Abstract
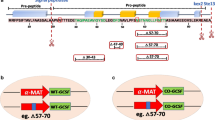
Granulocyte colony-stimulating factor (GCSF) has therapeutic applications due to its proven efficacy in different forms of neutropenia and chemotherapy-induced leucopenia. The original 564-bp nucleotide sequence from NCBI was codon optimized and assembled by overlapping PCR method comprising of 16 oligos of 50-nt length with 15 base overhang. The synthetic gene (CO-GCSF) was cloned under glucose utilizing glyceraldehyde 3-phosphate dehydrogenase (GAP) and methanol-utilizing alcohol oxidase (AOX1) promoters and expressed in Pichia pastoris SMD1168 strain. Constitutive expression under GAP resulted in cellular toxicity while AOX1 promoter controlled expression was stable. Variation in the levels of expression was observed among the transformant colonies with transformant #2 secreting up to ∼4 mg/L of GCSF. The molecular mass of the expressed GCSF in P. pastoris was ∼19.0 kDa. Quatitation of the expressed protein was carried out by a highly reproducible gel densitometric method. Effect of several operational and nutritional conditions was studied on GCSF production and the results suggest a general approach for increasing the yield of GCSF several folds (2- to 5-fold) over the standard conditions employed currently. Cultivation of the single-copy integrant in the chemically defined medium in a 5-L fermenter resulted in a volumetric productivity of ∼0.7 mg/L/h at the end of the induction phase, which was about 4-fold higher than attained in the shake flask.





Similar content being viewed by others
References
Biethahna, S., Alvesa, F., Wildea, S., Hiddemanna, W., & Spiekermann, K. (1999). Expression of granulocyte colony-stimulating factor and granulocyte-macrophage colony stimulating factor associated signal transduction proteins of the JAK/STAT pathway in normal granulopoiesis and in blast cells of acute myelogenous leukemia. Experimental Hematology, 27, 885–894.
Viret, F., Goncalves, A., Tarpin, C., Chabannon, C., et al. (2006). GCSF in oncology. Bulletins Cancer, 93, 463–471.
Bell, E. (2009). Transplantation: GCSF therapy after BMT: getting the timing right. Nature Reviews in Immunology, 9, 308–309.
Nagata, S., Tsuchiya, M., Asano, S., Yamamoto, O., Hirata, Y., Kubota, N., Oheda, M., Nomura, H., & Yamazaki, T. (1986). The chromosomal gene structure and two mRNAs for human granulocyte colony stimulating factor. EMBO Journal, 5, 575–581.
Kubota, N., Orita, T., Hattori, K., Oh-eda, M., et al. (1990). Structural characterization of natural and recombinant human granulocyte colony-stimulating factors. Journal of Biochemistry, 107, 486–492.
Oh-eda, M., Hasegawa, M., Hattori, K., Kuboniwa, H., et al. (1990). O-linked sugar chain of human granulocyte colony-stimulating factor protects it against polymerization and denaturation allowing it to retain its biological activity. Journal of Biological Chemistry, 265, 11432–11435.
Carter, C. R. D., Whitmore, K. M., & Thorpe, R. (2004). The significance of carbohydrates on G-CSF: differential sensitivity of GCSFs’ to human neutrophil elastase degradation. Journal of Leukocyte Biology, 75, 515–522.
Maccani, A., Landes, N., Stadlmayr, G., Maresch, D., Leitner, C., Maurer, M., Gasser, B., Ernst, W., Kunert, R., & Mattanovich, D. (2014). Pichia pastoris secretes recombinant proteins less efficiently than Chinese hamster ovary cells but allows higher space-time yields for less complex proteins. Biotechnology Journal, 9, 526–537.
Werten, M. W. T., Wisselink, W. H., Jansen-van den Bosch, T. J., de Bruin, E. C., et al. (2001). Secreted production of a custom-designed, highly hydrophilic gelatin in Pichia pastoris. Protein Engineering, 14, 447–454.
Trinh, L. B., Phue, J. N., & Shiloach, J. (2003). Effect of methanol feeding strategies on production and yield of recombinant mouse endostatin from Pichia pastoris. Biotechnology and Bioengineering, 82, 438–444.
Fickers, P. (2014). Pichia pastoris: a workhorse for recombinant protein production. Current research in Microbiology and Biotechnology, 2, 354–363.
Saeedinia, A., Shamsara, M., Bahrami, A., Zeinoddini, M., et al. (2008). Heterologous expression of human granulocyte colony stimulating factor in Pichia pastoris. Biotechnology, 2, 1–5.
Bahrami, A., Shojaosadati, S. A., Khalilzadeh, R., Mohammadian, J., Farahani, E. V., & Masoumian, M. R. (2009). Prevention of human granulocyte colony-stimulating factor protein aggregation in recombinant Pichia pastoris fed-batch fermentation using additives. Biotechnology and Applied Biochemistry, 52, 141–148.
Lasnik, M. A., Porekar, V. G., & Stalc', A. (2001). Human granulocyte colony stimulating factor (hG-CSF) expressed by methylotrophic yeast Pichia pastoris. Pfliigers Archives of European Journal of Physiology, 442, R184–R186.
Apte-Deshpande, A., Somani, S., Mandal, G., Soorapaneni, S., & Padmanabhan, S. (2009). Over expression and analysis of O-glycosylated recombinant human granulocyte colony stimulating factor in Pichia pastoris using Agilent 2100 Bioanalyzer. Journal of Biotechnology, 143, 44–50.
