Abstract
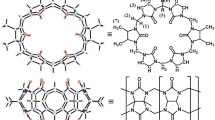
In recent years, symmetrical α,α′,δ,δ′-tetramethyl-cucurbit[6]uril (TMeQ[6]) has attracted much attention from many researchers due to its interesting assembly pattern. Herein, we have investigated the host–guest interactions pattern of the symmetric TMeQ[6] with (E)-1-(5-carboxypentyl)-4-(2-(pyridin-4-yl)vinyl)pyridin-1-ium. (ECPP). Furthermore, their binding behaviors were investigated by 1H NMR spectroscopy, MALDI-TOF mass spectrometry, isothermal titration calorimeter, fluorescence, and UV–vis spectrum. The results indicate that TMeQ[6] and ECPP form host–guest complexes in a host/guest molar ratio of 1:1. TMeQ[6] is bound with the vinylpyridine group of ECPP. In addition, TMeQ[6]-ECPP exhibits a high association constant Ka = (1.520 ± 0.58) × 106 M−1, ΔH = − 53.40 kJ/mol, ΔS = − 60 J/mol·K. which provides a promising foundation for potential studies of the assemblies.
Graphical abstract






Similar content being viewed by others
Data availability
Data in the manuscript and references were completed by the authors and data are available.
References
Bai H, He W, Chau JH, Zheng CZ, Kwok RTK, Lam JWY, Tang BZ (2021) AIEgens for microbial detection and antimicrobial therapy. Biomaterials 268:120598. https://doi.org/10.1016/j.biomaterials.2020.120598
Bardelang D, Udachin AK, Leeka MD, Ripmeester AJ (2007) Highly symmetric columnar channels in metal-free cucurbit[n]uril hydrate crystals (n = 6, 8). CrystEngComn 9:973–975. https://doi.org/10.1039/B711285H
Cen R, Liu M, Lu JH, Zeng X, Zhang WF, Dai JJ, Tao Z, Xiao X (2022) Synthesis and characterization of asensitive and selective Fe3+ fluorescent sensor based on novel sulfonated calix[4]arene-basedhost-guestcomplex. Chinese Chem Lett 33:2469–2472. https://doi.org/10.1016/j.cclet.2021.12.005
Chen K, Zhu ZQ, Zhang MH, Yang X, Li J, Chen C, Redshaw C (2023) 4,4′-Biphenyldisulfonic acid induced coordination polymers of symmetrical tetramethyl cucurbit[6]uril with alkaline-earth metals for detection of antibiotics. CrystEngComm 25:961–970. https://doi.org/10.1039/D2CE01470J
Cong H, Ni XL, Xiao X, Huang Y, Zhu QJ, Xue SF, Tao Z, Lindoy LF, Wei G (2016) Synthesis and separation of cucurbit[n]urils and their derivatives. Org Biomol Chem 14:4335–4364. https://doi.org/10.1039/C6OB00268D
Dai XY, Hu YY, Sun YH, Huo M, Dong XY, Liu Y (2022) A highly efficient phosphorescence/fluorescence supramolecular switch based on a bromoisoquinoline cascaded assembly in aqueous solution. Adv Sci 9(14):2200524. https://doi.org/10.1002/advs.202200524
Elbashir AA, Aboul-Enein YH (2015) Supramolecular. analytical application of Cucurbit[n]urils using fluorescence spectroscopy. Crit Rev Anal Chem 45:52–61. https://doi.org/10.1080/10408347.2013.876354
Gong YY, Tan YQ, Mei J, Zhang YR, Yuan WZ, Zhang YM, Tang BZ (2013) Room temperature phosphorescence from natural products: crystallization matters. Sci China Chem 56:1178–1182. https://doi.org/10.1007/s11426-013-4923-8
Hou RX, Hu JH, Li Q, Zhang JY, Pan DW, Xiong Y, Liu ZN, Xiao X (2023) A symmetric tetramethyl cucurbit[6]uril-based supramolecular assembly for the visual detection of CrO42- in water. J Mol Struct 1294:136447. https://doi.org/10.1016/j.molstruc.2023.136447
