72, 533–542 (2015). https://doi.org/10.1007/s12013-014-0499-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0499-y