Abstract
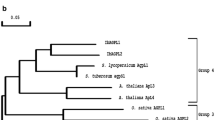
In higher plants, ADP-glucose pyrophosphorylase (EC 2.7.7.27, AGPase), a heterotetrameric enzyme comprised of two small and two large subunits, carries out the first committed step of starch synthesis. To characterize the AGPase genes in lotus, we cloned a small subunit and a large subunit of AGPase using the rapid amplification of cDNA ends (RACE) technique, designated NnAGPS1 and NnAGPL1 genes, respectively. The full-length cDNA of NnAGPS1 was 2,168 bp which codes for a protein of 524 amino acids and of NnAGPL1 was 2,304 bp encoding a protein of 528 amino acids. Complete genomic structures of NnAGPS1 (comprising 9 exons and 8 introns) and NnAGPL1 (comprising 14 exons and 13 introns) were distributed over 3.8 and 4.8 kb, separately. Phylogenetic tree divided the AGPase subunits into two classes: small subunit (SSU) and large subunit (LSU). Sequence alignment of these two genes with other plant species displayed a vast dissimilarity in the N-terminal regions. Results of quantitative real-time RT-PCR (qRT-PCR) showed that both genes were expressed in all tissues, including leaf, leafstalk, rhizome and root. The highest expression of NnAGPS1 was observed in leaves and NnAGPL1 seemed to be preferentially expressed in rhizomes. Besides, transcripts of NnAGPS1 decreased significantly while NnAGPL1 kept a steady level during the early to middle rhizome-swelling stages. Our study will provide important molecular information for the genetic modification and regulation of starch content in lotus.





Similar content being viewed by others
Abbreviations
- AGPase:
-
ADP-glucose pyrophosphorylase
- LSU:
-
Large subunit
- SSU:
-
Small subunit
- AGPS:
-
ADP-glucose pyrophosphorylase small subunit
- AGPL:
-
ADP-glucose pyrophosphorylase large subunit
- RACE:
-
Rapid amplification of cDNA ends
- RT-PCR:
-
Reverse transcription PCR
- qRT-PCR:
-
Quantitative real-time RT-PCR
References
Akihiro T, Mizuno K, Fujimura T (2005) Gene expression of ADP-glucose pyrophosphorylase and starch contents in rice cultured cells are cooperatively regulated by sucrose and ABA. Plant Cell Physiol 46:937–946. doi:10.1093/pcp/pci101
Ballicora MA, Laughlin MJ, Fu Y, Okita TW, Barry GF, Preiss J (1995) Adenosine 5′-diphosphate-glucose pyrophosphorylase from potato tuber. Significance of the N terminus of the small subunit for catalytic properties and heat stability. Plant Physiol 109:245–251
Ballicora MA, Iglesias AA, Preiss J (2004) ADP-glucose pyrophosphorylase: a regulatory enzyme for plant starch synthesis. Photosynth Res 79:1–24. doi:10.1023/B:PRES.0000011916.67519.58
Bejar CM, Ballicora MA, Iglesias AA, Preiss J (2006) ADPglucose pyrophosphorylase’s N-terminus: structural role in allosteric regulation. Biochem Biophys Res Commun 343:216–221. doi:10.1016/j.bbrc.2006.02.123
Choi SB, Kim KH, Kavakli IH, Lee SK, Okita TW (2001) Transcriptional expression characteristics and subcellular localization of ADP-glucose pyrophosphorylase in the oil plant Perilla frutescens. Plant Cell Physiol 42:146–153
Cook FR, Fahy B, Trafford K (2012) A rice mutant lacking a large subunit of ADP-glucose pyrophosphorylase has drastically reduced starch content in the culm but normal plant morphology and yield. Funct Plant Biol 39:1068. doi:10.1071/fp12186
Crevillen P, Ballicora MA, Merida A, Preiss J, Romero JM (2003) The different large subunit isoforms of Arabidopsis thaliana ADP-glucose pyrophosphorylase confer distinct kinetic and regulatory properties to the heterotetrameric enzyme. J Biol Chem 278:28508–28515. doi:10.1074/jbc.M304280200
