Abstract
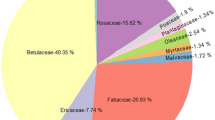
Botanical gardens could serve as pollinator-friendly gardens and are suitable locations for investigating plant–pollinator interactions. Pollination in botanical gardens is a challenging topic of research, and its ecological function remains unclear in terms of pollination management for both pollinators and plants. Objective monitoring methods—including the selection of insect taxa and analytical methods—are required to elucidate plant–pollinator interactions in botanical gardens. In this study, we evaluated pollen transport networks using generalist hoverflies, as they are important pollinator insects, generalist flower visitors, and heterospecific pollen vectors that carry pollen over large distances. Hoverflies can form generalized pollen transport networks with an adequate abundance and wide distribution, and are easily collectable using sticky traps. Pollen grains on hoverfly bodies were identified based on pollen DNA barcoding using the ITS, rbcL, and trn regions. Bipartite pollen transport networks in the study period in 2018 and 2019 consisted of 116 plant taxa that were significantly generalized and showed similar structures in both years. The dominant plant taxa were two invasive species—Solidago canadensis and Bidens sp.—which had a significantly high eigenvector centrality in unipartite pollen transport networks. Hoverfly-mediated pollen transfer distances were 116–131 m on average and were significantly shorter in heterogeneous open environments. These findings suggest that hoverflies are non-negligible actors in pollen transport networks in the studied botanical garden.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to sensitive geolocation information of plants but are available from the corresponding author on reasonable request.
Code availability
The statistical scripts generated during and/or analyzed during the current study are not publicly available due to sensitive geolocation information of plants but are available from the corresponding author on reasonable request.
References
Abhilasha D, Quintana N, Vivanco J, Joshi J (2008) Do allelopathic compounds in invasive Solidago canadensis s.l. restrain the native European flora? J Ecol 96:993–1001. https://doi.org/10.1111/j.1365-2745.2008.01413.x
Ashman TL, Arceo-Gómez G, Bennett JM, Knight TM (2020) Is heterospecific pollen receipt the missing link in understanding pollen limitation of plant reproduction? Am J Bot 107:845–847. https://doi.org/10.1002/ajb2.1477
Ashton PS (1988) Conservation of biological diversity in botanical gardens. In: Wilson EO, Peter FM (eds) Biodiversity. National Academy Press, Washington DC, pp 269–278
Baldock KCR, Goddard MA, Hicks DM et al (2019) A systems approach reveals urban pollinator hotspots and conservation opportunities. Nat Ecol Evol 3:363–373. https://doi.org/10.1038/s41559-018-0769-y
Bartomeus I, Vilà M, Santamaría L (2008) Contrasting effects of invasive plants in plant–pollinator networks. Oecologia 155:761–770. https://doi.org/10.1007/s00442-007-0946-1
Bell KL, De Vere N, Keller A, Richardson RT, Gous A, Burgess KS, Brosi JB (2016) Pollen DNA barcoding: current applications and future prospects. Genome 59:629–640. https://doi.org/10.1139/gen-2015-0200
Bhattacharya A (2010) Conservation of pollinator resources in botanic gardens. Our Nat 8:332–335. https://doi.org/10.3126/on.v8i1.4341
Blackmore LM, Goulson D (2014) Evaluating the effectiveness of wildflower seed mixes for boosting floral diversity and bumblebee and hoverfly abundance in urban areas. Insect Conserv Divers 7:480–484. https://doi.org/10.1111/icad.12071
