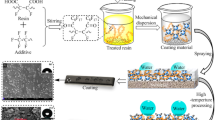
Abstract
Inspired from nature, superhydrophobic surfaces having selective water repelling properties were facilely fabricated without using any fluorinated chemicals. The sessile water contact angle (CA) ≈ 168° with low contact angle hysteresis (CAH) was achieved on this reduced graphene oxide (rGO) surface. Typically, low surface free energy chemicals are employed to fabricate extreme water repellent surface. Herein, graphene oxide (GO) and rGO were coated by drop casting method. Superhydrophobic surfaces were fabricated by coating rGO on one side of the double-sided adhesive tape. The surface wettability of the GO and rGO surfaces were characterized by drop shape analyzer. The water contact angle increases from 61° on GO coated surface to CA ≈ 168° extreme partial wetting (superhydrophobic) for rGO coatings. The CAH reduces from ~ 27° to less than 5° for rGO coated surface. The adhesion of GO and rGO was also achieved using epoxy resin and the surface wettability was compared. The unique combination of surface chemistry and roughness provided very high CA. The surface was stable against hot water, chemicals (acids, bases, alkali) and mechanical abrasion. On such low CAH surface, the self-cleaning ability was shown by removal of deposited impurity by jet of water drops.








Similar content being viewed by others
References
Aliofkhazraei M (2015) Re-derivation of Young’s equation, Wenzel equation, and Cassie–Baxter equation based on energy minimization. In: surface energy. IntechOpen, London, pp 1–21
Autumn K, Liang YA, Hsieh ST, Zesch W, Chan WP, Kenny TW, Fearing R, Full RJ (2000) Adhesive force of a single gecko foot-hair. Nature 405:681–685. https://doi.org/10.1038/35015073
Ball DW (2006) Fisico-Quimica, vol 2. Thomson, Sao Paulo, pp 1–877
Bartali R, Zhang G, Tong X, Speranza G, Micheli V, Gottardi G, Michele F, Filippo P, Shuhui S, Nadhira L, Tavares AC (2020) Graphene oxide/reduced graphene oxide films as protective barriers on lead against differential aeration corrosion induced by water drops. Nanoscale Adv 2:5412–5420. https://doi.org/10.1039/d0na00212g
Barthlott W, Neinhuis C (1997) Purity of the sacred lotus, or escape from contamination in biological surfaces. Planta 202:1–8. https://doi.org/10.1007/s004250050096
Belyaeva LA, Schneider GF (2020) Wettability of graphene. Surf Sci Rep. https://doi.org/10.1016/j.surfrep.2020.100482
Bharathidasan T, Narayanan TN, Sathyanaryanan S, Sreejakumari SS (2015) Above 170° water contact angle and oleophobicity of fluorinated graphene oxide based transparent polymeric films. Carbon 84:207–213. https://doi.org/10.1016/j.carbon.2014.12.004
Blissessur R, Scully SF (2007) Intercalation of solid polymer electrolytes into graphite oxide. Solid State Ionics 178:877–882. https://doi.org/10.1016/j.ssi.2007.02.030
Brassard J-D, Sarkar DK, Perron J (2012) Fluorine based superhydrophobic coatings. Appl Sci 2:453–464. https://doi.org/10.3390/app2020453
Crick CR, Parkin IP (2010) Preparation and characterisation of super-hydrophobic surfaces. Chem Eur J 16:3568–3588. https://doi.org/10.1002/chem.200903335
Ding G, Jiao W, Chen L, Yan M, Hao L, Wang R (2018) A self-sensing, superhydrophobic, heterogeneous graphene network with controllable adhesion behaviour. J Mater Chem A 6:16992–17000
Du F, Huang J, Duan H, Xiong C, Wang J (2018) Wetting transparency of supported graphene is regulated by polarities of liquids and substrates. Appl Surf Sci 454:249–255. https://doi.org/10.1016/j.apsusc.2018.05.098
Erbil HY (2020) Practical applications of superhydrophobic materials and coatings: problems and perspectives. Langmuir 36(10):2493–2509. https://doi.org/10.1021/acs.langmuir.9b03908
Ewing GW (2009) Metodos instrumentais de analise Quimica, vol 2. Blucher, Sao Paulo, pp 1–230
