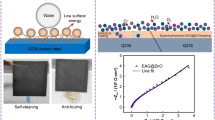
Abstract
In order to achieve superhydrophobic characteristics, Zn–CaO nanoparticles has been electropositively coated on mild steel substrates and then functionalized using silicone rubber. SEM morphological characteristics reveal that the electrodeposited zinc films and functionalized silicone reveal a porous, rectangular structure. Infrared spectroscopy and X-ray diffraction (XRD) were used to determine the functional group and phases Zn–CaO nanoparticles functionalized with silicone has been discovered and offers superhydrophobic qualities with a 175° contact angle. Superhydrophobic zinc-coated steel has been shown to be more corrosion resistant than mild steel in simulated saltwater. Similarly, the centrifugal adhesion test results show that the observed superhydrophobic surfaces' ice adhesion strength was 6.5 times lower than that of mild steel. This coating has potential uses in marine environments to lessen ice adhesion and prevent corrosion.











Similar content being viewed by others
Availability of data and materials
The authors confirm that the data supporting this study's conclusions is included in the publication.
References
Adams SM, Anianwu FO, Aigbodion VS (2022) Ecofriendly new nanocomposites coating formulation of zinc reinforced with calcium oxide nanoparticles synthesis from oyster shell. J Indian Chem Soc 99(8):100609. https://doi.org/10.1016/j.jics.2022.100609
Adams SM, Anianwu FO, Aigbodion VS (2023) High-performance multi-response optimization of new nanocomposite coating formulation of zinc reinforced with calcium oxide nanoparticle synthesis from oyster shell via one-step electrodeposition approach. Emergent Mater. https://doi.org/10.1007/s42247-023-00458-3
Aigbodion VS, Akinlabi ET (2019) Explicit microstructural evolution and electrochemical performance of zinc-eggshell particles composite coating on mild steel. Surf Interfaces. https://doi.org/10.1016/j.surfin.2019.100387
Aigbodion V, Neife S, Suleiman I (2018) Surface modification of mild steel by co-deposition using Zn–ZnO–Ant hill particulate composite coating in simulated sea water. Proc Inst Mech Eng Part M J Eng Marit Environ. https://doi.org/10.1177/1475090218792382
Bakkar A, Neubert V (2007) Electrodeposition onto magnesium in air and water stable ionic liquids: from corrosion to successful plating. Electrochem Commun 9(9):2428–2435. https://doi.org/10.1016/j.elecom.2007.07.010
Berger F, Delhalle J, Mekhalif Z (2009) Undec-10-ene-1-thiol multifunctional molecular layer as a junction between metallic zinc and polymer coatings on steel. Electrochim Acta 54(26):6464–6471. https://doi.org/10.1016/j.electacta.2009.06
Brassard J-D, Sarkar DK, Perron J (2012) Fluorine based superhydrophobic coatings. Appl Sci 2(2):453–464. https://doi.org/10.3390/app2020453
Cassie ABD, Baxter S (1944) Wettability of porous surfaces. Trans Faraday Soc 40:546–551
Chang Y-T, Wen N-T, Chen W-K, Ger M-D, Pan G-T, Yang TC-K (2008) The effects of immersion time on morphology and electrochemical properties of the Cr(III)-based conversion coatings on zinc coated steel surface. Corros Sci 50(12):3494–3499. https://doi.org/10.1016/j.corsci.2008.08.051
Chen H, Wang F, Fan H, Hong R, Li W (2020) Construction of MOF-based superhydrophobic composite coating with excellent abrasion resistance and durability for self-cleaning, corrosion resistance, anti-icing, and loading-increasing research. Chem Eng J 456:127343. https://doi.org/10.1016/j.cej.2020.127343
