Abstract
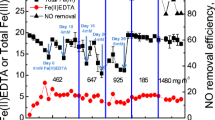
In this work, several conditions of pH and inlet load (IL) were applied to a scale laboratory biofilter treating n-hexane vapors during 143 days. During the first 79 days of operation (period 1, P1), the system was fed with neutral pH mineral medium (MM) and the IL was progressively decreased from 177 to 16 g m−3 h−1. A maximum elimination capacity (EC) of 30 g m−3 h−1 was obtained at an IL of 176.9 ± 9.8 g m−3 h−1. During the following 64 days (period 2, P2), acidic conditions were induced by feeding the biofilter with acidic buffer solution and pH 4 MM in order to evaluate the effect of bacterial community changes on EC. Within the acidic period, a maximum EC of 54 g m−3 h−1 (IL 132.3 ± 13 g m−3 h−1) was achieved. Sequence analysis of 16S rDNA genes amplified from the consortium revealed the presence of Sphingobacteria, Actinobacteria, and α-, β- and γ-Proteobacteria. An Actinobacteria of the Mycobacterium genus had presence throughout the whole experiment of biofiltration showing resistance to fluctuating pH and IL conditions. Batch tests confirm the bacterial predominance and a negligible contribution of fungi in the degradation of n-hexane.




Similar content being viewed by others
References
Environmental Protection Agency (EPA) (2007). n-Hexane, http://epa.gov/ttn/atw/hlthef.html. Accessed 25 March 2013.
Gutierrez-Acosta, O. B., Arriaga, S., Escobar-Barrios, V. A., Casas-Flores, S., & Almendarez-Camarillo, A. (2012). Performance of innovative PU-foam and natural fiberbased composites for the biofiltration of a mixture of volatile organic compounds by a fungal biofilm. Journal of Hazardous Materials, 201–202, 202–208.
Muñoz, R., Arriaga, S., Hernández, S., Guieysse, B., & Revah, S. (2006). Enhanced hexane biodegradation in a two phase partitioning bioreactor: overcoming pollutant transport limitations. Process Biochemistry, 41, 1614–1619.
Galindo, H., Revah, S., Cervantes, F. J., & Arriaga, S. (2011). Effect of surfactant and oil additions in the biodegradation of hexane and toluene vapors in batch tests. Environmental Technology, 32, 167–173.
Hassan, A. A., & Sorial, G. A. (2010). Biofiltration of n-hexane in the presence of benzene vapors. Journal of Chemical Technology and Biotechnology, 85, 371–377.
Zedraoui, A., Aly, H. A., & Sorial, G. A. (2012). Effect of methanol on the biofiltration of n-hexane. Journal of Hazardous Materials, 138, 543–548.
Arriaga, S., & Revah, S. (2005). Improving hexane removal by enhancing fungal development in a microbial consortium biofilter. Biotechnology and Bioengineering, 90, 107–115.
Aizpuru, A., Dunat, B., Christen, P., Auria, R., García-Peña, I., & Revah, S. (2005). Fungal biofiltration of toluene on ceramic rings. Journal of Environmental Engineering, 131, 396–402.
Prenafeta-Boldú, F. X., Summerbell, R., & de Hoog, G. S. (2006). Fungi growing on aromatic hydrocarbons: biotechnology’s unexpected encounter with biohazard? FEMS Microbiology Ecology, 30, 109–130.
Barcón, T., Alonso-Gutiérrez, J., & Omil, F. (2012). Molecular and physiological approaches to understand the ecology of methanol degradation during the biofiltration of air streams. Chemosphere, 87, 1179–1185.
Okunishi, S., Morita, Y., Higuchi, T., Maeda, H., & Nishi, K. (2012). Transformation of microflora during degradation of gaseous toluene in a biofilter detected using PCR-DGGE. Journal of the Air & Waste Management Association, 62, 748–757.
Prenafeta-Boldú, F. X., Guivernau, M., Gallastegui, G., Viñas, M., de Hoog, G. S., & Elías, A. (2012). Fungal/bacterial interactions during the biodegradation of TEX hydrocarbons (toluene, etylbenzene and p-xylene) in gas biofilters operated under xerophilic conditions. FEMS Microbiology, 80, 722–734.
Amouric, A., Verhé, F., Auria, R., & Casalot, L. (2006). Study of a hexane degrading consortium in a biofilter and in a liquid culture: biodiversity, kinetics, and characterization of degrading strains. FEMS Microbiology Ecology, 55, 239–247.
Friedrich, M., & Lipski, A. (2010). Characterization of hexane-degrading microorganisms in a biofilter by stable isotope-based fatty acid analysis, FISH and cultivation. Applied Microbiology and Biotechnology, 85, 1189–1199.
Sharefdeen, Z., & Singh, A. (2005). Biotechnology for odor and air pollution control (pp. 29–64). Berlin: Springer.
García-Perez, T., Aizpuru, A., & Arriaga, S. (2013). By-passing acidification limitations during the biofiltration of highformaldehyde loads via the application of ozone pulses. Journal of Hazardous Materials, 262, 732–740.
