Abstract
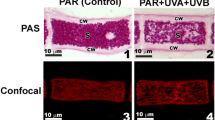
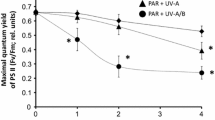
Lichens are phototrophic organisms tolerant to adverse environmental conditions. However, the mechanisms underlying their stress tolerance are not fully understood. For photosynthetic organisms depending on solar radiation, UV-B radiation (280–320 nm) acts as a stress factor. We studied the pro-/antioxidant and respiratory metabolism of Peltigera aphthosa to identify adaptive responses of lichen to a physiological dose of UV-B radiation (14 kJ day−1 for 10 days). A browning of the upper cortex, the appearance of dark spots in the medulla layer of treated thalli, and an increase in the browning reflectance index indicated the synthesis of protective UV screening pigments. UV-B treatment did not cause significant changes in the photosynthetic activity of thalli and isolated algal cells. More intense lipid peroxidation activity and transient changes in H2O2 content accompanied the acclimation process. Higher superoxide dismutase and catalase isoenzyme levels and activity were noted 4 days following the termination of the UV-B treatment. Increased alternative respiration capacity (AP) and a contribution of this energy-dissipating respiratory pathway of up to 45% of the total respiration rate were noted in treated thalli, but not in isolated algal cells. These data demonstrate the UV-B effect on the Peltigera aphthosa respiratory metabolism to be higher due to reactions of the mycobiont than those of the photobiont. We suggest that the activation of the energy-dissipating AP in mycobiont mitochondria may be associated with the synthesis of protective pigments.




Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods in enzymology. Academic Press, Cambridge, pp 121–126
Bahr JT, Bonner WD (1973) Cyanide-insensitive respiration. I. The steady states of skunk cabbage spadix and bean hypocotyl mitochondria. J Biol Chem 248:3441–3445
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Beckett RP, Kranner I, Minibayeva FV (2008) Stress physiology and the symbiosis. In: Nash TH (ed) Lichen biology, 2nd edn. Cambridge University Press, Cambridge, pp 134–151
Beckett RP, Minibayeva FV, Liers C (2013) On the occurrence of peroxidase and laccase activity in lichens. Lichenologist 45:277–283. https://doi.org/10.1017/S0024282912000771
Beckett RP, Minibayeva F, Solhaug KA, Roach T (2021) Photoprotection in lichens: adaptations of photobionts to high light. Lichenologist 53:21–33. https://doi.org/10.1017/S0024282920000535
Bellincampi D, Dipierro N, Salvi G et al (2000) Extracellular H2O2 induced by oligogalacturonides is not involved in the inhibition of the auxin-regulated rolB gene expression in tobacco leaf explants. Plant Physiol 122:1379–1386
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brandt A, de Vera J-P, Onofri S, Ott S (2015) Viability of the lichen Xanthoria elegans and its symbionts after 18 months of space exposure and simulated Mars conditions on the ISS. Int J Astrobiol 14:411–425. https://doi.org/10.1017/S1473550414000214
Caldwell MM, Bornman JF, Ballaré CL et al (2007) Terrestrial ecosystems, increased solar ultraviolet radiation, and interactions with other climate change factors. Photochem Photobiol Sci 6:252–266. https://doi.org/10.1039/B700019G
Chivkunova O, Solovchenko A, Sokolova S et al (2001) Reflectance spectral features and detection of superficial scald–induced browning in storing apple fruit. J Russ Phytopathol Soc 2:73–77
