Abstract
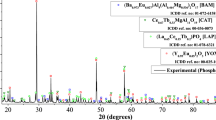
Waste fluorescent lamps containing a significantly high quantity of rare earth metals have great potential to be an unconventional source of critical metals if exploited efficiently for resource recovery. Therefore, the present study dealt with the selective leaching of red phosphor rare earths from waste fluorescent lamps. The parametric effects of the acid media and their concentrations, addition of H2O2, pulp density, temperature, and time were studied. The results revealed that 2.0 M HCl with 5 vol.% H2O2 yielded 100% yttrium and more than 95% europium compared to only 92% and 96% yttrium and 89% and 91% europium while using H2SO4 and HNO3, respectively. The green phosphor compounds Ce0.67Tb0.33MgAl11O19 and (La0.65Ce0.15Tb0.2)PO4 were undissolved in a residual mass that can be handled separately. Kinetics data followed logarithmic rate law, and the chemically-controlled mechanism was indicated by the values of apparent activation energy (i.e., Ea(Y), 87.8 kJ/mol and Ea(Eu), 54.1 kJ/mol).








Similar content being viewed by others
References
S. Ilyas, H. Kim, and R.R. Srivastava, Sep. Purif. Technol. 254, 117634 (2021).
F. Habashi, Extractive metallurgy of rare earths. Can. Metall. Quart. 52, 224 (2013).
S. Ilyas, H. Kim, R.R. Srivastava, and S. Choi, J. Clean. Prod. 278, 123435 (2021).
G. Prameswara, I. Trisnawati, P. Mulyono, A. Prasetya, and H.T.B.M. Petrus, JOM 73, 988 (2021).
G. Hearty, “Rare earths: next element in the trade war?” (Center for Strategic and International Studies, 2019). https://www.csis.org/analysis/rare-earths-next-element-trade-war#:%7e:text%3dWith%20the%20trade%20war%20having%2cnext%20salvo%20in%20the%20conflict.%26text%3dA1%253A%20Rare%252Dearth%20elements%20are%2cwhich%20possess%20similar%20chemical%20properties. Accessed 29 Sept 2021
S. Ilyas, H. Kim, and R.R. Srivastava, Sustainable Urban Mining of Precious Metals (CRC, Boca Raton, 2021).
J.-H. Lee, and J. Woo, Sustainability 12, 10191 (2020).
The EU Green Deal – a roadmap to sustainable economie (Swich2green), https://www.switchtogreen.eu/the-eu-green-deal-promoting-a-green-notable-circular-economy/. Accessed 29 Sept 2021
R. Galvin, and N. Healy, Energy Res. Soc. Sci. 67, 101529 (2020).
K. Binnemans, P.T. Jones, B. Blanpain, T. Van Gerven, Y. Yang, A. Walton, and M. Buchert, J. Clean. Prod. 51, 1 (2013).
Memoria anual 2016 (Ambilamp, 2017), https://ambilamp.es/sites/default/files/memoria-actividad/files/memoria2016-ambilamp.pdf. Accessed 29 Sept 2021
L. Yurramendi, L. Gijsemans, F. Forte, J.L. Aldana, C. del Río, and K. Binnemans, Hydrometallurgy 187, 38 (2019).
A. Miskufova, A. Kochmanova, T. Havlik, H. Horvathova, and P. Kuruc, Hydrometallurgy 176, 216 (2018).
N. Shukla, and N. Dhawan, JOM 73, 1090 (2021).
Q. Tan, J. Li, and X. Zeng, Crit. Rev. Environ. Sci. Technol. 45, 749 (2015).
M. Tanaka, K. Koyama, H. Narita, and T. Oishi, Recycling valuable metals via hydrometallurgical routes, in Design for Innovative Value Towards a Sustainable Society. ed. by M. Matsumoto, Y. Umeda, K. Masui, and S. Fukushige (Springer, Amsterdam, 2012), p. 507.
A. Tuncuk, V. Stazi, A. Akcil, E.Y. Yazici, and H. Deveci, Miner. Eng. 25, 28 (2012).
C.H. Yan, J.T. Jia, C.S. Liao, S. Wu, and G.X. Xu, Tsinghua. Sci. Technol. 11, 241 (2006).
S. Zielinski, and A. Szczepanik, Hydrometallurgy 33, 219 (1993).
R. Shimizu, K. Sawada, Y. Enokida, and I. Yamamoto, J. Supercrit. Fluids 33, 235 (2005).
Z. Hubicki, and M. Olszak, J. Chromatogr. A 955, 257 (2002).
H.Y. Liu, J. Chen, and D.Q. Li, Sep. Sci. Technol. 47, 223 (2012).
B.J. Beaudry, and K.A. Gschneidner Jr., Chapter 2, preparation and basic properties of the rare earth metals, in Handbook on the Physics and Chemistry of Rare Earths. ed. by K.A. Gschneidner Jr., and L. Eyring (Elsevier, New York, 1978), p. 173.
C.H. Huang, W. Wang, Y.J. Liu, and J.G. Wu, Inorganic Chemistry Series Volume VII: Scandium, Rare Earth Elements (Science Press, Beijing, 1992).
B.B. Mishra, N. Devi, and K. Sarangi, Miner. Eng. 136, 43 (2019).
L.V. Resende, and C.A. Morais, Miner. Eng. 70, 217 (2015).
S. Ilyas, H. Kim, and R.R. Srivastava, JOM 73, 19 (2021).
C.M. Lousada, M. Yang, K. Nilsson, and M. Jonsson, J. Mol. Catal. A-Chem. 379, 178 (2013).
V. Diesen, and M. Jonsson, J. Adv. Oxid. Technol. 16, 16 (2013).
C.M. Lousada, A.J. Johansson, T. Brinck, and M. Jonsson, J. Phys. Chem. C 116, 9533 (2012).
R. Sattar, S. Ilyas, S. Kousar, A. Khalid, M. Sajid, and S.I. Bhukhari, Environ. Eng. Res. 25, 88 (2020).
S. Ilyas, R.R. Srivastava, H. Kim, and H.A. Cheema, Sep. Purif. Technol. 248, 117029 (2020).
F. Habashi, Principles of Extractive Metallurgy: Hydrometallurgy, Gordon and Breach, vol I. (Science Publishers, New York, 1969).
O. Levenspiel, Chemical Reaction Engineering, 3rd edn. (Wiley, New York, 1999).
K. Chabhadiya, R.R. Srivastava, and P. Pathak, J. Environ. Chem. Eng. 9, 105232 (2021).
H. Munir, R.R. Srivastava, H. Kim, S. Ilyas, M.K. Khosa, and B. Yameen, J. Chem. Technol. Biotechnol. 95, 2286 (2020).
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (Project Nos. 2020R1A6A3A13073210 and 2020R1I1A1A01074249) and by the Brain Pool Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2019H1D3A2A02101993). The authors are thankful to Dr. Dipti Tanna for the language editing support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Choi, S., Ilyas, S. & Kim, H. Intensive Leaching of Red Phosphor Rare Earth Metals from Waste Fluorescent Lamp: Parametric Optimization and Kinetic Studies. JOM 74, 1054–1060 (2022). https://doi.org/10.1007/s11837-021-05112-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-021-05112-z