Abstract
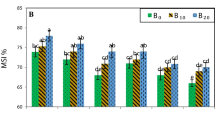
Salt stress is one of the important abiotic factors responsible for inhibiting the growth and development of commercially important crops. The demand for salt-tolerant varieties is growing tremendously with increasing soil salinity. In the present study, the effect of salt stress on four commercially important potato cultivars [‘Kufri Chipsona-1’ (CS-1), ‘Kufri Chandramukhi’ (KCM), ‘Kufri Pukhraj’ (KP), and ‘Kufri Jyoti’ (KJ)] was examined to identify the salt-tolerant levels using in vitro screening. The nodes from microshoots of these cultivars were cultured for 40 days on MS1 medium (basal MS medium + 10 µM AgNO3) containing 0, 100, 200, and 300 mM NaCl. Compared to the control, the morpho-physiological traits varied significantly with maximum growth reduction in cultivar ‘KJ’ cultivar and minimum in ‘CS-1’. The cultivar ‘CS-1’ showed no sign of leaf senescence and remained green on all the salt concentrations, while the other three cultivars (‘KCM’, ‘KP’, and ‘KJ’) turned brown and did not tolerate higher (300 mM) NaCl concentration. Moreover, cultivar ‘CS-1’ was found to have high chlorophyll, carotenoid, proline, sugars, and soluble protein contents on a medium containing 300 mM NaCl compared to the other three cultivars. As part of the antioxidant defense system, the analysis of various enzymes (peroxidase, ascorbate peroxidase, catalase, and superoxide dismutase) also showed a significant increase in their activity with an increasing NaCl concentration in cultivar ‘CS-1’. The study concluded that cultivar ‘CS-1’ possessed a higher level of salt tolerance and could adapt to saline environment. We can further conclude that the accumulation of osmolytes and increased antioxidant enzyme activities have an important role in imparting salt tolerance to the plants. Therefore, the outcomes of the study can be utilized as a platform to develop screening strategies for the improvement of commercially important crops.




Similar content being viewed by others
Availability of data and material
Authors confirm that data and material used in this study support our publication claims.
Code availability
Not Applicable.
References
Acosta-Motos JR, Ortuño MF, Bernal-Vicente A, Diaz-Vivancos P, Sanchez-Blanco MJ, Hernandez JA (2017) Plant responses to salt stress: adaptive mechanisms. Agronomy 7:18. https://doi.org/10.3390/agronomy7010018
Aebi H (1984) Catalase in vitro. In: Methods in enzymology, vol 105. Elsevier, pp 121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Amini F, Ehsanpour AA (2005) Soluble proteins, proline, carbohydrates and Na+/K+ changes in two tomato (Lycopersicon esculentum Mill.) cultivars under in vitro salt stress. Am J Biochem Biotechnol 1:204–208. https://doi.org/10.3844/ajbbsp.2005.204.208
Armstrong MR, Vossen J, Lim TY, Hutten RC, Xu J, Strachan SM, Harrower B, Champouret N, Gilroy EM, Hein I (2019) Tracking disease resistance deployment in potato breeding by enrichment sequencing. Plant Biotechnol J 17:540–549. https://doi.org/10.1111/pbi.12997
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Ayala-Astorga GI, Alcaraz-Meléndez L (2010) Salinity effects on protein content, lipid peroxidation, pigments, and proline in Paulownia imperialis (Siebold & Zuccarini) and Paulownia fortunei (Seemann & Hemsley) grown in vitro. Electron J Biotechnol 13:13–14. https://doi.org/10.2225/vol13-issue5-fulltext-13
Azooz M, Ismail A, Elhamd MA (2009) Growth, lipid peroxidation and antioxidant enzyme activities as a selection criterion for the salt tolerance of maize cultivars grown under salinity stress. Int J Agric Biol 11:21–26
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Benavídes MP, Marconi PL, Gallego SM, Comba ME, Tomaro ML (2000) Relationship between antioxidant defence systems and salt tolerance in Solanum tuberosum. Funct Plant Biol 27:273–278. https://doi.org/10.1071/PP99138
Cha-Um S, Charoenpanich A, Roytrakul S, Kirdmanee C (2009) Sugar accumulation, photosynthesis and growth of two indica rice varieties in response to salt stress. Acta Physiol Plant 31:477–486. https://doi.org/10.1007/s11738-008-0256-1