Gutierrez-Acosta, O., Escobar-Barrios, V., & Arriaga, S. (2010). Batch biodegradation of hydrocarbon vapors using a modified polymeric support. Journal of Chemical Technology and Biotechnology, 85, 410–415.
Gabor, E. M., de Vries, E. J., & Janssen, D. B. (2003). Efficient recovery of environmental DNA for expression cloning by indirect extraction methods. FEMS Microbiology Ecology, 44, 153–163.
Turner, S., Pryer, K. M., Miao, V. P. W., & Palmer, J. D. (1999). Investigating deep phylogenetic relationships among cyanobacteria and plastids by small subunit rRNA sequence analysis. Journal of Eukaryotic Microbiology, 46, 327–338.
Birnboim, H. C., & Doly, J. (1979). A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Research, 7, 1513–1523.
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M., & Kumar, S. (2011). Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular Biology and Evolution, 28, 2731–2739.
Vigueras, G., Arriaga, S., Shirai, K., Morales, M., & Revah, S. (2009). Hydrophobic response of the fungus Rhinocladiella similis in the biofiltration with volatile organic compounds with different polarity. Biotechnological Letters, 31, 1203–1209.
Groesnetijn, J. W., & Lake, M. E. (1999). Elimination of alkanes from off-gases using biotrickling filters containing two liquid phases. Environmental Progress, 18, 151–155.
Hernández-Meléndez, O., Bárzana, E., Arriaga, S., Hernández-Luna, M., & Revah, S. (2008). Fungal removal of gaseous hexane in biofilters packed with poly (ethylene carbonate), pine sawdust or peat composites. Biotechnology and Bioengineering, 100, 864–871.
Kibazohi, O., Yun, S., & Anderson, W. A. (2004). Removal of hexane in biofilters packed with perlite and a peat-perlite mixture. World Journal of Microbiology and Biotechnology, 20, 337–343.
Wang, C., Kong, X., & Zhang, X. Y. (2012). Mesophilic and thermophilic biofiltration of gaseous toluene in a long-term operation: Performance evaluation, biomass accumulation, mass balance analysis and isolation identification. Journal of Hazardous Materials, 229–230, 94–99.
Saucedo-Lucero, J. O., & Arriaga, S. (2013). Photocatalytic degradation of hexane vapors in batch and continuous systems using impregnated ZnO nanoparticles. Chemical Engineering Journal, 218, 358–367.
Saadoun, I., Al-Akhras, M. A., & Abu-Ashour, J. (1999). Bacterial degradation of hydrocarbons as evidenced by respirometric analysis. Microbios, 100, 19–25.
Roy, S., Gendron, J., Delhomenie, M. C., Bibeau, L., Heitz, M., & Brzezinski, R. (2003). Pseudomonas putida as the dominant toluene-degrading bacterial species during air decontamination by biofiltration. Applied Microbiology and Biotechnology, 61, 366–373.
Lee, E. H., Kim, J., Cho, K. S., Ahn, Y. G., & Hwang, G. H. (2009). Degradation of hexane and other recalcitrant hydrocarbons by a novel isolate Rhodococcus sp. EH831. Environmental Science and Pollution Research, 17, 64–77.
Vergara-Fernández, A., Van Haaren, B., & Revah, S. (2006). Phase partition of gaseous hexane and surface hydrophobicity of Fusarium solani when grown in liquid and solid media with hexanol and hexane. Biotechnological Letters, 28, 2011–2017.
Mazumder, S., Falkinham, J. O., Dietrich, A. M., & Puri, I. K. (2009). Role of hydrophobicity in bacterial adherence to carbon nanostructures and biofilm formation. Biofouling, 26, 333–339.
Prenafeta-Boldú, F. X., Ortega, O., Arimany, M., & Canalias, F. (2012). Assessment of process limiting factors during the biofiltration of odorous VOCs in a full-scale composting plant. Compost Science and Utilization, 20, 73–78.
Kiel, L. J., & Olson, F. N. (2000). The physicochemical surface characteristics of Lactobacillus casei. Food Microbiology, 17, 277–291.
Acknowledgments
We would like to thank CONACYT for its financial support to Sonia Arriaga (SEP-CONACYT-CB-2009-133930 Grant). Edgardo Valenzuela Reyes is thankful to CONACYT for the Bachelor scholarship (Number: 17434). The use of the analytical infrastructure of LINAN and LANBAMA are also acknowledged. Special gratitude is expressed to M.Sc. Gladis Labrada, M.Sc. JP. Rodas, M.Sc. Dulce Partida, and M.Sc. Guillermo Vidriales for their technical assistance. We thank Dr. Aitor Aizpuru and Anne Jennifer Eckerly for their suggestions to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valenzuela-Reyes, E., Casas-Flores, S., Isordia-Jasso, I. et al. Performance and Bacterial Population Composition of an n-Hexane Degrading Biofilter Working Under Fluctuating Conditions. Appl Biochem Biotechnol 174, 832–844 (2014). https://doi.org/10.1007/s12010-014-1079-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1079-8