Chowdhury DP, Solhaug KA, Gauslaa Y (2017) Ultraviolet radiation reduces lichen growth rates. Symbiosis 73:27–34. https://doi.org/10.1007/s13199-016-0468-x
Del-Saz NF, Ribas-Carbo M, McDonald AE et al (2018) An in vivo perspective of the role(s) of the alternative oxidase pathway. Trends Plant Sci 23:206–219. https://doi.org/10.1016/j.tplants.2017.11.006
Eisenman HC, Casadevall A (2012) Synthesis and assembly of fungal melanin. Appl Microbiol Biotechnol 93:931–940. https://doi.org/10.1007/s00253-011-3777-2
Elix JA, Stocker-Wörgötter E (2008) Biochemistry and secondary metabolites. In: Nash TH (ed) Lichen biology, 2nd edn. Cambridge University Press, Cambridge, pp 104–133
Fernández MB, Tossi V, Lamattina L, Cassia R (2016) A comprehensive phylogeny reveals functional conservation of the UV-B photoreceptor UVR8 from green algae to higher plants. Front Plant Sci 7:1–6. https://doi.org/10.3389/fpls.2016.01698
Frohnmeyer H, Staiger D (2003) Ultraviolet-B radiation-mediated responses in plants. Balancing Damage and Protection Plant Physiol 133:1420–1428. https://doi.org/10.1104/pp.103.030049
Garmash EV, Velegzhaninov IO, Ermolina KV et al (2020) Altered levels of AOX1a expression result in changes in metabolic pathways in Arabidopsis thaliana plants acclimated to low dose rates of ultraviolet B radiation. Plant Sci 291:110332. https://doi.org/10.1016/j.plantsci.2019.110332
Gauslaa Y, Solhaug KA (2001) Fungal melanins as a sun screen for symbiotic green algae in the lichen Lobaria pulmonaria. Oecologia 126:462–471. https://doi.org/10.1007/s004420000541
Gauslaa Y, Ustvedt EM (2003) Is parietin a UV-B or a blue-light screening pigment in the lichen Xanthoria parietina? Photochem Photobiol Sci 2:424–432. https://doi.org/10.1039/B212532C
Gauslaa Y, Alam MdA, Lucas P-L et al (2017) Fungal tissue per se is stronger as a UV-B screen than secondary fungal extrolites in Lobaria pulmonaria. Fungal Ecol 26:109–113. https://doi.org/10.1016/j.funeco.2017.01.005
Gitelson A, Merzlyak MN (1994) Quantitative estimation of chlorophyll-a using reflectance spectra: experiments with autumn chestnut and maple leaves. J Photochem Photobiol B 22:247–252. https://doi.org/10.1016/1011-1344(93)06963-4
Golovko TK, Shelyakin MA, Zakhozhiy IG et al (2018) The response of lichens to the environmental pollution under the bauxite mining in the taiga zone. Theor Appl Ecol. https://doi.org/10.25750/1995-4301-2018-2-044/2-053/1 (in Russian)
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hideg É, Strid Å (2017) The effects of UV-B on the biochemistry and metabolism of plants. In: Jordan BR (ed) UV-B radiation and plant life: molecular biology to ecology. CABI, Wallingford, pp 90–110
Jansen MAK, Gaba V, Greenberg BM (1998) Higher plants and UV-B radiation: balancing damage, repair and acclimation. Trends Plant Sci 3:131–135. https://doi.org/10.1016/S1360-1385(98)01215-1
Jenkins GI (2009) Signal transduction in responses to UV-B radiation. Annu Rev Plant Biol 60:407–431. https://doi.org/10.1146/annurev.arplant.59.032607.092953
Jenkins GI (2014) The UV-B photoreceptor UVR8: from structure to physiology. Plant Cell 26:21–37. https://doi.org/10.1105/tpc.113.119446
Jensen M (2002) Measurement of chlorophyll fluorescence in lichens. In: Kranner IC, Beckett RP, Varma AK (eds) Protocols in lichenology: culturing, biochemistry, ecophysiology and use in biomonitoring. Springer, Berlin, pp 135–151
Kershaw KA, Macfarlane JD (1980) Physiological-environmental interactions in lichens X. Light as an ecological factor. New Phytol 84:687–702. https://doi.org/10.1111/j.1469-8137.1980.tb04781.x
