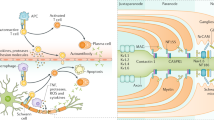
Abstract
Purpose of review
The purpose of this review article is to discuss the pathogenesis of acute and chronic immune-mediated neuropathies along with the recent advances in their treatment.
Recent findings
Since the first description of Guillain-Barre syndrome (GBS) more than a century ago, there have been numerous forms of immune-mediated neuropathies described expanding the spectrum. Understanding the role of the immune system in the pathogenesis of immune-mediated neuropathies has been an advancement towards the diagnosis and treatment.
Summary
It is postulated that immune-mediated neuropathies are a group of diseases resulting from autoimmunity towards multiple components of peripheral nervous system. These have a wide range of pathologic mechanisms, defined clinical presentations, electro-diagnostic and laboratory findings which help in diagnosis and management. Although immunosuppression is the common modality of treatment for these disorders, uncovering distinct pathogenic mechanisms can allow for targeted immunomodulation.


Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Barohn RJ. Approach to peripheral neuropathy and neuronopathy. Semin Neurol. 1998;18:7–18.
Willison HJ, Jacobs BC, van Doorn PA. Guillain-Barre syndrome. Lancet. 2016;388:717–27.
•• Esposito S, Longo MR. Guillain-Barre syndrome. Autoimmun Rev. 2017;16:96–101. This review summarises the clinical features and diagnostic criteria of GBS and proposes the algorithm for management.
Asbury AK, Cornblath DR. Assessment of current diagnostic criteria for Guillain-Barré syndrome. Ann Neurol. 1990;27 Suppl:S21–4.
van den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs BC, van Doorn PA. Guillain-Barre syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol. 2014;10:469–82.
Ropper AH. The Guillain-Barré syndrome. N Engl J Med. 1992;326:1130–6.
•• Dalakas MC. Pathogenesis of immune-mediated neuropathies. Biochim Biophys Acta. 2015;1852:658–66. This article reviews the pathogenesis of GBS, CIDP, MMN and anti-MAG neuropathy.
•• Kieseier BC, Mathey EK, Sommer C, Hartung H-P. Immune-mediated neuropathies. Nat Rev Dis Prim. 2018;4:31. This is a recent comprehensive review paper on pathogenesis, diagnosis and management of AIDP, CIDP and MMN.
Lawn ND, Fletcher DD, Henderson RD, Wolter TD, Wijdicks EF. Anticipating mechanical ventilation in Guillain-Barre syndrome. Arch Neurol. 2001;58:893–8.
Ruts L, Drenthen J, Jongen JLM, Hop WCJ, Visser GH, Jacobs BC, et al. Pain in Guillain-Barre syndrome: a long-term follow-up study. Neurology. 2010;75:1439–47.
The Guillain-Barré Syndrome Study Group. Plasmapheresis and acute Guillain-Barre syndrome. Neurology. 1985;35:1096 LP–1096.
van der Meché FGA, Schmitz PIM. A randomized trial comparing intravenous immune globulin and plasma exchange in Guillain-Barré syndrome. N Engl J Med. 1992;326:1123–9.
Hughes RAC, Swan A V, van Doorn PA. Intravenous immunoglobulin for Guillain-Barre syndrome. Cochrane Database Syst Rev. 2014;CD002063.
Hughes RAC, Swan AV, Raphaël J-C, Annane D, van Koningsveld R, van Doorn PA. Immunotherapy for Guillain-Barré syndrome: a systematic review. Brain. 2007;130:2245–57.
Hughes RA. Plasma exchange versus intravenous immunoglobulin for Guillain-Barré syndrome. Ther Apher. 1997;1:129–30.
Hughes RAC, Brassington R, Gunn AA, van Doorn PA. Corticosteroids for Guillain-Barré syndrome. Cochrane Database Syst Rev. 2016.