Huang SM, Huang SJ, Yan YJ, Yu SH, Chou M, Yang HW, Chang YS, Chen RS (2017) Highly responsive photoconductance in a Sb2SeTe2 topological insulator nanosheet at room temperature. RSC Adv 7:39057–39062. https://doi.org/10.1039/C7RA06151J
Isaacs L (2009) Cucurbit[n]urils: from mechanism to structure and function. Chem Commun 6:619–629. https://doi.org/10.1039/B814897J
Kim K, Murray J, Ko YH, Hwang I (2018) Cucurbiturils. World Scientific London, Chemistry, Supramolecular Chemistry and Applications
Lagona J, Fettinger CJ, Isaacs L (2003) Cucurbit[n]uril analogues. Org Lett 5(20):3745–3747. https://doi.org/10.1021/ol035468w
Lampel A, Ulijn RV, Tuttle T (2018) Guiding principles for peptide nanotechnology through directed discovery. Chem Soc Rev 47:3737–3758. https://doi.org/10.1039/C8CS00177D
Lim S, Kim H, Selvapalam N, Kim KJ, Cho SJ, Seo G, Kim K (2008) Cucurbit[6]uril: organic molecular porous material with permanent porosity, exceptional stability, and acetylene sorption properties. Angew Chem Int Ed 47(18):3352–3355. https://doi.org/10.1002/anie.200800772
Lin RL, Fang GS, Sun WQ, Liu JX (2016) Aniline-containing guests recognized by α, α′, δ, δ′-tetramethyl-cucurbit[6]uril host. Sci Rep 6:39057. https://doi.org/10.1038/srep39057
Luo Y, Zhang W, Ren Q, Tao Z, Xiao X (2022) Highly efficient artificial light-harvesting systems constructed in aqueous solution based on twisted Cucurbit[14]uril. ACS Appl Mater Interfaces 14(26):29806–29812. https://doi.org/10.1021/acsami.2c05599
Mao Z, Yang Z, Fan ZG, Ubba E, Li WL, Li Y, Zhao J, Yang ZY (2019) Matthew P. Aldreda, Zhenguo Chi. The methylation effect in prolonging the pure organic room temperature phosphorescence lifetime. Chem Sci 10:179–184. https://doi.org/10.1039/C8SC03019G
Meng D, Liang H, Chen Q, Shen XH (2018) Preparation and characterization of a novel single crystal of Th(IV) with cucurbit[6]uril. Chin Chem Lett 29:447–450. https://doi.org/10.1016/j.cclet.2017.09.030
Mu L, Yang XB, Xue SF, Zhu QJ, Tao Z, Zeng X (2007) Cucurbit[n]urils-induced room temperature phosphorescence of quinoline derivatives. Anal Chim Acta 597(1):90–96. https://doi.org/10.1016/j.aca.2007.06.049
Nowicki J, Jaroszewska K, Nowakowska-Bogdan E, Szmato M, Iłowska J (2018) Synthesis of 2,2,4-trimethyl-1,2-H-dihydroquinoline (TMQ) over selected organosulfonic acid silica catalysts: Selectivity aspects. Mol Catal 454:94–103. https://doi.org/10.1016/j.mcat.2018.05.016
Peerannawar RS, Gobre VV, Gejji SP (2012) Binding of viologen derivatives to cucurbit[8]uril. Comput Theor Chem 983:16–24. https://doi.org/10.1016/j.comptc.2011.12.013
Shan PH, Hu JH, Liu M, Tao Z, Xiao X, Redshaw C (2022) Progress in host-guest macrocycle/pesticide research: Recognition, detection, release and application. Coord Chem Rev 467:214580. https://doi.org/10.1016/j.ccr.2022.214580
Wang YZ, Xin B, Chen XH, Zheng SY, Yuan Y, Zhang M (2018) Emission and emissive mechanism of nonaromatic oxygen clusters. Macromol Rapid Commun 39:1800528. https://doi.org/10.1002/marc.201800528
Xiao ZY, Lin RL, Tao Z, Liu QY, Liu JX, Xiao X (2017) Multiple noncovalent interaction constructed polymeric supramolecular crystals: recognition of butyl viologen bypara-dicyclohexanocucurbit[6]uril and α, α′, δ, δ′-tetramethylcucurbit[6]uril. Org Chem Front 4:2422. https://doi.org/10.1039/C7QO00708F
Xu W, Kan J, Yang B, Prior JT, Bian B, Xiao X, Tao Z, Redshaw C (2019) A Study of the interaction between Cucurbit[8]uril and alkyl-substituted 4-Pyrrolidinopyridinium salts. Chem-An Asian J 4(1):235–242. https://doi.org/10.1002/asia.201801498