Dahal K, Li X-Q, Tai H, Creelman A, Bizimungu B (2019) Improving potato stress tolerance and tuber yield under a climate change scenario–a current overview. Front Plant Sci 10:563. https://doi.org/10.3389/fpls.2019.00563
de Azevedo Neto AD, Prisco JT, Enéas-Filho J, de Abreu CEB, Gomes-Filho E (2006) Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ Exper Bot 56:87–94. https://doi.org/10.1016/j.envexpbot.2005.01.008
Doganlar ZB, Demir K, Basak H, Gul I (2010) Effects of salt stress on pigment and total soluble protein contents of three different tomato cultivars. Afr J Agric Res 5:2056–2065. https://doi.org/10.5897/AJAR10.258
Dubois M, Gilles KA, Hamilton JK, Rebers PT, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Gopal J, Iwama K (2007) In vitro screening of potato against water-stress mediated through sorbitol and polyethylene glycol. Plant Cell Rep 26:693–700. https://doi.org/10.1007/s00299-006-0275-6
Gupta VK, Luthra SK, Singh BP (2015) Storage behaviour and cooking quality of Indian potato varieties. J Food Sci Technol 52:4863–4873. https://doi.org/10.1007/s13197-014-1608-z
Hasanuzzaman M, Bhuyan M, Zulfiqar F, Raza A, Mohsin SM, Mahmud JA, Fujita M, Fotopoulos V (2020) Reactive oxygen species and antioxidant defense in plants under abiotic stress: Revisiting the crucial role of a universal defense regulator. Antioxidants 9:681. https://doi.org/10.3390/antiox9080681
Hasanuzzaman M, Hossain MA, da Silva JAT, Fujita M (2012) Plant response and tolerance to abiotic oxidative stress: antioxidant defense is a key factor. In: Crop stress and its management: Perspectives and strategies. Springer, pp 261–315. https://doi.org/10.1007/978-94-007-2220-0_8
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7:1456–1466. https://doi.org/10.4161/psb.21949
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hernández JA, Almansa MS (2002) Short-term effects of salt stress on antioxidant systems and leaf water relations of pea leaves. Physiol Plant 115:251–257. https://doi.org/10.1034/j.1399-3054.2002.1150211.x
Isayenkov SV, Maathuis FJ (2019) Plant salinity stress: many unanswered questions remain. Front Plant Sci 10:80. https://doi.org/10.3389/fpls.2019.00080
Jaarsma R, de Vries RS, de Boer AH (2013) Effect of salt stress on growth, Na+ accumulation and proline metabolism in potato (Solanum tuberosum) cultivars. PLoS ONE 8:e60183. https://doi.org/10.1371/journal.pone.0060183
Kaur A, Reddy MS, Kumar A (2017) Efficient, one step and cultivar independent shoot organogenesis of potato. Physiol Mol Biol Plants 23:461–469. https://doi.org/10.1007/s12298-017-0418-y
Kaur A, Reddy MS, Kumar A (2018a) Direct somatic embryogenesis of potato [Solanum tuberosum (L.)] cultivar ‘Kufri Chipsona 2.’ Plant Cell Tissue Organ Cult 134:457–466. https://doi.org/10.1007/s11240-018-1435-4
Kaur A, Singh D, Gupta N, Kumar A (2018b) In Vitro Propagation of Important Rootstocks of Apple for Rapid Cloning and Improvement. In: Biotechnologies of Crop Improvement, Volume 1. Springer, pp 215–241. https://doi.org/10.1007/978-3-319-78283-6_6
Kaymakanova M, Stoeva N (2008) Physiological reaction of bean plants (Phaseolus vulg. L.) to salt stress. Gen Appl Plant Physiol Special 34:177–188
Kerepesi I, Galiba G (2000) Osmotic and salt stress-induced alteration in soluble carbohydrate content in wheat seedlings. Crop Sci 40:482–487. https://doi.org/10.2135/cropsci2000.402482x
Kumar A, Palni L, Nandi S (2003) The effect of light source and gelling agent on micropropagation of Rosa damascena Mill. and Rhynchostylis retusa (L.) Bl. J Hortic Sci Biotech 78:786–792. https://doi.org/10.1080/14620316.2003.11511700
Londhe S (2016) Cultivation of potato on abiotic stress-affected soils of India. Curr Sci 111:21
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
McEwen Jr CM (1971) Monoamine oxidase [rabbit serum]. In: Methods in enzymology, vol 17. Elsevier, pp 686–692. https://doi.org/10.1016/0076-6879(71)17119-4
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Ouzounidou G, Giannakoula A, Ilias I, Zamanidis P (2016) Alleviation of drought and salinity stresses on growth, physiology, biochemistry and quality of two Cucumis sativus L. cultivars by Si application. Braz J Bot 39:531–539. https://doi.org/10.1007/s40415-016-0274-y
