Abstract
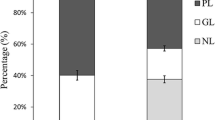
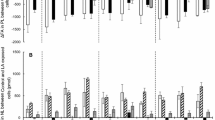
sn-2-Monoacylglycerol (2-MAG) and sn-1(3)-monoacylglycerol [1(3)-MAG] are important but yet little studied intermediates in lipid metabolism. The current study compared the metabolic fate of 2-MAG and 1(3)-MAG in isolated caecal enterocytes and hepatocytes of brown trout (Salmo trutta). 1(3)-Oleoyl [9,10-3H(N)]-glycerol and 2-Oleoyl [9,10-3H(N)]-glycerol were prepared by pancreatic lipase digestion of triolein [9,10-3H(N)]. The 1(3)-MAG and 2-MAG were efficiently absorbed by enterocytes and hepatocytes at similar rates. The 2-MAG was quickly resynthesized into TAG through the monoacylglycerol acyltransferase (EC: 2.3.1.22, MGAT) pathway in both tissues, whereas 1(3)-MAG was processed into TAG and phospholipids at a much slower rate, suggesting 2-MAG was the preferred substrates for MGAT. Further analysis showed that 1(3)-MAG was synthesized into 1,3-DAG, but there were no accumulation of 1,3-DAG in either enterocytes or hepatocytes, which contrasts that of mammalian studies. Some of the 1(3)-MAG may be acylated to 1,2(2,3)-DAG and then utilized for TAG synthesis. Alternatively, 1(3)-MAG can be hydrolyzed to free fatty acid and glycerol, and re-synthesized into TAG through the glycerol-3-phosphate (Gro-3-P) pathway. The overall data suggested that the limiting step of the intracellular 1(3)-MAG metabolism is the conversion of 1(3)-MAG itself.






Similar content being viewed by others
Abbreviations
- 1(3)-MAG:
-
sn-1(3)-Monoacylglycerol
- 1,2(2,3)-DAG:
-
sn-1,2(2,3)-Diacylglycerol
- 1,3-DAG:
-
sn-1,3-Diacylglycerol
- 1-MAG:
-
sn-1-Monoacylglycerol
- 2-MAG:
-
sn-2-Monoacylglycerol
- BSDL:
-
Bile salt-dependent lipase
- DAG:
-
Diacylglycerol(s)
- DGAT:
-
Diacylglycerol acyltransferase
- FAF-BSA:
-
Fatty acid free bovine serum albumin
- FFA:
-
Unesterified fatty acids
- Gro-3-P:
-
Glycerol-3-phosphate
- HBSS:
-
Hank’s balanced salt solution
- HPTLC:
-
High performance thin layer chromatography
- lysoPtdOH:
-
Lysophosphatidic acid
- MAG:
-
Monoacylglycerol(s)
- MEM:
-
Minimum essential medium
- MGAT:
-
Monoacylglycerol acyltransferase
- MGL:
-
Monoacylglycerol lipase
- MS222:
-
Tricaine methanesulfonate
- PtdCho:
-
Phosphatidylcholine
- PtdEtn:
-
Phosphatidylethanolamine
- PtdIns:
-
Phosphatidylinositol
- PtdOH:
-
Phosphatidic acid
- PtdSer:
-
Phosphatidylserine
- TAG:
-
Triacylglycerol(s)
References
Decaro A, Figarella C, Amic J, Michel R, Guy O (1977) Human pancreatic lipase—glycoprotein. Biochim Biophys Acta 490:411–419
Mattson FH, Volpenhein RA (1964) The digestion and absorption of triglycerides. J Biol Chem 239:2772–2777
Mattson FH, Volpenhein RA (1968) Hydrolysis of primary and secondary esters of glycerol by pancreatic juice. J Lipid Res 9:79–84
Wang CS, Kuksis A, Manganaro F, Myher JJ, Downs D, Bass HB (1983) Studies on the substrate specificity of purified human milk bile salt-activated lipase. J Biol Chem 258:9197–9202
Lombardo D (2001) Bile salt-dependent lipase: its pathophysiological implications. BBA-Mol Cell Biol Lipids 1533:1–28
Olsen RE, Ringø E (1997) Lipid digestibility in fish: a review. Recent Res Dev Lipid Res 1:199–265
Tocher DR, Sargent JR (1984) Studies on triacylglycerol, wax ester and sterol ester hydrolases in intestinal ceca of rainbow trout (Salmo gairdneri) fed diets rich in triacylglycerols and wax esters. Comp Biochem Physio 77:561–571
Iijima N, Tanaka S, Ota Y (1998) Purification and characterization of bile salt-activated lipase from the hepatopancreas of red sea bream, Pagrus major. Fish Physiol Biochem 18:59–69