Angov, E. (2011). Codon usage: nature’s roadmap to expression and folding of protein. Biotechnology Journal, 6, 650–659.
Macauley-Patrick, S., Fazenda, M. L., McNeil, B., & Harvey, L. M. (2005). Heterologous protein production using the Pichia pastoris expression system. Yeast, 22, 249–270.
Pal, Y., Khusboo, A., & Mukherjee, K. J. (2006). Process optimization of constitutive human granulocyte macrophage colony-stimulating factor (hGM-CSF) expression in Pichia pastoris fed batch culture. Applied Microbiology and Biotechnology, 69, 650–657.
Vogl, T., & Glieder, A. (2013). Regulation of Pichia pastoris promoters and its consequences for protein production. Nature Biotechnology, 30, 385–404.
Viader-Salvado, J. M., Cab-Barrera, E. L., Galan-Wong, L. J., & Guerrero-Olazaran, M. (2006). Genotyping of recombinant Pichia pastoris strains. Cellular and Molecular Biology Letters, 11, 348–359.
Jafari, R., Sundstrom, B. E., & Holm, P. (2011). Optimization of production of the anti keratin 8 single chain Fv TS-218 in Pichia pastoris using design of experiments. Microbial Cell Factories, 10, 34.
Jahic, M., Wallberg, F., Bollock, M., Garcia, P., & Enfors, S. O. (2003). Temperature limited fed-batch technique for control of proteolysis in Pichia pastoris bioreactor cultures. Microbial Cell Factories, 2, 6.
André, N., Cherouati, N., Prual, C., Steffan, T., Zeder-Lutz, G., Magnin, T., Pattus, F., Michel, H., Wagner, R., & Reinhart, C. (2006). Enhancing functional production of G protein-coupled receptors in Pichia pastoris to levels required for structural studies via a single expression system. Protein Science, 15, 1115–1126.
Batra, J., Beri, D., & Mishra, S. (2014). Response surface methodology based optimization of β–glucosidase production from Pichia pastoris. Applied Biochemistry and Biotechnology, 172, 380–393.
Athmaram, T. N., Singh, A. K., Saraswat, S., Srivastava, S., Miisra, P., Rao, M. K., Gopalan, N., & Rao, P. V. L. (2013). A simple Pichia pastoris fermentation and downstream processing strategy for making recombinant pandemic swine origin influenza A virus hemagglutinin protein. Journal of Industrial Microbiology and Biotechnology, 40, 245–255.
Brierley, R. A., Davis, G. R., Holtz, G.. C., Gleeson, M. A., Howard, B. D. (1997) Production of insulin-like growth factor-1 in methylotropic yeast cells. Google Patents, US 08/308, 196.
Werten, M. W. T., Wisselink, W. H., Jansen-van Den Bosch, T. J. C., de Bruin, E. A., & de Wolf, F. (2001). Secreted production of a custom-designed, highly hydrophilic gelatin in Pichia pastoris. Protein Engineering, 14, 447–454.
Ricci, M. S., Sarkar, C. A., Fallon, E. M., Lauffenburger, D. A., & Brems, D. N. (2003). pH dependence of structural stability of interleukin-2 and granulocyte colony-stimulating factor. Protein Science, 12, 1030–38.
Gorgens, J. F., van Zyl, W. H., Knoetze, J. H., & Hahn-Hagerdal, B. (2000). Amino acid supplementation improves heterologous protein production by Saccharomyces cerevisiae in defined medium. Applied Microbiology and Biotechnology, 67, 684–691.
Heyland, J., Fu, J., Blank, L. M., & Schmid, A. (2011). Carbon metabolism limits recombinant protein production in Pichia pastoris. Biotechnology and Bioengineering, 108, 1942–1953.
Ramon, R., Ferrer, P., & Valero, F. (2007). Sorbitol co-feeding reduces metabolic burden caused by the overexpression of Rhizopus oryzae lipase in Pichia pastoris. Journal of Biotechnology, 130, 39–46.
Cos, O., Ramon, R., Montesinos, J. L., & Valero, F. (2006). Operational strategies, monitoring and control of heterologous protein production in the methylotrophic yeast Pichia pastoris under different promoters: a review. Microbial Cell Factories, 5, 17.
Maurer, M., Kuhleitner, M., Gasser, B., & Mattanovich, D. (2006). Versatile modeling and optimization of fed batch processes for the production of secreted heterologous proteins with Pichia pastoris. Microbial Cell Factories, 5, 37.
Werner, R. J. (2004). Economic aspects of commercial manufacture of biopharmaceuticals. Journal of Biotechnology, 113, 171–182.
Chien, S.-F. (2010). Cloning and expression of bioactive human granulocyte colony stimulating factor in Pichia pastoris. Journal of Chinese Chemical Society, 57, 850–856.
Acknowledgments
The authors wish to thank IIT Delhi for providing funds to carry out this research under the “High impact research and technology leadership project.”
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maity, N., Thawani, A., Sharma, A. et al. Expression and Control of Codon-Optimized Granulocyte Colony-Stimulating Factor in Pichia pastoris . Appl Biochem Biotechnol 178, 159–172 (2016). https://doi.org/10.1007/s12010-015-1865-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1865-y