Ouzounidou G, Ilias I, Giannakoula A, Theoharidou I (2014) Effect of water stress and NaCl triggered changes on yield, physiology, biochemistry of broad bean (Vicia faba) plants and on quality of harvested pods. Biologia 69:1010–1017. https://doi.org/10.2478/s11756-014-0397-1
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60:324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Passamani LZ, Barbosa RR, Reis RS, Heringer AS, Rangel PL, Santa-Catarina C, Grativol C, Veiga CF, Souza-Filho GA, Silveira V (2017) Salt stress induces changes in the proteomic profile of micropropagated sugarcane shoots. PLoS ONE 12:e0176076. https://doi.org/10.1371/journal.pone.0176076
Qados AMA (2011) Effect of salt stress on plant growth and metabolism of bean plant Vicia faba (L.). J Saudi Soc Agric Sci 10:7–15. https://doi.org/10.1016/j.jssas.2010.06.002
Queirós F, Rodrigues JA, Almeida JM, Almeida DP, Fidalgo F (2011) Differential responses of the antioxidant defence system and ultrastructure in a salt-adapted potato cell line. Plant Physiol Biochem 49:1410–1419. https://doi.org/10.1016/j.plaphy.2011.09.020
Radi AA, Farghaly FA, Hamada AM (2013) Physiological and biochemical responses of salt-tolerant and salt-sensitive wheat and bean cultivars to salinity. J Biol Earth Sci 3:72–88
Rai MK, Kalia RK, Singh R, Gangola MP, Dhawan A (2011) Developing stress tolerant plants through in vitro selection—an overview of the recent progress. Environ Exper Bot 71:89–98. https://doi.org/10.1016/j.envexpbot.2010.10.021
Rana RK, Anwer M (2018) Potato production scenario and analysis of its total factor productivity in India. Indian J Agr Sci 88:1354–1361
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–26. https://doi.org/10.1155/2012/217037
Singh D, Kaur S, Kumar A (2020) In vitro drought tolerance in selected elite clones of Eucalyptus tereticornis Sm. Acta Physiol Plant 42:17. https://doi.org/10.1007/s11738-019-3009-4
Singh D, Kumar A (2021a) In Vitro Screening and Characterization of Selected Elite Clones of Eucalyptus tereticornis Sm. for Salt Stress. J Plant Growth Regul 40:694–706. https://doi.org/10.1007/s00344-020-10138-9
Singh D, Kumar A (2021b) Multivariate screening approach indicated adaptive tolerance to salt stress in the seedlings of an agroforestry tree, Eucalyptus tereticornis Sm. Plant Cell Tissue Organ Cult 145:545–560. https://doi.org/10.1007/s11240-021-02025-2
Singh D, Sillu D, Kumar A, Agnihotri S (2021) Dual nanozyme characteristics of iron oxide nanoparticles alleviate salinity stress and promote the growth of an agroforestry tree, Eucalyptus tereticornis Sm. Environ Sci-Nano 8:1308–1325. https://doi.org/10.1039/D1EN00040C
Slama I, Abdelly C, Bouchereau A, Flowers T, Savoure A (2015) Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann Bot 115:433–447. https://doi.org/10.1093/aob/mcu239
Sumner JB, Howell SF (1935) A method for determination of saccharase activity. J Biol Chem 108:51–54. https://doi.org/10.1016/S0021-9258(18)75307-6
Zinnah K, Zobayer N, Sikdar SU, Liza LN, Chowdhury MAN, Ashrafuzzaman M (2013) In vitro regeneration and screening for salt tolerance in rice (Oryza sativa L.). Int Res J Biol Sci 2:29–36
Acknowledgements
Authors are thankful to TIFAC-CORE, Thapar Institute of Engineering & Technology for providing all required facilities and Council of Scientific.
Funding
This work was funded by the Council of Scientific and Industrial Research, Extramural Research Division, New Delhi through the Project 38(1465)/18/EMR-II.
Author information
Authors and Affiliations
Contributions
DS, AK and MK conducted experiments, DS and AK compiled data and wrote the initial draft of the manuscript, Anil Kumar finalized the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not Applicable.
Consent to participate
Not Applicable.
Consent for publication
Authors give their consent to publish the results and data reported in this study.
Conflict of Interests
On behalf of all the authors, I confirm that there is no conflict of interest in publication of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, D., Kaur, A., Kaur, M. et al. In vitro screening of Indian potato cultivars for the salt stress and associated physio-biochemical changes. Biologia 77, 627–639 (2022). https://doi.org/10.1007/s11756-021-00949-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00949-w