Bogevik AS, Tocher DR, Waagbo R, Olsen RE (2008) Triacylglycerol-, wax ester- and sterol ester-hydrolases in midgut of Atlantic salmon (Salmo salar). Aquac Nutr 14:93–98
Bogevik AS, Oxley A, Olsen RE (2008) Hydrolysis of acyl-homogeneous and fish oil triacylglycerols using desalted midgut extract from Atlantic salmon, Salmo salar. Lipids 43:655–662
Gjellesvik DR (1991) Fatty acid specificity of bile salt-dependent lipase enzyme recognition and super-substrate effects. Biochim Biophys Acta 1086:167–172
Gjellesvik DR, Lombardo D, Walther BT (1992) Pancreatic bile salt dependent lipase from cod (Gadus morhua) purification and properties. Biochim Biophys Acta 1124:123–134
Lehner R, Kuksis A (1996) Biosynthesis of triacylglycerols. Prog Lipid Res 35:169–201
Oxley A, Torstensen BE, Rustan AC, Olsen RE (2005) Enzyme activities of intestinal triacylglycerol and phosphatidylcholine biosynthesis in Atlantic salmon (Salmo salar L.). Comp Biochem Physiol B-Biochem Mol Biol 141:77–87
Oxley A, Jutfelt F, Sundell K, Olsen RE (2007) Sn-2-monoacylglycerol, not glycerol, is preferentially utilised for triacylglycerol and phosphatidylcholine biosynthesis in Atlantic salmon (Salmo salar L.) intestine. Comp Biochem Physiol B-Biochem Mol Biol 146:115–123
Caballero MJ, Gallardo G, Robaina L, Montero D, Fernandez A, Izquierdo M (2006) Vegetable lipid sources affect in vitro biosynthesis of triacylglycerols and phospholipids in the intestine of sea bream (Sparus aurata). Br J Nutr 95:448–454
Li KS, Olsen RE, Ostensen MA, Altin D, Kjorsvik E, Olsen Y (2016) Atlantic cod (Gadus morhua) larvae can biosynthesis phospholipid de novo from 2-oleoyl-glycerol and glycerol precursors. Fish Physiol Biochem 42:137–147
Senior JR, Isselbacher KJ (1963) Demonstration of an intestinal monoglyceride lipase: an enzyme with a possible role in the intracellular completion of fat digestion. J Clin Invest 42:187–195
De Jong B, Hülsmann W (1978) Monoacylglycerol hydrolase activity of isolated rat small intestinal epithelial cells. BBA-Lipid Lipid Metab 528:36–46
Yen CLE, Farese RV (2003) MGAT2, a monoacylglycerol acyltransferase expressed in the small intestine. J Biol Chem 278:18532–18537
Cao JS, Lockwood J, Burn P, Shi YG (2003) Cloning and functional characterization of a mouse intestinal Acyl-CoA: monoacylglycerol acyltransferase, MGAT2. J Biol Chem 278:13860–13866
Kondo H, Hase T, Murase T, Tokimitsu I (2003) Digestion and assimilation features of dietary DAG in the rat small intestine. Lipids 38:25–30
Murase T, Aoki M, Wakisaka T, Hase T, Tokimitsu I (2002) Anti-obesity effect of dietary diacylglycerol in C57BL/6 J mice: dietary diacylglycerol stimulates intestinal lipid metabolism. J Lipid Res 43:1312–1319
Hibi M, Takase H, Meguro S, Tokimitsu I (2009) The effects of diacylglycerol oil on fat oxidation and energy expenditure in humans and animals. BioFactors 35:175–177
Tocher DR, Fonseca-Madrigal J, Bell JG, Dick JR, Henderson RJ, Sargent JR (2002) Effects of diets containing linseed oil on fatty acid desaturation and oxidation in hepatocytes and intestinal enterocytes in Atlantic salmon (Salmo salar). Fish Physiol Biochem 26:157–170
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Myher J, Kuksis A (1979) Stereospecific analysis of triacylglycerols via racemic phosphatidylcholines and phospholipase C. Can J Biochem 57:117–124
Thomas A III, Scharoun J, Ralston H (1965) Quantitative estimation of isomeric monoglycerides by thin-layer chromatography. J Am Oil Chem Soc 42:789–792