Davidson AI, Halstead SK, Goodfellow JA, Chavada G, Mallik A, Overell J, et al. Inhibition of complement in Guillain-Barre syndrome: the ICA-GBS study. J Peripher Nerv Syst. 2017;22:4–12.
Misawa S, Kuwabara S, Sato Y, Yamaguchi N, Nagashima K, Katayama K, et al. Safety and efficacy of eculizumab in Guillain-Barré syndrome: a multicentre, double-blind, randomised phase 2 trial. Lancet Neurol. 2018;17:519–29.
Herbison GJ, Jaweed MM, Ditunno JFJ. Exercise therapies in peripheral neuropathies. Arch Phys Med Rehabil. 1983;64:201–5.
Lunn MPT, Manji H, Choudhary PP, Hughes RAC, Thomas PK. Chronic inflammatory demyelinating polyradiculoneuropathy: a prevalence study in south east England. J Neurol Neurosurg Psychiatry. 1999;66:677–80.
Mathey EK, Park SB, Hughes RAC, Pollard JD, Armati PJ, Barnett MH, et al. Chronic inflammatory demyelinating polyradiculoneuropathy: from pathology to phenotype. J Neurol Neurosurg Psychiatry. 2015;86:973–85.
Katz JS, Saperstein DS, Gronseth G, Amato AA, Barohn RJ. Distal acquired demyelinating symmetric neuropathy. Neurology. 2000;54:615–20.
Lewis RA, Sumner AJ, Brown MJ, Asbury AK. Multifocal demyelinating neuropathy with persistent conduction block. Neurology. 1982;32:958–64.
Sinnreich M, Klein CJ, Daube JR, Engelstad J, Spinner RJ, Dyck PJB. Chronic immune sensory polyradiculopathy. Neurology. 2004;63:1662–9.
Querol L, Illa I. Paranodal and other autoantibodies in chronic inflammatory neuropathies. Curr Opin Neurol. 2015;28:474–9.
Berger M, Allen JA. Optimizing IgG therapy in chronic autoimmune neuropathies: a hypothesis driven approach. Muscle Nerve. 2015;51:315–26.
Cocito D, Paolasso I, Antonini G, Benedetti L, Briani C, Comi C, et al. A nationwide retrospective analysis on the effect of immune therapies in patients with chronic inflammatory demyelinating polyradiculoneuropathy. Eur J Neurol. 2010;17:289–94.
Hughes RAC, Donofrio P, Bril V, Dalakas MC, Deng C, Hanna K, et al. Intravenous immune globulin (10% caprylate-chromatography purified) for the treatment of chronic inflammatory demyelinating polyradiculoneuropathy (ICE study): a randomised placebo-controlled trial. Lancet Neurol. 2008;7:136–44.
• van Schaik IN, Bril V, van Geloven N, Hartung H-P, Lewis RA, Sobue G, et al. Subcutaneous immunoglobulin for maintenance treatment in chronic inflammatory demyelinating polyneuropathy (PATH): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2018;17:35–46 Study on SCIg in CIDP.
Markvardsen LH, Sindrup SH, Christiansen I, Olsen NK, Jakobsen J, Andersen H. Subcutaneous immunoglobulin as first-line therapy in treatment-naive patients with chronic inflammatory demyelinating polyneuropathy: randomized controlled trial study. Eur J Neurol. 2017;24:412–8.
Dyck PJ, O’Brien PC, Oviatt KF, Dinapoli RP, Daube JR, Bartleson JD, et al. Prednisone improves chronic inflammatory demyelinating polyradiculoneuropathy more than no treatment. Ann Neurol. 1982;11:136–41.
Nobile-Orazio E, Cocito D, Jann S, Uncini A, Beghi E, Messina P, et al. Intravenous immunoglobulin versus intravenous methylprednisolone for chronic inflammatory demyelinating polyradiculoneuropathy: a randomised controlled trial. Lancet Neurol. 2012;11:493–502.
van Schaik IN, Eftimov F, van Doorn PA, Brusse E, van den Berg LH, van der Pol WL, et al. Pulsed high-dose dexamethasone versus standard prednisolone treatment for chronic inflammatory demyelinating polyradiculoneuropathy (PREDICT study): a double-blind, randomised, controlled trial. Lancet Neurol. 2010;9:245–53.