Cross JM, Clancy M, Shaw JR, Greene TW, Schmidt RR, Okita TW, Hannah LC (2004) Both subunits of ADP-glucose pyrophosphorylase are regulatory. Plant Physiol 135:137–144. doi:10.1104/pp.103.036699
Emanuelsson O, Nielsen H, von Heijne G (1999) ChloroP, a neural network-based method for predicting chloroplast transit peptides and their cleavage sites. Protein Sci 8:978–984. doi:10.1110/ps.8.5.978
Emanuelsson O, Nielsen H, Brunak S, von Heijne G (2000) Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J Mol Biol 300:1005–1016. doi:10.1006/jmbi.2000.3903
Frueauf JB, Ballicora MA, Preiss J (2001) Aspartate residue 142 is important for catalysis by ADP-glucose pyrophosphorylase from Escherichia coli. J Biol Chem 276:46319–46325. doi:10.1074/jbc.M107408200
Frueauf JB, Ballicora MA, Preiss J (2003) ADP-glucose pyrophosphorylase from potato tuber: site-directed mutagenesis of homologous aspartic acid residues in the small and large subunits. Plant J 33:503–511
Fu Y, Ballicora MA, Preiss J (1998) Mutagenesis of the glucose-1-phosphate-binding site of potato tuber ADP-glucose pyrophosphorylase. Plant Physiol 117:989–996
Gibson K et al (2011) Exploiting leaf starch synthesis as a transient sink to elevate photosynthesis, plant productivity and yields. Plant Sci 181:275–281. doi:10.1016/j.plantsci.2011.06.001
Giroux MJ, Shaw J, Barry G, Cobb BG, Greene T, Okita T, Hannah LC (1996) A single mutation that increases maize seed weight. Proc Natl Acad Sci USA 93:5824–5829
Greene TW, Chantler SE, Kahn ML, Barry GF, Preiss J, Okita TW (1996) Mutagenesis of the potato ADPglucose pyrophosphorylase and characterization of an allosteric mutant defective in 3-phosphoglycerate activation. Proc Natl Acad Sci USA 93:1509–1513
Gu C et al (2013) Characterization of genes encoding granule-bound starch synthase in sacred lotus reveals phylogenetic affinity of nelumbo to proteales. Plant Mol Biol Rep 31:1157–1165. doi:10.1007/s11105-013-0605-0
Guo HB (2008) Cultivation of lotus (Nelumbo nucifera Gaertn. ssp. nucifera) and its utilization in China. Genet Resour Crop Evol 56:323–330. doi:10.1007/s10722-008-9366-2
Hannah LC et al (2012) A shrunken-2 transgene increases maize yield by acting in maternal tissues to increase the frequency of seed development. Plant Cell 24:2352–2363. doi:10.1105/tpc.112.100602
Huang B, Hennen-Bierwagen TA, Myers AM (2014) Functions of multiple genes encoding ADP-glucose pyrophosphorylase subunits in maize endosperm, embryo, and leaf. Plant Physiol 164:596–611. doi:10.1104/pp.113.231605
Huggett J, Dheda K, Bustin S, Zumla A (2005) Real-time RT-PCR normalisation; strategies and considerations. Genes Immun 6:279–284. doi:10.1038/sj.gene.6364190
Iglesias AA et al (1993) Expression of the potato tuber ADP-glucose pyrophosphorylase in Escherichia coli. J Biol Chem 268:1081–1086
Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci 8:275–282
Kang G et al (2013) Increasing the starch content and grain weight of common wheat by overexpression of the cytosolic AGPase large subunit gene. Plant Physiol Biochem 73:93–98. doi:10.1016/j.plaphy.2013.09.003
Kavakli IH, Slattery CJ, Ito H, Okita TW (2000) The conversion of carbon and nitrogen into starch and storage proteins in developing storage organs: an overview. Funct Plant Biol 27:561–570. doi:10.1071/PP99176
Kavakli IH, Park JS, Slattery CJ, Salamone PR, Frohlick J, Okita TW (2001) Analysis of allosteric effector binding sites of potato ADP-glucose pyrophosphorylase through reverse genetics. J Biol Chem 276:40834–40840. doi:10.1074/jbc.M106310200
