Abstract
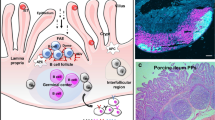
Pathological conditions and harmful drugs cause many gastrointestinal diseases in broiler chicken. The current study was conducted to investigate the effect of trace elements zinc (Zn) and selenium (Se) supplementation on histomorphology, immunological role, and functional activity of goblet cells (GCs) of the small intestine. The Alcian blue-periodic acid-Schiff (AB-PAS) was performed to assess the histomorphological changes in GCs, which revealed the regular dispersion with high electron density of GCs throughout the mucosal surface in the supplemented group. However, irregular dispersion with low electron density of GCs was present in the control group. The immunological functional role of GCs within the small intestine was examined by mucicarmine staining, immunohistochemistry, and immunofluorescence. The results showed a high mucin glycol protein secretion in the supplemented group, whereas limited mucin glycol protein secretion in the control group. Furthermore, the biological significance showed a high and low immunoreactivity of Muc2 and Muc13 in the supplemented and control groups, respectively. Immunofluorescence was used to confirm the immunosignaling of Muc2. Results revealed high immunosignaling of Muc2 at the apical part of the small intestine in the supplementation group, while low immunosignaling of Muc2 in the control group. Results suggest that trace element supplementation had significant effect on morphology and immunological role of GCs, which might be essential for immune function and health status of broiler chicken.






Similar content being viewed by others
Data Availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Juul-Madsen H, Su G, Sørensen P (2004) Influence of early or late start of first feeding on growth and immune phenotype of broilers. Br Poult Sci 45(2):210–222
Uni Z, Ferket P, Tako E, Kedar O (2005) In ovo feeding improves energy status of late-term chicken embryos. Poult Sci 84(5):764–770
Ohta Y, Tsushima N, Koide K, Kidd M, Ishibashi T (1999) Effect of amino acid injection in broiler breeder eggs on embryonic growth and hatchability of chicks. Poult Sci 78(11):1493–1498
Bhanja S, Mandal A (2005) Effect of in ovo injection of critical amino acids on pre-and post-hatch growth, immunocompetence and development of digestive organs in broiler chickens. Asian Australas J Anim Sci 18(4):524–531
Jha R, Singh AK, Yadav S, Berrocoso JFD, Mishra B (2019) Early nutrition programming (in ovo and post-hatch feeding) as a strategy to modulate gut health of poultry. Front Vet Sci 6:82. https://doi.org/10.3389/fvets.2019.00082
Sawosz F, Pineda L, Hotowy A, Hyttel P, Sawosz E, Szmidt M, Niemiec T, Chwalibog A (2012) Nano-nutrition of chicken embryos The effect of silver nanoparticles and glutamine on molecular responses and the morphology of pectoral muscle. Baltic J Comp Clin Syst Biol 2:29–45
Josuha P, Valli C, Balakrishnan V (2016) Effect of in ovo supplementation of nano forms of zinc, copper, and selenium on post-hatch performance of broiler chicken. Vet, World 9(3):287–294
Bartlett J, Smith M (2003) Effects of different levels of zinc on the performance and immunocompetence of broilers under heat stress. Poult Sci 82(10):1580–1588
Sunder GS, Panda A, Gopinath N, Rao SR, Raju M, Reddy M, Kumar CV (2008) Effects of higher levels of zinc supplementation on performance, mineral availability, and immune competence in broiler chickens. J Appl Poultry Res 17(1):79–86
Surai P (2002) Selenium in poultry nutrition 2. Reprod, Egg Meat Qual Pract Appl, World’s Poultry Sci J 58(4):431–450
Dharmani P, Srivastava V, Kissoon-Singh V, Chadee K (2009) Role of intestinal mucins in innate host defense mechanisms against pathogens. J Innate Immun 1(2):123–135
Herbert DBR, Yang J-Q, Hogan SP, Groschwitz K, Khodoun M, Munitz A, Orekov T, Perkins C, Wang Q, Brombacher F (2009) Intestinal epithelial cell secretion of RELM-β protects against gastrointestinal worm infection. J Exp Med 206(13):2947–2957
Johansson ME, Thomsson KA, Hansson GC (2009) Proteomic analyses of the two mucus layers of the colon barrier reveal that their main component, the Muc2 mucin, is strongly bound to the Fcgbp protein. J Proteome Res 8(7):3549–3557
Taupin D, Podolsky DK (2003) Trefoil factors: initiators of mucosal healing. Nat Rev Mol Cell Biol 4(9):721–732
Guy JS (1998) Virus infections of the gastrointestinal tract of poultry. Poult Sci 77(8):1166–1175
Yegani M, Korver D (2008) Factors affecting intestinal health in poultry. Poult Sci 87(10):2052–2063
Contreras-Ruiz L, Ghosh-Mitra A, Shatos MA, Dartt DA, Masli S (2013) Modulation of conjunctival goblet cell function by inflammatory cytokines. Mediators of Inflamm 2013:636812. https://doi.org/10.1155/2013/636812
Kim YS, Ho SB (2010) Intestinal goblet cells and mucins in health and disease: recent insights and progress. Curr Gastroenterol Rep 12(5):319–330
McGuckin MA, Eri R, Simms LA, Florin TH, Radford-Smith G (2009) Intestinal barrier dysfunction in inflammatory bowel diseases. Inflamm Bowel Dis 15(1):100–113