Botanic Gardens Conservation International (2020) Botanic Gardens Conservation International education review: pollination and botanic gardens. Roots. New Holland Publishers, London
Branquart E, Hemptinne J-L (2000) Selectivity in the exploitation of floral resources by hoverflies (Diptera: Syrphinae). Ecography 23:732–742. https://doi.org/10.1111/j.1600-0587.2000.tb00316.x
Butts CT (2016) sna: tools for social network analysis. R package version 2.6. Available at https://CRAN.R-project.org/package=sna
Cavender N, Smith P, Marfleet K (2019) BGCI technical review: the role of botanic gardens in urban greening and conserving urban biodiversity. Botanic Gardens Conservation International, Richmond
Chen G, Sun W (2018) The role of botanical gardens in scientific research, conservation, and citizen science. Plant Divers 40:181–188. https://doi.org/10.1016/j.pld.2018.07.006
Chen TY, Chu CC, Fitzgerald G, Natwick E, Henneberry TJ (2004) Trap evaluations for thrips (Thysanoptera: Thripidae) and hoverflies (Diptera: Syrphidae). Environ Entomol 33:1416–1420. https://doi.org/10.1603/0046-225X-33.5.1416
Cranmer L, McCollin D, Ollerton J (2012) Landscape structure influences pollinator movements and directly affects plant reproductive success. Oikos 121:562–568. https://doi.org/10.1111/j.1600-0706.2011.19704.x
Csárdi G, Nepusz T (2006) The Igraph software package for complex network research. InterJ Complex Syst. https://doi.org/10.3724/sp.j.1087.2009.02191
Dormann CF, Frund J, Bluthgen N, Gruber B (2009) Indices, graphs and null models: analyzing bipartite ecological networks. Open Ecol J 2:7–24. https://doi.org/10.2174/1874213000902010007
Dormann CF, Fruend J, Gruber B et al (2020) Package ‘bipartite’
Dormann CF (2020) Using bipartite to describe and plot two-mode networks in R
Doyle T, Hawkes WLS, Massy R, Powney GD, Menz MHM, Wotton KR (2020) Pollination by hoverflies in the Anthropocene. Proc Biol Sci 287:20200508. https://doi.org/10.1098/rspb.2020.0508
Dylewski Ł, Maćkowiak Ł, Banaszak-Cibicka W (2019) Are all urban green spaces a favourable habitat for pollinator communities? Bees, butterflies and hoverflies in different urban green areas. Ecol Entomol 44:678–689. https://doi.org/10.1111/een.12744
Evans TM, Cavers S, Ennos R, Vanbergen AJ, Heard MS (2017) Florally rich habitats reduce insect pollination and the reproductive success of isolated plants. Ecol Evol 7:6507–6518. https://doi.org/10.1002/ece3.3186
Farine DR (2017) A guide to null models for animal social network analysis. Methods Ecol Evol 8:1309–1320. https://doi.org/10.1111/2041-210X.12772
Fenesi A, Vágási CI, Beldean M et al (2015) Solidago canadensis impacts on native plant and pollinator communities in different-aged old fields. Basic Appl Ecol 16:335–346. https://doi.org/10.1016/j.baae.2015.03.003
Fontaine C, Dajoz I, Meriguet J, Loreau M (2006) Functional diversity of plant-pollinator interaction webs enhances the persistence of plant communities. PLoS Biol 4:0129–0135. https://doi.org/10.1371/journal.pbio.0040001
Fukase J, Simons AM (2016) Increased pollinator activity in urban gardens with more native flora. Appl Ecol Environ Res 14:297–310. https://doi.org/10.15666/aeer/1401_297310
Gyan KY, Woodell SRJ (1987) Analysis of insect pollen loads and pollination efficiency of some common insect visitors of four species of woody Rosaceae. Funct Ecol 1:269–274. https://doi.org/10.2307/2389430
Hall DM, Camilo GR, Tonietto RK et al (2017) The city as a refuge for insect pollinators. Conserv Biol 31:24–29. https://doi.org/10.1111/cobi.12840