Lee SK et al (2007) Identification of the ADP-glucose pyrophosphorylase isoforms essential for starch synthesis in the leaf and seed endosperm of rice (Oryza sativa L.). Plant Mol Biol 65:531–546. doi:10.1007/s11103-007-9153-z
Li N, Zhang S, Zhao Y, Li B, Zhang J (2011) Over-expression of AGPase genes enhances seed weight and starch content in transgenic maize. Planta 233:241–250. doi:10.1007/s00425-010-1296-5
Linebarger CR, Boehlein SK, Sewell AK, Shaw J, Hannah LC (2005) Heat stability of maize endosperm ADP-glucose pyrophosphorylase is enhanced by insertion of a cysteine in the N terminus of the small subunit. Plant Physiol 139:1625–1634. doi:10.1104/pp.105.067637
Lu Y, Li L, Zhou Y, Gao Q, Liang G, Chen X, Qi X (2012) Cloning and characterization of the Wx gene encoding a granule-bound starch synthase in Lotus (Nelumbo nucifera Gaertn). Plant Mol Biol Rep 30:1210–1217. doi:10.1007/s11105-012-0430-x
Mangelsen E, Wanke D, Kilian J, Sundberg E, Harter K, Jansson C (2010) Significance of light, sugar, and amino acid supply for diurnal gene regulation in developing barley caryopses. Plant Physiol 153:14–33. doi:10.1104/pp.110.154856
Martin C, Smith AM (1995) Starch biosynthesis Plant Cell 7:971–985. doi:10.1105/tpc.7.7.971
Ming R et al (2013) Genome of the long-living sacred lotus (Nelumbo nucifera Gaertn.). Genome Biol 14:R41. doi:10.1186/gb-2013-14-5-r41
Morell MK, Bloom M, Knowles V, Preiss J (1987) Subunit structure of spinach leaf ADPglucose pyrophosphorylase. Plant Physiol 85:182–187
Nakamura Y, Kawaguchi K (1992) Multiple forms of ADP-glucose pyrophorylase of rice endosperm. Physiol Plant 84:336–342. doi:10.1111/j.1399-3054.1992.tb04673.x
Ni XM (1987) Lotus of China. Science Press, Beijing
Nielsen H, Engelbrecht J, Brunak S, von Heijne G (1997) Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng 10:1–6
Obana Y et al (2006) Enhanced turnover of transitory starch by expression of up-regulated ADP-glucose pyrophosphorylases in Arabidopsis thaliana. Plant Sci 170:1–11. doi:10.1016/j.plantsci.2005.07.019
Okita TW, Nakata PA, Anderson JM, Sowokinos J, Morell M, Preiss J (1990) The subunit structure of potato tuber ADPglucose pyrophosphorylase. Plant Physiol 93:785–790
Preiss J (1988) Biosynthesis of starch and its regulation, vol 14. The biochemistry of plants, NewYork
Preiss J (1991) Biology and molecular biology of starch synthesis and its regulation. In: Mifflin B (ed) Oxford surveys of plant molecular and cell biology. Oxford University Press, Oxford
Rani S, Sharma P, Sharma A, Chatrath R (2013) Comparative computational analysis of ADP Glucose pyrophosphorylase in plants. Bioinformation 9:572–576. doi:10.6026/97320630009572
Sakulsingharoj C et al (2004) Engineering starch biosynthesis for increasing rice seed weight: the role of the cytoplasmic ADP-glucose pyrophosphorylase. Plant Sci 167:1323–1333. doi:10.1016/j.plantsci.2004.06.028
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108. doi:10.1038/nprot.2008.73
Seferoglu AB, Baris I, Morgil H, Tulum I, Ozdas S, Cevahir G, Kavakli IH (2013) Transcriptional regulation of the ADP-glucose pyrophosphorylase isoforms in the leaf and the stem under long and short photoperiod in lentil. Plant Sci 205–206:29–37. doi:10.1016/j.plantsci.2013.01.006
Shen-Miller J et al (2002) Long-living lotus: germination and soil γ-irradiation of centuries-old fruits, and cultivation, growth, and phenotypic abnormalities of offspring. Am J Bot 89:236–247. doi:10.3732/ajb.89.2.236
Sivak MN, Preiss J (1998) Starch: basic science and biotechnology. Adv Food Nutr Res 41:1–131
Slattery CJ, Kavakli IH, Okita TW (2000) Engineering starch for increased quantity and quality. Trends Plant Sci 5:291–298