Darmanin T, de Givenchy ET, Amigoni S, Guittard F (2013) Superhydrophobic surfaces by electrochemical processes. Adv Mater 25(10):1378–1394. https://doi.org/10.1002/adma.201204300
Ghalmi Z, Farzaneh M (2014) Durability of nanostructured coatings based on PTFE nanoparticles deposited on porous aluminum alloy. Appl Surf Sci 314:564–569. https://doi.org/10.1016/j.apsusc.2014.05.194
Han D, Steckl AJ (2009) Superhydrophobic and oleophobic fibers by coaxial electrospinning. Langmuir 25(16):9454–9462. https://doi.org/10.1021/la900660v
Ishizaki T, Sakamoto M (2011) Facile formation of biomimetic color-tuned superhydrophobic magnesium alloy with corrosion resistance. Langmuir 27(6):2375–2381. https://doi.org/10.1021/la1051029
Ishizaki T, Masuda Y, Sakamoto M (2011) Corrosion resistance and durability of superhydrophobic surface formed on magnesium alloy coated with nanostructured cerium oxide film and fluoroalkylsilane molecules in corrosive NaCl aqueous solution. Langmuir 27(8):4780–4788. https://doi.org/10.1021/la2002783
Jeong C, Choi C-H (2012) Single-step direct fabrication of pillar-on-pore hybrid nanostructures in anodizing aluminum for superior superhydrophobic efficiency. ACS Appl Mater Interfaces 4(2):842–848. https://doi.org/10.1021/am201514n
Kang M, Kim I, Kim SW, Ryu J-W, Park HY (2011) Metal-insulator transition without structural phase transition in V2O5 film. Appl Phys Lett 98(13):131907. https://doi.org/10.1063/1.3571557
Lazauskas A, Guobienė A, Prosyčevas I, Baltrušaitis I, Grigaliūnas I, Narmontas P, Baltrusaitis I (2013) Water droplet behavior on superhydrophobic SiO2 nanocomposite films during icing/deicing cycles. Mater Charact 82:9–16. https://doi.org/10.1016/j.matchar.2013.04.017
Li M, Feng C, Zhang Z, Sugiura N (2009) Efficient electrochemical reduction of nitrate to nitrogen using Ti/IrO2–Pt anode and different cathodes. Electrochim Acta 54(20):4600–4606. https://doi.org/10.1016/j.electacta.2009.03.0
Liu H, Szunerits S, Xu W, Boukherroub R (2009) Preparation of superhydrophobic coatings on zinc as effective corrosion barriers. ACS Appl Mater Interfaces 1(6):1150–1153. https://doi.org/10.1021/am900100q
Lu X, Zhou J, Zhao Y, Qiu Y, Li J (2008) Room temperature ionic liquid based polystyrene nanofibers with superhydrophobicity and conductivity produced by electrospinning. Chem Mater 20(10):3420–3424. https://doi.org/10.1021/cm800045h
Lu Y, Song J, Liu X, Xu W, Xing Y, Wei Z (2012) Preparation of superoleophobic and superhydrophobic titanium surfaces via an environmentally friendly electrochemical etching method. ACS Sustain Chem Eng 1(1):102–109. https://doi.org/10.1021/sc3000527
Manca M, Cannavale A, De Marco L, Aricò AS, Cingolani R, Gigli G (2009) Durable superhydrophobic and antireflective surfaces by trimethylsilanized silica nanoparticles-based sol−gel processing. Langmuir 25(11):6357–6362. https://doi.org/10.1021/la804166t
Morón LE, Méndez A, Castañeda F, Flores JG, Ortiz-Frade L, Meas Y, Trejo G (2011) Electrodeposition and corrosion behavior of Zn coatings formed using as brighteners arene additives of different structure. Surf Coat Technol 205(21–22):4985–4992. https://doi.org/10.1016/j.surfcoat.2011.04.09
Ou J, Hu W, Xue M, Wang F, Li W (2013) Superhydrophobic surfaces on light alloy substrates fabricated by a versatile process and their corrosion protection. ACS Appl Mater Interfaces 8:3101–3107. https://doi.org/10.1021/am4000134
Polyakov NA, Botryakova IG, Glukhov VG, Red’kina GV, Kuznetsov YI (2020) Formation and anticorrosion properties of superhydrophobic zinc coatings on steel. Chem Eng J. https://doi.org/10.1016/j.cej.2020.127775