Hodgkiss D, Brown MJF, Fountain MT (2018) Syrphine hoverflies are effective pollinators of commercial strawberry. J Pollinat Ecol 22:55–66. https://doi.org/10.26786/1920-7603(2018)five
Hunneman H, Hoffmann F, Kwak MM (2004) The importance of syrphid flies as pollinators of Succisa pratensis (Dipsacaceae). Proc Neth Entomol Soc 15:53–58
Inouye DW, Larson BMH, Ssymank A, Kevan PG (2015) Flies and flowers III: ecology of foraging and pollination. J Pollinat Ecol 16:115–133. https://doi.org/10.26786/1920-7603(2015)15
Jauker F, Bondarenko B, Becker HC, Steffan-Dewenter I (2012) Pollination efficiency of wild bees and hoverflies provided to oilseed rape. Agric For Entomol 14:81–87. https://doi.org/10.1111/j.1461-9563.2011.00541.x
Johnson AL, Fetters AM, Ashman TL (2017) Considering the unintentional consequences of pollinator gardens for urban native plants: is the road to extinction paved with good intentions? New Phytol 215:1298–1305. https://doi.org/10.1111/nph.14656
Kéfi S, Miele V, Wieters EA, Navarrete SA, Berlow EL (2016) How structured is the entangled bank? The surprisingly simple organization of multiplex ecological networks leads to increased persistence and resilience. PLoS Biol 14:e1002527. https://doi.org/10.1371/journal.pbio.1002527
King C, Ballantyne G, Willmer PG (2013) Why flower visitation is a poor proxy for pollination: measuring single-visit pollen deposition, with implications for pollination networks and conservation. Methods Ecol Evol 4:811–818. https://doi.org/10.1111/2041-210X.12074
Klecka J, Hadrava J, Biella P, Akter A (2018) Flower visitation by hoverflies (Diptera: Syrphidae) in a temperate plant-pollinator network. PeerJ 6:e6025. https://doi.org/10.7717/peerj.6025
Levé M, Baudry E, Bessa-Gomes C (2019) Domestic gardens as favorable pollinator habitats in impervious landscapes. Sci Total Environ 647:420–430. https://doi.org/10.1016/j.scitotenv.2018.07.310
Lopezaraiza-Mikel ME, Hayes RB, Whalley MR, Memmott J (2007) The impact of an alien plant on a native plant–pollinator network: an experimental approach. Ecol Lett 10:539–550. https://doi.org/10.1111/j.1461-0248.2007.01055.x
Loughnan D, Thompson JD, Ogilvie JE, Gilbert B (2014) Taraxacum officinale pollen depresses seed set of montane wildflowers through pollen allelopathy. J Pollinat Ecol 13:146–150. https://doi.org/10.26786/1920-7603(2014)13
Lozada-Gobilard S, Pánková H, Zhu J et al (2020) Potential risk of interspecific hybridization in ex situ collections. J Nat Conserv 58:125912. https://doi.org/10.1016/j.jnc.2020.125912
Lucas A, Bodger O, Brosi BJ, Ford CR, Forman DW, Greig C, Hegarty M, Neyland PJ, de Vere N (2018) Generalisation and specialisation in hoverfly (Syrphidae) grassland pollen transport networks revealed by DNA metabarcoding. J Anim Ecol 87:1008–1021. https://doi.org/10.1111/1365-2656.12828
Majewska AA, Altizer S (2020) Planting gardens to support insect pollinators. Conserv Biol 34:15–25. https://doi.org/10.1111/cobi.13271
Mayer C, Van Rossum F, Jacquemart AL (2012) Evaluating pollen flow indicators for an insect-pollinated plant species. Basic Appl Ecol 13:690–697. https://doi.org/10.1016/j.baae.2012.09.012
Morales CL, Traveset A (2008) Interspecific pollen transfer: magnitude, prevalence and consequences for plant fitness. Crit Rev Plant Sci 27:221–238. https://doi.org/10.1080/07352680802205631
Murphy SD (2001) The role of pollen allelopathy in weed ecology. Weed Technol 15:867–872. https://doi.org/10.1614/0890-037X(2001)015[0867:TROPAI]2.0.CO;2
Murphy SD, Aarssen LW (1995) Allelopathic pollen extract from Phleum pratense L. (Poaceae) reduces germination, in vitro, of pollen of sympatric species. Int J Plant Sci 156:435–444. https://doi.org/10.1111/j.1469-8137.1989.tb02385.x