Mehndiratta MM, Hughes RAC, Pritchard J. Plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev. 2015;CD003906.
Good JL, Chehrenama M, Mayer RF, Koski CL. Pulse cyclophosphamide therapy in chronic inflammatory demyelinating polyneuropathy. Neurology. 1998;51:1735 LP–1738.
Brannagan TH 3rd, Pradhan A, Heiman-Patterson T, Winkelman AC, Styler MJ, Topolsky DL, et al. High-dose cyclophosphamide without stem-cell rescue for refractory CIDP. Neurology. 2002;58:1856–8.
Mahdi-Rogers M, Brassington R, Gunn AA, van Doorn PA, Hughes RA. Immunomodulatory treatment other than corticosteroids, immunoglobulin and plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev. 2017;5:CD003280.
Querol L, Rojas-Garcia R, Diaz-Manera J, Barcena J, Pardo J, Ortega-Moreno A, et al. Rituximab in treatment-resistant CIDP with antibodies against paranodal proteins. Neurol Neuroimmunol Neuroinflammation. 2015;2:e149.
Van den Bergh PYK, Hadden RDM, Bouche P, Cornblath DR, Hahn A, Illa I, et al. European Federation of Neurological Societies/peripheral nerve society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripher. Eur J Neurol. 2010;17:356–63.
van Nes SI, Vanhoutte EK, van Doorn PA, Hermans M, Bakkers M, Kuitwaard K, et al. Rasch-built overall disability scale (R-ODS) for immune-mediated peripheral neuropathies. Neurology. 2011;76:337–45.
Draak THP, Pruppers MHJ, van Nes SI, Vanhoutte EK, Bakkers M, Gorson KC, et al. Grip strength comparison in immune-mediated neuropathies: vigorimeter vs. Jamar. J Peripher Nerv Syst. 2015;20:269–76.
Magy L, Chassande B, Maisonobe T, Bouche P, Vallat J-M, Leger J-M. Polyneuropathy associated with IgG/IgA monoclonal gammopathy: a clinical and electrophysiological study of 15 cases. Eur J Neurol. 2003;10:677–85.
Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF, et al. A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med. 2002;346:564–9.
Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Offord JR, et al. Prevalence of monoclonal gammopathy of undetermined significance. N Engl J Med. 2006;354:1362–9.
Tatum AH. Experimental paraprotein neuropathy, demyelination by passive transfer of human IgM anti-myelin-associated glycoprotein. Ann Neurol. 1993;33:502–6.
Dalakas MC. Pathogenesis and treatment of anti-MAG neuropathy. Curr Treat Options Neurol. 2010;12:71–83.
Dalakas MC. Rituximab in anti-MAG neuropathy: more evidence for efficacy and more predictive factors. J Neurol Sci. 2017;377:224–6.
•• Dalakas MC. Advances in the diagnosis, immunopathogenesis and therapies of IgM-anti-MAG antibody-mediated neuropathies. Ther Adv Neurol Disord. 2018;11:1756285617746640. Detailed review on anti-MAG neuropathy.
Galassi G, Tondelli M, Ariatti A, Benuzzi F, Nichelli P, Valzania F. Long-term disability and prognostic factors in polyneuropathy associated with anti-myelin-associated glycoprotein (MAG) antibodies. Int J Neurosci. 2017;127:439–47.
Gazzola S, Delmont E, Franques J, Boucraut J, Salort-Campana E, Verschueren A, et al. Predictive factors of efficacy of rituximab in patients with anti-MAG neuropathy. J Neurol Sci. 2017;377:144–8.