Extrand CW (2002) Model for contact angles and hysteresis on rough and ultraphobic surfaces. Langmuir 18:7991–7999. https://doi.org/10.1021/la025769z
Feng L, Li S, Li Y, Li H, Zhang L, Zhai J, Song Y, Liu B, Jiang L, Zhu D (2002) Super-hydrophobic surfaces: From natural to artificial. Adv Mater 14:1857–1860. https://doi.org/10.1002/adma.200290020
Fu Y, Zhang J, Liu H, Hiscox WC, Gu Y (2013) Ionic liquid-assisted exfoliation of graphite oxide for simultaneous reduction and functionalization to graphenes with improved properties. J Phys Chem A 1:2663. https://doi.org/10.1039/c2ta00353h
Gao X, Jiang L (2004) Water-repellent legs of water striders. Nature 432:36. https://doi.org/10.1038/432036a
Gu Z, Uetsuka H, Takahashi K, Nakajima R, Onishi H, Fujishima A, Sato O (2004) Structural color and the Lotus effect. Angew Chem Int Ed 42:922–925. https://doi.org/10.1002/ange.200390204
Han JH, Cho KW, Lee K-H, Kim H (1998) Porous graphite matrix for chemical heat pumps. Carbon 36:1801–1810. https://doi.org/10.1016/S0008-6223(98)00150-X
Klinowski J, He H, Forster M, Lerf A (1998) A new structural model for graphite oxide. Chem Phys Lett 287:53–56. https://doi.org/10.1016/S0009-2614(98)00144-4
Kozbial A, Li Z, Sun J, Gong X, Zhou F, Wang Y, Xu H, Liu H, Li L (2014) Understanding the intrinsic water wettability of graphite. Carbon 74:218–225. https://doi.org/10.1016/j.carbon.2014.03.025
Krishnamurthy G, Namitha RB (2013) Synthesis of structurally novel carbon micro/nanosphere by low temperatutre-hydrothermal process. J Chil Chem Soc 58:1930–1933. https://doi.org/10.4067/S0717-97072013000300030
Kuila T, Bose S, Khanra P, Mishra AK, Kim NH, Lee JH (2012) A green approach for the reduction of graphene oxide by wild carrot root. Carbon 50:914–921. https://doi.org/10.1016/j.carbon.2011.09.053
Le GTT, Manyam J, Opaprakasit P, Chanlek N, Grisdanurak N, Sreearunothai P (2018) Divergent mechanisms for thermal reduction of graphene oxide and their highly different ion affinities. Diam Relat Mater 89:246–256. https://doi.org/10.1016/j.diamond.2018.09.006
Lerf A, He H, Forster M, Klinowski J (1998) Structure of graphite oxide revisited. J Phys Chem B 5647:4477–4482. https://doi.org/10.1021/jp9731821
Li S, Li H, Wang X, Song Y, Liu Y, Jiang L, Zhu D (2002) Super-hydrophobicity of large-area honeycomb-like aligned carbon nanotubes. J Phys Chem B 106:9274–9276. https://doi.org/10.1021/jp0209401
Li Z, Wang Y, Kozbial A, Shenoy G, Zhou F, McGinley R, Ireland P, Morganstein B, Kunkel A, Surwade SP, Li L, Liu H (2013) Effect of airborne contaminants on the wettability of supported graphene and graphite. Nat Mater 12:925–931. https://doi.org/10.1038/nmat3709
Liang M, Wei Y, Hou L, Wang H, Li Y, Guo C (2016) Fabrication of a super-hydrophobic surface on a magnesium alloy by a simple method. J Alloys Compd 656:311–317. https://doi.org/10.1016/j.jallcom.2015.09.234
Lin Y, Ehlert GJ, Bukowsky C, Sodano HA (2011) Superhydrophobic functionalized graphene aerogels. Appl Mater Interfaces 3:2200–2203
Liu Y, Xiu YH, Hess DW, Wong CP (2010) Silicon surface structure-controlled oleophobicity. Langmuir 26:8908–8913. https://doi.org/10.1021/la904686c
Liu K, Cao M, Fujishima A, Jiang L (2014) Bio-inspired titanium dioxide materials with special wettability and their applications. Chem Rev 114:10044–10094. https://doi.org/10.1021/cr4006796
Liu X, Shao XY, Fang GB, He HF, Wan ZG (2016) Preparation and properties of chemically reduced graphene oxide/copolymer-polyamide nanocomposites. E-Polymers. https://doi.org/10.1515/epoly-2016-0094
Luna M, Colchero J, Baró AM (1999) Study of water droplets and films on graphite by noncontact scanning force microscopy. J Phys Chem B 103:9576–9581. https://doi.org/10.1021/jp991256y