Bhanja S, Mandal A, Johri T (2004) Standardization of injection site, needle length, embryonic age and concentration of amino acids for in ovo injection in broiler breeder eggs. Indian J Poult Sci 39(2):105–111
Tahmasebi S, Toghyani M (2016) Effect of arginine and threonine administered in ovo on digestive organ developments and subsequent growth performance of broiler chickens. J Anim Physiol Anim Nutr (Berl) 100(5):947–956
Luna LG (1968) Manual of histologic staining methods of the Armed Forces Institute of Pathology. In Manual of histologic staining methods of the Armed Forces Institute of Pathology. Google Scholar, pp xii–258
Osho SO, Wang T, Horn NL, Adeola O (2017) Comparison of goblet cell staining methods in jejunal mucosa of turkey poults. Poult Sci 96:556–59
Hedman HD, Vasco KA, Zhang L (2020) A review of antimicrobial resistance in poultry farming within low-resource settings. Animals 10(8):1264. https://doi.org/10.3390/ani10081264
Bergstrom KS, Kissoon-Singh V, Gibson DL, Ma C, Montero M, Sham HP, Ryz N, Huang T, Velcich A, Finlay BB (2010) Muc2 protects against lethal infectious colitis by disassociating pathogenic and commensal bacteria from the colonic mucosa. PLoS Pathog 6(5):e1000902
Koninkx J, Mirck M, Hendriks H, Mouwen J, Van Dijk J (1988) Nippostrongylus brasiliensis histochemical changes in the composition of mucins in goblet cells during infection in rats. Exp Parasitol 65(1):84–90
Smirnova MG, Guo L, Birchall JP, Pearson JP (2003) LPS up-regulates mucin and cytokine mRNA expression and stimulates mucin and cytokine secretion in goblet cells. Cell Immunol 221(1):42–49
Cornick S, Tawiah A, Chadee K (2015) Roles and regulation of the mucus barrier in the gut. Tissue barriers 3(1–2):e982426
Singh R, Kaushik S, Wang Y, Xiang Y, Novak I, Komatsu M, Tanaka K, Cuervo AM, Czaja MJ (2009) Autophagy regulates lipid metabolism. Nature 458(7242):1131
Atuma C, Strugala V, Allen A, Holm L (2001) The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am J Physiol-Gastrointestinal Liver Physiol 280(5):922–929
Maares M, Keil C, Koza J, Straubing S, Schwerdtle T, Haase H (2018) In vitro studies on zinc binding and buffering by intestinal mucins. Int J Mol Sci 19(9):2662. https://doi.org/10.3390/ijms19092662
Abbasi-Oshaghi E, Mirzaei F, Mirzaei A (2018) Effects of ZnO nanoparticles on intestinal function and structure in normal/high fat diet-fed rats and Caco-2 cells. Nanomedicine (Lond) 13(21):2791–2816
Zhang S, Cheng S, Jiang X, Zhang J, Bai L, Qin X, Zou Z, Chen C (2020) Gut-brain communication in hyperfunction of 5-hydroxytryptamine induced by oral zinc oxide nanoparticles exposure in young mice. Food Chem Toxicol : an Intl J Published Bri Ind Biol Res Assoc 135:110906
Maares M, Keil C, Straubing S, Robbe-Masselot C, Haase H (2020) Zinc deficiency disturbs mucin expression, O-glycosylation and secretion by intestinal goblet cells. Int J Mol Sci 21(17):6149. https://doi.org/10.3390/ijms21176149
Hafez A, Nassef E, Fahmy M, Elsabagh M (2020) Impact Diet Nano-zinc Oxide On Immune Response Antioxid Def Broiler Chickens 27(16):19108–19114
Khajeh Bami M, Afsharmanesh M, Espahbodi M, Esmaeilzadeh E (2022) Effects of dietary nano-selenium supplementation on broiler chicken performance meat selenium content, intestinal microflora intestinal morphology and immune response. J Trace Elem Med Biol : Organ Soc Minerals Trace Elem (GMS) 69:126897
Chen Y, Wu W, Zhou H, Liu X, Li S, Guo Y, Li Y, Wang Y, Yuan J (2022) Selenium nanoparticles improved intestinal health through modulation of the NLRP3 signaling pathway. Front Nutr 9:907386
Gupta BK, Maher DM, Ebeling MC, Stephenson PD, Puumala SE, Koch MR, Aburatani H, Jaggi M, Chauhan SC (2014) Functions and regulation of MUC13 mucin in colon cancer cells. J Gastroenterol 49(10):1378–1391
Acknowledgements
Firstly, the authors are thankful to the Central Animal House (CAH) section of Advanced Biological and Animal Research, DUHS, OJHA Campus Karachi, Pakistan, for the great support in data analysis and research sample processing. Additionally, the authors are very grateful to Professor Juan Liu, College of Veterinary Medicine, Southwest University, Chongqing, China, for technical assistance throughout all aspects of our study and for their help in writing the manuscript.
Funding
This work was financially supported by funds from the Pakistan Science Foundation (PSF) Islamabad under the project title “Investigate the effect of in ovo feeding of amino acids and trace elements on immunity and production performance of post-hatch broiler chicken” (award no.: PSF/NSLP/B-LUAWMS (763)).
Author information
Authors and Affiliations
Contributions
F.N. wrote the manuscript text and M.A.A., S.A.F., and M.K. prepared all figures and F.B., S.A., S.K.F., and J.L. reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nabi, F., Arain, M.A., Fazlani, S.A. et al. Effect of In Ovo Trace Element Supplementation on Immune-Related Cells of the Small Intestine of Post-hatched Broiler Chicken. Biol Trace Elem Res 201, 4052–4061 (2023). https://doi.org/10.1007/s12011-022-03492-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03492-0