Safaee A, Sarkar DK, Farzaneh M (2008) Superhydrophobic properties of silver-coated films on copper surface by galvanic exchange reaction. Appl Surf Sci 254(8):2493–2498. https://doi.org/10.1016/j.apsusc.2007.09.073
Sonawane SH, Bhanvase BA, Jamali AA, Dubey SK, Kale SS, Pinjari DV et al (2012) Improved active anticorrosion coatings using layer-by-layer assembled ZnO nanocontainers with benzotriazole. Chem Eng J 189–190:464–472. https://doi.org/10.1016/j.cej.2012.02.076
Tadanaga K, Morinaga J, Matsuda A, Minami T (2000) Superhydrophobic−superhydrophilic micropatterning on flowerlike alumina coating film by the sol−gel method. Chem Mater 12(3):590–592. https://doi.org/10.1021/cm990643h
Wang L, Gao Y, Xu T, Xue Q (2006) Corrosion resistance and lubricated sliding wear behaviour of novel Ni–P graded alloys as an alternative to hard Cr deposits. Appl Surf Sci 252(20):7361–7372. https://doi.org/10.1016/j.apsusc.2005.08.040
Xu W, Song J, Sun J, Lu Y, Yu Z (2011) Rapid fabrication of large-area, corrosion-resistant superhydrophobic Mg alloy surfaces. ACS Appl Mater Interfaces 3(11):4404–4414. https://doi.org/10.1021/am2010527
Zhang X, Shi F, Yu X, Liu H, Fu Y, Wang Z, Jiang L, Li X (2004) Polyelectrolyte multilayer as matrix for electrochemical deposition of gold clusters: toward super-hydrophobic surface. J Am Chem Soc 126(10):3064–3065. https://doi.org/10.1021/ja0398722
Zhang X, Liang J, Liu B, Peng Z (2014) Preparation of superhydrophobic zinc coating for corrosion protection. Colloids Surf A 454:113–118. https://doi.org/10.1016/j.colsurfa.2014.04.02
Zhang H, Zhang N, Fang F (2020) Synergistic effect of surfactant and saccharin on dispersion and crystal refinement for electrodeposition of nanocrystalline nickel/graphene oxide composite. Surf Coat Technol. https://doi.org/10.1016/j.surfcoat.2020.12629
Zhao N, Shi F, Wang Z, Zhang X (2005) Combining layer-by-layer assembly with electrodeposition of silver aggregates for fabricating superhydrophobic surfaces. Langmuir 21(10):4713–4716. https://doi.org/10.1021/la0469194
Acknowledgements
The author hereby appreciates and acknowledges the Africa Centre of Excellence for Sustainable Power and Energy Development, ACE-SPED, University of Nigeria, Nsukka; Energy materials research group, University of Nigeria, Nsukka, Nigeria; and Faculty of Engineering and Built Environment, University of Johannesburg, Auckland Park, South Africa for their supports.
Funding
There is no money available for this project.
Author information
Authors and Affiliations
Contributions
They contribute 100% to the study work's development, from experimentation to analysis.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicting interests to disclose in this study.
Acceptance of ethics
Because this experiment does not involve people or animals, no ethics committee approval is required.
Consent of participants
Because this study does not include people or animals, no permission is required to participate.
Publishing permission
The writers grant their permission for the work to be published by the publisher.
Consent to publish
The authors give the publisher the consent to publish the work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aigbodion, V.S. Unveiling anticorrosion and ice adhesion of superhydrophobic Zn–CaO nanoparticle derived from oyster composite coating functionalized with ultra-thin films silicone. Chem. Pap. 77, 6483–6490 (2023). https://doi.org/10.1007/s11696-023-02952-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-02952-8