Rakocevic G, Outschoorn UM, Dalakas M. Obinutuzumab (GAZYVA), a potent anti-B cell agent, in the treatment of rituximab-unresponsive IgM anti-myelin-associated-glycoprotein (MAG)-mediated-neuropathy (P1.450). Neurology. 2018;90:P1.450.
Mauermann ML, Sorenson EJ, Dispenzieri A, Mandrekar J, Suarez GA, Dyck PJ, et al. Uniform demyelination and more severe axonal loss distinguish POEMS syndrome from CIDP. J Neurol Neurosurg Psychiatry. 2012;83:480–6.
Li J, Zhang W, Jiao L, Duan M-H, Guan H-Z, Zhu W-G, et al. Combination of melphalan and dexamethasone for patients with newly diagnosed POEMS syndrome. Blood. 2011;117:6445–9.
Badros A, Porter N, Zimrin A. Bevacizumab therapy for POEMS syndrome. Blood. 2005;106:1135.
Chu BF, Shana’ah A, Hofmeister CC, Benson DM, Sell M, Tucker J, et al. Long-term therapy with lenalidomide in a patient with POEMS syndrome. Eur J Case Rep Intern Med. 2014;1:2014_000093.
Guimaraes-Costa R, Bombelli F, Leger J-M. Multifocal motor neuropathy. Curr Opin Neurol. 2013;26:503–9.
Traynor BJ, Codd MB, Corr B, Forde C, Frost E, Hardiman O. Amyotrophic lateral sclerosis mimic syndromes: a population-based study. Arch Neurol. 2000;57:109–13.
Vlam L, van den Berg LH, Cats EA, Piepers S, van der Pol W-L. Immune pathogenesis and treatment of multifocal motor neuropathy. J Clin Immunol. 2013;33(Suppl 1):S38–42.
Willison HJ, Yuki N. Peripheral neuropathies and anti-glycolipid antibodies. Brain. 2002;125:2591–625.
Uncini A, Santoro M, Corbo M, Lugaresi A, Latov N. Conduction abnormalities induced by sera of patients with multifocal motor neuropathy and anti-GM1 antibodies. Muscle Nerve. 1993;16:610–5.
Takigawa T, Yasuda H, Kikkawa R, Shigeta Y, Saida T, Kitasato H. Antibodies against GM1 ganglioside affect K+ and Na+ currents in isolated rat myelinated nerve fibers. Ann Neurol. 1995;37:436–42.
Kiernan MC, Guglielmi J, Kaji R, Murray NMF, Bostock H. Evidence for axonal membrane hyperpolarization in multifocal motor neuropathy with conduction block. Brain. 2002;125:664–75.
Uncini A, Kuwabara S. Nodopathies of the peripheral nerve: an emerging concept. J Neurol Neurosurg Psychiatry. 2015;86:1186–95.
van Schaik IN, Léger JM, Nobile-Orazio E, Cornblath DR, Hadden RD, Koski CL, et al. European Federation of Neurological Societies/peripheral nerve society guideline on management of multifocal motor neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society--first revisi. J Peripher Nerv Syst. 2010;15:295–301.
Carpo M, Cappellari A, Mora G, Pedotti R, Barbieri S, Scarlato G, et al. Deterioration of multifocal motor neuropathy after plasma exchange. Neurology. 1998;50:1480–2.
Cocito D, Merola A, Romagnolo A, Peci E, Toscano A, Mazzeo A, et al. Subcutaneous immunoglobulin in CIDP and MMN: a different long-term clinical response? J Neurol Neurosurg Psychiatry. 2016;87:791–3.
Kamiya-Matsuoka C, Shroff S, Gildersleeve K, Hormozdi B, Manning JT, Woodman KH. Neurolymphomatosis: a case series of clinical manifestations, treatments, and outcomes. J Neurol Sci. 2014;343:144–8.
Giometto B, Grisold W, Vitaliani R, Graus F, Honnorat J, Bertolini G. Paraneoplastic neurologic syndrome in the PNS Euronetwork database: a European study from 20 centers. Arch Neurol. 2010;67:330–5.