Kliebenstein DJ, Lim JE, Landry LG, Last RL (2002) Arabidopsis UVR8 regulates ultraviolet-B signal transduction and tolerance and contains sequence similarity to Human regulator of chromatin condensation 1. Plant Physiol 130:234–243. https://doi.org/10.1104/pp.005041
Kornfeld A, Heskel M, Atkin OK et al (2013) Respiratory flexibility and efficiency are affected by simulated global change in Arctic plants. New Phytol 197:1161–1172. https://doi.org/10.1111/nph.12083
Kotlova ER (2000) Possible role of carotenoids in green alga and cyanobacteria adaptation to the lichen symbiosis condition. Botanicheskii Zhurnal 85:103–114 (in Russian)
Kranner I, Cram WJ, Zorn M et al (2005) Antioxidants and photoprotection in a lichen as compared with its isolated symbiotic partners. PNAS 102:3141–3146. https://doi.org/10.1073/pnas.0407716102
Lambers H, Ribas-Carbo M (eds) (2005) Plant respiration: from cell to ecosystem. Springer, Dordrecht
Libik M, Konieczny R, Surowka E, Miszalski Z (2005) Superoxide dismutase activity in organs of Mesembryanthemum crystallinum L. at different stages of CAM development. Acta Biol Crac Ser Bot 47:199–204
Mafole TC, Chiang C, Solhaug KA, Beckett RP (2017) Melanisation in the old forest lichen Lobaria pulmonaria reduces the efficiency of photosynthesis. Fungal Ecol 29:103–110. https://doi.org/10.1016/j.funeco.2017.07.004
Mafole TC, Solhaug KA, Minibayeva FV, Beckett RP (2019a) Tolerance to photoinhibition within lichen species is higher in melanised thalli. Photosyntica 57:96–102. https://doi.org/10.32615/ps.2019.008
Mafole TC, Solhaug KA, Minibayeva FV, Beckett RP (2019b) Occurrence and possible roles of melanic pigments in lichenized ascomycetes. Fungal Biol Rev 33:159–165. https://doi.org/10.1016/j.fbr.2018.10.002
Matee LP, Beckett RP, Solhaug KA, Minibayeva FV (2016) Characterization and role of tyrosinases in the lichen Lobaria pulmonaria (L.) Hoffm. Lichenologist 48:311–322. https://doi.org/10.1017/S0024282916000293
Mayaba N, Beckett R (2001) The effect of desiccation on the activities of antioxidant enzymes in lichens from habitats of contrasting water status. Symbiosis 31:113–121
McDonald AE, Vanlerberghe GC (2006) Origins, evolutionary history, and taxonomic distribution of alternative oxidase and plastoquinol terminal oxidase. Comp Biochem Physiol Part D Genomics Proteomics 1:357–364. https://doi.org/10.1016/j.cbd.2006.08.001
McEvoy M, Gauslaa Y, Solhaug KA (2007) Changes in pools of depsidones and melanins, and their function, during growth and acclimation under contrasting natural light in the lichen Lobaria pulmonaria. New Phytol 175:271–282. https://doi.org/10.1111/j.1469-8137.2007.02096.x
Miszalski Z, Slesak I, Niewiadomska E et al (1998) Subcellular localization and stress responses of superoxide dismutase isoforms from leaves in the C3-CAM intermediate halophyte Mesembryanthemum crystallinum L. Plant Cell Environ 21:169–179. https://doi.org/10.1046/j.1365-3040.1998.00266.x
Møller IM, Bérczi A, van der Plas LHW, Lambers H (1988) Measurement of the activity and capacity of the alternative pathway in intact plant tissues: identification of problems and possible solutions. Physiol Plant 72:642–649. https://doi.org/10.1111/j.1399-3054.1988.tb09176.x
Morales M, Munné-Bosch S (2019) Malondialdehyde: facts and artifacts. Plant Physiol 180:1246–1250. https://doi.org/10.1104/pp.19.00405
Nguyen K-H, Chollet-Krugler M, Gouault N, Tomasi S (2013) UV-protectant metabolites from lichens and their symbiotic partners. Nat Prod Rep 30:1490–1508. https://doi.org/10.1039/C3NP70064J