National Institute for Environmental Studies (2020) Invasive species of Japan. Available at https://www.nies.go.jp/biodiversity/invasive/DB/etoc8_plants.html. Accessed 2 May 2020
Parra-Tabla V, Ashman T, Raguso R, Albor C, Sosenski P, Carmona D, Arceo-Gómez G (2021) Pollen transfer networks reveal alien species as main heterospecific pollen donors with fitness consequences for natives. J Ecol 109:939–951. https://doi.org/10.1111/1365-2745.13520
Pornon A, Escaravage N, Burrus M et al (2016) Using metabarcoding to reveal and quantify plant-pollinator interactions. Sci Rep 6:27282. https://doi.org/10.1038/srep27282
Pornon A, Andalo C, Burrus M, Escaravage N (2017) DNA metabarcoding data unveils invisible pollination networks. Sci Rep 7:16828. https://doi.org/10.1038/s41598-017-16785-5
Powney GD, Carvell C, Edwards M, Morris RKA, Roy HE, Woodcock BA, Isaac NJB (2019) Widespread losses of pollinating insects in Britain. Nat Commun 10:1018. https://doi.org/10.1038/s41467-019-08974-9
QGIS Development Team (2020) QGIS Geographic Information System. Available at https://www.qgis.org
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rader R, Howlett BG, Cunningham SA et al (2009) Alternative pollinator taxa are equally efficient but not as effective as the honeybee in a mass flowering crop. J Appl Ecol 46:1080–1087. https://doi.org/10.1111/j.1365-2664.2009.01700.x
Rader R, Edwards W, Westcott DA, Cunningham SA, Howlett BG (2011) Pollen transport differs among bees and flies in a human-modified landscape. Divers Distrib 17:519–529. https://doi.org/10.1111/j.1472-4642.2011.00757.x
Razanajatovo M, Föhr C, Fischer M, Prati D, van Kleunen M (2015) Non-naturalized alien plants receive fewer flower visits than naturalized and native plants in a Swiss botanical garden. Biol Conserv 182:109–116. https://doi.org/10.1016/j.biocon.2014.11.043
Rivera-Hutinel A, Bustamante R, Marín V, Medel A (2012) Effects of sampling completeness on the structure of plant-pollinator networks. Ecology 93:1593–1603
Rollings R, Goulson D (2019) Quantifying the attractiveness of garden flowers for pollinators. J Insect Conserv 23:803–817. https://doi.org/10.1007/s10841-019-00177-3
Rossi J, Gamba U, Pinna M, Spagnolo S, Visentin C, Alma A (2006) Hoverflies in organic apple orchards in north-western Italy. Bull Insectol 59:111–114
Russo L, Debarros N, Yang S, Shea K, Mortensen D (2013) Supporting crop pollinators with floral resources: network–based phenological matching. Ecol Evol 3:3125–3140. https://doi.org/10.1002/ece3.703
Sarthou JP, Ouin A, Arrignon F, Barreau G, Bouyjou B (2005) Landscape parameters explain the distribution and abundance of Episyrphus balteatus (Diptera: Syrphidae). Eur J Entomol 102:539–545
Soderstrom M (2008) Botanical gardens. In: Jørgensen SW, Fath BD (eds) Encyclopedia of ecology, 1st edn. Elsevier, Oxford, pp 495–502. https://doi.org/10.1016/B978-008045405-4.00320-7
Stan Development Team (2018a) RStan: the R interface to Stan. R Package version 2173
Stan Development Team (2018b) Stan modeling language users guide and reference manual. Version 2180
Stevens GA, Tang CS (1985) Inhibition of seedling growth of crop species by recirculating root exudates of Bidens pilosa L. J Chem Ecol 11:1411–1425. https://doi.org/10.1007/BF01012141
Suyama Y (2011) Procedure for single-pollen genotyping. In: Isagi Y, Suyama Y (eds) Single-pollen genotyping. Ecological research monographs. Springer, Tokyo, pp 7–15
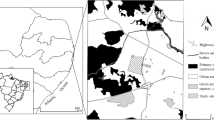
Tsukuba Botanical Garden (2019) A list of vascular plants in Tsukuba Botanical Garden 2018. Bull Natl Museum Nat Sci 45:1–270