Gwathmey KG. Sensory neuronopathies. Muscle Nerve. 2016;53:8–19.
Darnell RB. Paraneoplastic neurologic disorders: windows into neuronal function and tumor immunity. Arch Neurol. 2004;61:30–2.
Rudnicki SA, Dalmau J. Paraneoplastic syndromes of the peripheral nerves. Curr Opin Neurol. 2005;18:598–603.
Antoine JC, Honnorat J, Camdessanche JP, Magistris M, Absi L, Mosnier JF, et al. Paraneoplastic anti-CV2 antibodies react with peripheral nerve and are associated with a mixed axonal and demyelinating peripheral neuropathy. Ann Neurol. 2001;49:214–21.
Muppidi S, Vernino S. Paraneoplastic neuropathies. Continuum (Minneap Minn). 2014;20:1359–72.
Koike H, Sobue G. Paraneoplastic neuropathy. Handb Clin Neurol. 2013;115:713–26.
Graus F, Keime-Guibert F, Reñe R, Benyahia B, Ribalta T, Ascaso C, et al. Anti-Hu-associated paraneoplastic encephalomyelitis: analysis of 200 patients. Brain. 2001;124:1138–48.
Gwathmey KG, Burns TM, Collins MP, Dyck PJB. Vasculitic neuropathies. Lancet Neurol. 2014;13:67–82.
Gorson KC. Therapy for vasculitic neuropathies. Curr Treat Options Neurol. 2006;8:105–17.
Levy Y, Uziel Y, Zandman GG, Amital H, Sherer Y, Langevitz P, et al. Intravenous immunoglobulins in peripheral neuropathy associated with vasculitis. Ann Rheum Dis. 2003;62:1221–3.
De Vita S, Quartuccio L, Isola M, Mazzaro C, Scaini P, Lenzi M, et al. A randomized controlled trial of rituximab for the treatment of severe cryoglobulinemic vasculitis. Arthritis Rheum. 2012;64:843–53.
Thaipisuttikul I, Chapman P, Avila EK. Peripheral neuropathy associated with ipilimumab: a report of 2 cases. J Immunother. 2015;38:77–9.
de Maleissye M-F, Nicolas G, Saiag P. Pembrolizumab-induced demyelinating polyradiculoneuropathy. N Engl J Med. 2016;375:296–7.
Uyanikoglu A, Ermis F, Akyuz F, Pinarbasi B, Baran B, Aydogan T, et al. Infliximab in inflammatory bowel disease: attention to adverse events. Eur Rev Med Pharmacol Sci. 2014;18:2337–42.
Seo B, Jeong YJ, Hong S, Kim Y-G, Lee C-K, Yoo B. A case of infliximab-induced multifocal motor neuropathy in a patient with rheumatoid arthritis and literature review. J Rheum Dis. 2016;23:250–5.
Stubgen J-P. Tumor necrosis factor-alpha antagonists and neuropathy. Muscle Nerve. 2008;37:281–92.
Kaltsonoudis E, Zikou AK, Voulgari PV, Konitsiotis S, Argyropoulou MI, Drosos AA. Neurological adverse events in patients receiving anti-TNF therapy: a prospective imaging and electrophysiological study. Arthritis Res Ther. 2014;16:R125.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Mithila Fadia and Sheetal Shroff each declare no potential conflicts of interest. Ericka Simpson has served on the speaker bureau for indications for subcutaneous IgG in CIDP patients sponsored by CSL Behring, Inc.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuromuscular Disorders
Rights and permissions
About this article
Cite this article
Fadia, M., Shroff, S. & Simpson, E. Immune-Mediated Neuropathies. Curr Treat Options Neurol 21, 28 (2019). https://doi.org/10.1007/s11940-019-0569-y
Published:
DOI: https://doi.org/10.1007/s11940-019-0569-y