Nybakken L, Solhaug KA, Bilger W, Gauslaa Y (2004) The lichens Xanthoria elegans and Cetraria islandica maintain a high protection against UV-B radiation in Arctic habitats. Oecologia 140:211–216. https://doi.org/10.1007/s00442-004-1583-6
Palmqvist K (2000) Carbon economy in lichens. New Phytol 148:11–36
Pezzoni M, Pizarro RA, Costa CS (2018) Detection of catalase activity by polyacrylamide gel electrophoresis (PAGE) in cell extracts from Pseudomonas aeruginosa. Bio Protoc. https://doi.org/10.21769/BioProtoc.2869
Raggio J, Pintado A, Ascaso C et al (2011) Whole lichen thalli survive exposure to space conditions: results of lithopanspermia experiment with Aspicilia fruticulosa. Astrobiology 11:281–292. https://doi.org/10.1089/ast.2010.0588
Rizzini L, Favory J-J, Cloix C et al (2011) Perception of UV-B by the Arabidopsis UVR8 protein. Science 332:103–106. https://doi.org/10.1126/science.1200660
Sánchez FJ, Meeßen J, Ruiz MDC et al (2014) UV-C tolerance of symbiotic Trebouxia sp. in the space-tested lichen species Rhizocarpon geographicum and Circinaria gyrosa: role of the hydration state and cortex/screening substances. Int J Astrobiol 13:1–18. https://doi.org/10.1017/S147355041300027X
Schlee D, Kandzia R, Tintemann H, Türk R (1995) Activity of superoxide dismutase and malondialdehyde content in lichens along an altitude profile. Phyton 35:233–242
Shelyakin MA, Zakhozhiy IG, Golovko TK (2018) Changes of total respiration and respiratory pathways ratio in lichens adaptation to UV-B radiation. Izvestia Ufimskogo Nauchnogo Tsentra RAN 3:100–104. https://doi.org/10.31040/2222-8349-2018-5-3-100-104 (in Russian)
Shelyakin M, Zakhozhiy I, Golovko T (2020) The effect of temperature on Antarctic lichen cytochrome and alternative respiratory pathway rates. Polar Biol 43:2003–2010. https://doi.org/10.1007/s00300-020-02758-4
Shelyakin MA, Silina EV, Golovko TK (2021a) The effect of UV-B radiation on the antioxidant system in the Peltigera aphthosa and Peltigera rufescens lichens. J Sib Fed Biol 14:328–338. https://doi.org/10.17516/1997-1389-0359
Shelyakin MA, Zakhozhiy IG, Dalke IV et al (2021b) Photosynthetic and respiratory capacity of foliose lichen Lobaria pulmonaria throughout the annual cycle. Russ J Plant Physiol 68:1048–1058. https://doi.org/10.1134/S1021443721060182
Silberstein L, Siegel BZ, Siegel SM et al (1996) Comparative studies on Xanthoria Parietina, a pollution resistant lichen, and Ramalina Duriaei, a sensitive species. I. Effects of air pollution on physiological processes. Lichenologist 28:355–365. https://doi.org/10.1017/S0024282996000461
Sircar D, Cardoso HG, Mukherjee C et al (2012) Alternative oxidase (AOX) and phenolic metabolism in methyl jasmonate-treated hairy root cultures of Daucus carota L. J Plant Physiol 169:657–663. https://doi.org/10.1016/j.jplph.2011.11.019
Smirnoff N, Arnaud D (2019) Hydrogen peroxide metabolism and functions in plants. New Phytol 221:1197–1214. https://doi.org/10.1111/nph.15488
Solhaug KA, Gauslaa Y (2012) Secondary lichen compounds as protection against excess solar radiation and herbivores. In: Lüttge U, Beyschlag W, Büdel B, Francis D (eds) Progress in botany. Springer, Berlin, pp 283–304
Solhaug KA, Gauslaa Y, Nybakken L, Bilger W (2003) UV-induction of sun-screening pigments in lichens. New Phytol 158:91–100. https://doi.org/10.1046/j.1469-8137.2003.00708.x
Thomson J (1984) American arctic lichens. I. The macrolichens. Columbia University Press, New York