Vamosi JC, Gong YB, Adamowicz SJ, Packer L (2017) Forecasting pollination declines through DNA barcoding: the potential contributions of macroecological and macroevolutionary scales of inquiry. New Phytol 214:11–18. https://doi.org/10.1111/nph.14356
van Rossum F, Stiers I, van Geert A, Triest L, Hardy OJ (2011) Fluorescent dye particles as pollen analogues for measuring pollen dispersal in an insect-pollinated forest herb. Oecologia 165:663–674. https://doi.org/10.1007/s00442-010-1745-7
Van Rossum F, Triest L (2010) Pollen dispersal in an insect-pollinated wet meadow herb along an urban river. Landsc Urban Plan 95:201–208. https://doi.org/10.1016/j.landurbplan.2010.01.004
Villa M, Santos SAP, Marrão R et al (2016) Syrphids feed on multiple patches in heterogeneous agricultural landscapes during the autumn season, a period of food scarcity. Agric Ecosyst Environ 233:262–269. https://doi.org/10.1016/j.agee.2016.09.014
Wang J, Qigang Y, Ming K, Huang H (2008) Novel polymorphic microsatellite loci and patterns of pollen-mediated gene flow in an ex situ population of Eurycorymbus cavaleriei (Sapindaceae) as revealed by categorical paternity analysis. Conserv Genet 9:559–567. https://doi.org/10.1007/s10592-007-9369-0
Wojcik VA, Buchmann S (2012) A review of pollinator conservation and management on infrastructure supporting rights-of-way. J Poll Ecol 7:16–26. https://doi.org/10.26786/1920-7603(2012)5
Wotton KR, Gao B, Menz MHM, Morris RKA, Ball SG, Lim KS, Reynolds DR, Hu G, Chapman JW (2019) Mass seasonal migrations of hoverflies provide extensive pollination and crop protection services. Curr Biol 29(2167–2173):E5. https://doi.org/10.1016/j.cub.2019.05.036
Wray JC, Elle E (2014) Flowering phenology and nesting resources influence pollinator community composition in a fragmented ecosystem. Landsc Ecol 30:261–272. https://doi.org/10.1007/s10980-014-0121-0
Ye QG, Yao XH, Zhang SJ, Kang M, Huang HW (2006) Potential risk of hybridization in ex situ collections of two endangered species of Sinojackia Hu (Styracaceae). J Integr Plant Biol 48:867–872. https://doi.org/10.1111/j.1744-7909.2006.00260.x
Zhang JJ, Ye QG, Yao XH, Huang HW (2010) Spontaneous interspecific hybridization and patterns of pollen dispersal in ex situ populations of a tree species (Sinojackia xylocarpa) that is extinct in the wild. Conserv Biol 24:246–255. https://doi.org/10.1111/j.1523-1739.2009.01357.x
Acknowledgements
We thank Kanoko Takarabe and Kyoka Tsuda for their help with conducting the research. We also wish to thank Editage (www.editage.com) for English language editing. This work was supported by the Eco-Ibaraki Environmental Conservation Fund (Grant No. 19-70).
Author information
Authors and Affiliations
Contributions
HY: Conceptualization, HY: Data curation, HY: Formal analysis, HY: Funding acquisition, HY and IC: Investigation, HY and IC: Methodology; HY: Project administration; TN: Resources; HY: Software; TN and KT: Supervision; HY: Validation; HY: Visualization; HY: Roles/Writing-original draft; TN and KT: Writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest to disclose.
Additional information
Handling Editor: Isabel Alves dos Santos.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Horiuchi, Y., Ishii, C., Kamijo, T. et al. Generalist flower-visiting hoverflies as non-negligible actors in pollen transport network in a botanical garden: verification of a hoverfly pollen transport network using pollen DNA barcoding. Arthropod-Plant Interactions 16, 171–181 (2022). https://doi.org/10.1007/s11829-022-09896-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-022-09896-y