Yang B, Xiao X, Zhang YQ, Feng XS, Tao Z, Wei G (2014) Inclusion of 4-pyrrolidinopyridine derivatives in a symmetrical α, α′, δ, δ′-tetramethyl-cucurbit[6]uril and a Ba2+-driven pseudorotaxane with characteristic UV absorption changes. RSC Adv 4:44359–44366. https://doi.org/10.1039/C4RA07314B
Yang D, Liu M, Xiao X, Tao Z, Redshaw C (2021) Polymeric self-assembled cucurbit[n]urils: synthesis, structures and applications. Coordin Chem Rev 434:213733. https://doi.org/10.1016/j.ccr.2020.213733
Yu DH, Ni XL, Tian ZC, Zhang YQ, Xue SF, Tao Z, Zhu QJ (2008) Host-guest inclusion complexes of four partial alkyl-substituted cucurbit[6]urils with some probe guests. J Mol Struct 891:247–253. https://doi.org/10.1016/j.molstruc.2008.03.045
Zhang M, Cao LP, Isaacs L (2014) Cucurbit[6]uril-cucurbit[7]uril heterodimer promotes controlled self-assembly of supramolecular networks and supramolecular micelles by self-sorting of amphiphilic guests. Chem Commun 50:14756–14759. https://doi.org/10.1039/C4CC07268E
Zhang ML, Lin RL, Sun WQ, Liu JX (2018) Anion encapsulation and complexation by cucurbit[n]urils and their derivatives. J Incl Phenom Macrocycl Chem 90:173–187. https://doi.org/10.1007/s10847-017-0777-6
Zhang XD, Zhao Y, Chen K, Guo JH, Wang P, Wu H, Sun WY (2019a) Cucurbit[6]uril-based supramolecular assemblies incorporating metal complexes with multiaromatic ligands as structure-directing agent for detection of aromatic amines and nitroaromatic compounds. Sens Actuators, B Chem 282:844–853. https://doi.org/10.1016/j.snb.2018.11.128
Zhang K, Sun W, Lin R, Xiao X, Bian B, Tao Z (2019) Liu J (2019b) controlled encapsulation and release of an organic guest in the cavity of α, α′, δ, δ′-Tetramethylcucurbit[6]uril. Eur J Org Chem 7:1503–1507. https://doi.org/10.1002/ejoc.201801652
Zhang W, Luo Y, Zhou Y, Liu M, Xu WT, Bian B, Tao Z, Xiao X (2020) A highly selective fluorescent chemosensor probe for detection of Fe3+ and Ag+ based on supramolecular assembly of cucurbit[10]uril with a pyrene derivative. Dyes Pigm 176:108235. https://doi.org/10.1016/j.dyepig.2020.108235
Zhang XD, Chen K, Sun WY (2021) Potential applications of cucurbit[n]urils and their derivatives in the capture of hazardous chemicals. Chem Eur J 27:5107–5119. https://doi.org/10.1002/chem.202004711
Zhao YJ, Xue SF, Zhu QJ, Tao Z, Zhang JX, Wei ZB, Long LS, Hu ML, Xiao HP, Day A (2004) Synthesis of a symmetrical tetrasubstituted cucurbit[6]uril and its host-guest inclusion complex with 2,2′-bipyridine. Chin Sci Bull 49:1111–1116. https://doi.org/10.1360/04wb0031
Funding
This work was supported by the Science and Technology Fund of Guizhou Province and the Innovation Program for High-level Talents of Guizhou Province (No. 2016–5657).
Author information
Authors and Affiliations
Contributions
All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. C-LY and HY-W have contributed equally as first authors.
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yue, CL., Wang, HY., Yang, MX. et al. Supramolecular self-assembly between symmetric tetramethyl cucurbit[6]uril and 1,2-Di(4-pyridyl)ethylene derivative. Chem. Pap. 77, 7625–7631 (2023). https://doi.org/10.1007/s11696-023-03074-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-03074-x