Christie W (2003) Lipid analysis, isolation, separation and structural analysis of lipids. J Barnes and Associates, Dundee
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Olsen RE, Henderson RJ (1989) The rapid analysis of neutral and polar marine lipids using double-development HPTLC and scanning densitometry. J Exp Mar Biol Ecol 129:189–197
Ho SY, Delgado L, Storch J (2002) Monoacylglycerol metabolism in human intestinal Caco-2 cells—evidence for metabolic compartmentation and hydrolysis. J Biol Chem 277:1816–1823
Oxley A, Tocher DR, Torstensen BE, Olsen RE (2005) Fatty acid utilisation and metabolism in caecal enterocytes of rainbow trout (Oncorhynchus mykiss) fed dietary fish or copepod oil. BBA-Mol Cell Biol Lipids 1737:119–129
Diaz-Lopez M, Perez MJ, Acosta NG, Jerez S, Dorta-Guerra R, Tocher DR, Lorenzo A, Rodriguez C (2010) Effects of dietary fish oil substitution by Echium oil on enterocyte and hepatocyte lipid metabolism of gilthead seabream (Sparus aurata L.). Comp Biochem Physiol B-Biochem Mol Biol 155:371–379
Hiramine Y, Emoto H, Takasuga S, Hiramatsu R (2010) Novel acyl-coenzyme A: monoacylglycerol acyltransferase plays an important role in hepatic triacylglycerol secretion. J Lipid Res 51:1424–1431
Young SG, Zechner R (2013) Biochemistry and pathophysiology of intravascular and intracellular lipolysis. Genes Dev 27:459–484
Chon SH, Zhou YX, Dixon JL, Storch J (2007) Intestinal monoacylglycerol metabolism: developmental and nutritional regulation of monoacylglycerol lipase and monoacylglycerol acyltransferase. J Biol Chem 282:33346–33357
Duncan M, Thomas AD, Cluny NL, Patel A, Patel KD, Lutz B, Piomelli D, Alexander SP, Sharkey KA (2008) Distribution and function of monoacylglycerol lipase in the gastrointestinal tract. Am J Physiol Gastrointest Liver Physiol 295:G1255–G1265
Tornqvist H, Belfrage P (1976) Purification and some properties of a monoacylglycerol-hydrolyzing enzyme of rat adipose tissue. J Biol Chem 251:813–819
Sun J, Ji H, Li XX, Shi XC, Du ZY, Chen LQ (2016) Lipolytic enzymes involving lipolysis in Teleost: synteny, structure, tissue distribution, and expression in grass carp (Ctenopharyngodon idella). Comp Biochem Physiol B Biochem Mol Biol 198:110–118
Lehner R, Kuksis A, Itabashi Y (1993) Stereospecificity of monoacylglycerol and diacylglycerol acyltransferases from rat intestine as determined by chiral phase high-performance liquid chromatography. Lipids 28:29–34
Brown JL, Johnston JM (1964) The mechanism of intestinal utilization of monoglycerides. BBA-Lipid Lipid Metab 84:264–274
Pieringer RA, Hokin LE (1962) Biosynthesis of lysophosphatidic acid from monoglyceride and adenosine triphosphate. J Biol Chem 237:653–658
Wiggins D, Gibbons GF (1992) The lipolysis/esterification cycle of hepatic triacylglycerol. Its role in the secretion of very-low-density lipoprotein and its response to hormones and sulphonylureas. Biochem J 284(Pt 2):457–462
Acknowledgements
This work was supported by the Norwegian Research Council (Grant No. 245327). The authors wish to thank the staff at NTNU Sealab for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to report.
About this article
Cite this article
Li, K., Olsen, R.E. Metabolism of sn-1(3)-Monoacylglycerol and sn-2-Monoacylglycerol in Caecal Enterocytes and Hepatocytes of Brown Trout (Salmo trutta). Lipids 52, 61–71 (2017). https://doi.org/10.1007/s11745-016-4215-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-016-4215-0