Smidansky ED, Clancy M, Meyer FD, Lanning SP, Blake NK, Talbert LE, Giroux MJ (2002) Enhanced ADP-glucose pyrophosphorylase activity in wheat endosperm increases seed yield. Proc Natl Acad Sci USA 99:1724–1729. doi:10.1073/pnas.022635299
Smidansky ED, Martin JM, Hannah LC, Fischer AM, Giroux MJ (2003) Seed yield and plant biomass increases in rice are conferred by deregulation of endosperm ADP-glucose pyrophosphorylase. Planta 216:656–664. doi:10.1007/s00425-002-0897-z
Smidansky ED, Meyer FD, Blakeslee B, Weglarz TE, Greene TW, Giroux MJ (2007) Expression of a modified ADP-glucose pyrophosphorylase large subunit in wheat seeds stimulates photosynthesis and carbon metabolism. Planta 225:965–976. doi:10.1007/s00425-006-0400-3
Smith SM et al (2004) Diurnal changes in the transcriptome encoding enzymes of starch metabolism provide evidence for both transcriptional and posttranscriptional regulation of starch metabolism in arabidopsis leaves. Plant Physiol 136:2687–2699. doi:10.1104/pp.104.044347
Smith-White BJ, Preiss J (1992) Comparison of proteins of ADP-glucose pyrophosphorylase from diverse sources. J Mol Evol 34:449–464
Stark DM, Timmerman KP, Barry GF, Preiss J, Kishore GM (1992) Regulation of the amount of starch in plant tissues by ADP glucose pyrophosphorylase. Science 258:287–292. doi:10.1126/science.258.5080.287
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599. doi:10.1093/molbev/msm092
Tenorio G, Orea A, Romero JM, Merida A (2003) Oscillation of mRNA level and activity of granule-bound starch synthase I in Arabidopsis leaves during the day/night cycle. Plant Mol Biol 51:949–958
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Tuncel A, Okita TW (2013) Improving starch yield in cereals by over-expression of ADPglucose pyrophosphorylase: expectations and unanticipated outcomes. Plant Sci 211:52–60. doi:10.1016/j.plantsci.2013.06.009
Ventriglia T et al (2008) Two Arabidopsis ADP-glucose pyrophosphorylase large subunits (APL1 and APL2) are catalytic. Plant Physiol 148:65–76. doi:10.1104/pp.108.122846
Wu MX, Preiss J (1998) The N-terminal region is important for the allosteric activation and inhibition of the Escherichia coli ADP-glucose pyrophosphorylase. Arch Biochem Biophys 358:182–188. doi:10.1006/abbi.1998.0846
Wu MX, Preiss J (2001) Truncated forms of the recombinant Escherichia coli ADP-glucose pyrophosphorylase: the importance of the N-terminal region for allosteric activation and inhibition. Arch Biochem Biophys 389:159–165. doi:10.1006/abbi.2001.2327
Xu W et al (2011) Transcriptome phase distribution analysis reveals diurnal regulated biological processes and key pathways in rice flag leaves and seedling leaves. PLoS One 6:e17613. doi:10.1371/journal.pone.0017613
Zhao Y (1999) Chinese aquatic vegetables. Press of Chinese Agricultural University, Beijing
Acknowledgments
This work was supported by the National Science and Technology Supporting Program (No. 2012BAD27B01). The authors are very grateful to Mr. Hong-Liang Zhan, Jian-Xiong Wang and Mrs. Xia Yu (Wuhan University) for their help in the cultivation of study materials.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Abe.
Rights and permissions
About this article
Cite this article
Cheng, N., Zeng, XF., Zheng, XF. et al. Cloning and characterization of the genes encoding the small and large subunit of the ADP-glucose pyrophosphorylase in lotus (Nelumbo nucifera Gaertn). Acta Physiol Plant 37, 1734 (2015). https://doi.org/10.1007/s11738-014-1734-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-014-1734-2