Tretiach M, Bertuzzi S, Candotto Carniel F, Virgilio D (2013) Seasonal acclimation in the epiphytic lichen Parmelia sulcata is influenced by change in photobiont population density. Oecologia 173:649–663. https://doi.org/10.1007/s00442-013-2654-3
Umbach AL, Lacey EP, Richter SJ (2009) Temperature-sensitive alternative oxidase protein content and its relationship to floral reflectance in natural Plantago lanceolata populations. New Phytol 181:662–671. https://doi.org/10.1111/j.1469-8137.2008.02683.x
Ünal D, Uyanikgi̇L Y, (2011) UV-B induces cell death in the lichen Physcia semipinnata (J.F.Gmel). Turk J Biol 35:137–144. https://doi.org/10.3906/biy-0901-5
van Dongen JT, Gupta KJ, Ramírez-Aguilar SJ et al (2011) Regulation of respiration in plants: a role for alternative metabolic pathways. J Plant Physiol 168:1434–1443. https://doi.org/10.1016/j.jplph.2010.11.004
Weissman L, Garty J, Hochman A (2005) Characterization of enzymatic antioxidants in the lichen Ramalina lacera and their response to rehydration. Appl Environ Microbiol 71:6508–6514. https://doi.org/10.1128/AEM.71.11.6508-6514.2005
Weissman L, Fraiberg M, Shine L et al (2006) Responses of antioxidants in the lichen Ramalina lacera may serve as an early-warning bioindicator system for the detection of air pollution stress. FEMS Microbiol Ecol 58:41–53. https://doi.org/10.1111/j.1574-6941.2006.00138.x
Woodbury W, Spencer AK, Stahmann MA (1971) An improved procedure using ferricyanide for detecting catalase isozymes. Anal Biochem 44:301–305. https://doi.org/10.1016/0003-2697(71)90375-7
Yang S-H, Wang L-J, Li S-H et al (2007) The effects of UV-B radiation on photosynthesis in relation to Photosystem II photochemistry, thermal dissipation and antioxidant defenses in winter wheat (Triticum aestivum L.) seedlings at different growth temperatures. Funct Plant Biol 34:907–917
Zhang L, Oh Y, Li H et al (2012) Alternative oxidase in resistance to biotic stresses: Nicotiana attenuata AOX contributes to resistance to a pathogen and a piercing-sucking insect but not Manduca sexta larvae. Plant Physiol 160:1453–1467. https://doi.org/10.1104/pp.112.200865
Zhao M-G, Liu Y-G, Zhang L-X et al (2007) Effects of enhanced UV-B radiation on the activity and expression of alternative oxidase in red kidney bean leaves. J Integr Plant Biol 49:1320–1326. https://doi.org/10.1111/j.1744-7909.2007.00484.x
Fuller KK, Loros JJ, Dunlap JC (2015) Fungal photobiology: visible light as a signal for stress, space and time. Curr Genet 61:275–288. https://doi.org/10.1007/s00294-014-0451-0
Acknowledgements
This work was carried out as part of the “Photosynthesis, respiration and bioenergetics of plants and phototrophic organisms (physiological, biochemical, molecular, genetic and ecological aspects)” project (No. 122040600021-4). We thank Irina Novakovskaya (Department of Flora and Vegetation of the North of the Institute of Biology of the Komi Science Center of the Ural Branch of the Russian Academy of Sciences) for her help in identifying photobiont cells isolated from thalli. Furthermore, we would like to express our gratitude to Editage (www.editage.com) for competent English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by K. Jan Strzałka.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shelyakin, M., Malyshev, R., Silina, E. et al. UV-B induced changes in respiration and antioxidant enzyme activity in the foliose lichen Peltigera aphthosa (L.) Willd.. Acta Physiol Plant 44, 116 (2022). https://doi.org/10.1007/s11738-022-03457-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-022-03457-9