Ma ML, Hill RM (2006) Superhydrophobic surfaces. Curr Opin Colloid Interface Sci 11:193–202. https://doi.org/10.1016/j.cocis.2006.06.002
Manoharan K, Bhattacharya S (2019) Superhydrophobic surfaces review: functional application, fabrication techniques and limitations. J Micromanuf 2:59–78
Marmur A (2003) Wetting on hydrophobic rough surfaces: to be heterogeneous or not to be? Langmuir 19:8343–8348. https://doi.org/10.1021/la0344682
Marmur A, Lelah MD (1981) The spreading of aqueous surfactant solutions on glass. Chem Eng Commun 13:133–143. https://doi.org/10.1080/00986448108910901
Morcos I (1970) On contact angle and dispersion energy of the cleavage graphite/water system. J Colloid Interface Sci 34:469–471. https://doi.org/10.1016/0021-9797(70)90207-9
Morcos I (1972) Surface tension of stress-annealed pyrolytic graphite. J Chem Phys 57:1801–1803. https://doi.org/10.1063/1.1678482
Murakami D, Jinnai H, Takahara A (2014) Wetting transition from the Cassie–Baxter state to the Wenzel state on textured polymer surfaces. Langmuir 30:2061–2067. https://doi.org/10.1021/la4049067
Neinhuis C, Barthlott W (1997) Characterization and distribution of water-repellent, self-cleaning plant surfaces. Ann Bot 79:667–677. https://doi.org/10.1006/anbo.1997.0400
Nekahi PH, Haghshenas MD (2014) Transparent conductive thin film of ultra large reduced graphene oxide monolayers. Appl Surf Sci 295:59–65. https://doi.org/10.1016/j.apsusc.2014.01.004
Nosonovsky M (2007) Multiscale roughness and stability of superhydrophobic biomimetic interfaces. Langmuir 23:3157–3161. https://doi.org/10.1021/la062301d
Ou J, Wang J, Liu S, Mu B, Ren J, Wang H (2010) Tribology study of reduced graphene oxide sheets on silicon substrate synthesized via covalent assembly. Langmuir 26:15830–15836. https://doi.org/10.1021/la102862d
Parker AR, Lawrence CR (2001) Water capture by a desert beetle. Nature 414:33–34. https://doi.org/10.1038/35102108
Parvate S, Dixit P, Chattopadhyay S (2020) Superhydrophobic surfaces: Insights from theory and experiment. J Phys Chem B 124:1323–1360. https://doi.org/10.1021/acs.jpcb.9b08567
Pei S, Cheng HM (2012) The reduction of graphite oxide. Carbon 50:3210–3228. https://doi.org/10.1016/j.carbon.2011.11.010
Perrozzi F, Croce S, Treossi E, Palermo V, Santucci S, Fioravanti G, Ottaviano L (2014) Reduction dependent wetting properties of graphene oxide. Carbon 77:473–480. https://doi.org/10.1016/j.carbon.2014.05.052
Prakash CGJ, Prasanth R (2020) Recent trends in fabrication of Nepenthes inspired SLIPs: design strategies for self-healing efficient anti-icing surfaces. Surf Interfaces 21:100678–100690. https://doi.org/10.1016/j.surfin.2020.100678
Price EK, Bansala T, Achee TC, Sun W, Green MJ (2019) Tunable dispersibility and wettability of graphene oxide through one-pot functionalization and reduction. J Colloid Interface Sci 552:771–780. https://doi.org/10.1016/j.jcis.2019.05.097
Raj R, Maroo SC, Wang EN (2013) Wettability of graphene. Nano Lett 13:1509–1515. https://doi.org/10.1021/nl304647t
Ramos-Galicia L, Mendez LN, Martínez-Hernández AL, Espindola-Gonzalez A, Galindo-Esquivel IR, Fuentes-Ramirez R, Velasco-Santos C (2013) Improved performance of an epoxy matrix as a result of combining graphene oxide and reduced graphene. Int J Polym Sci 2013:1–7. https://doi.org/10.1155/2013/493147
Schrader ME (1975) Ultrahigh vacuum techniques in the measurement of contact angles. IV. Water on graphite (0001). J Phys Chem 79:2508–2515. https://doi.org/10.1021/j100590a013
Shibuichi S, Onda T, Satoh N, Tsujii K (1996) Super-water-repellent fractal surfaces. Langmuir 12:2125–2127. https://doi.org/10.1021/la950418o
Shin YJ, Wang Y, Huang H, Kalon G, Wee ATS, Shen Z (2010) Surface-energy engineering of graphene. Langmuir 26:3798–3802. https://doi.org/10.1021/la100231u
Smith AT, LaChance AM, Zeng S, Liu B, Sun L (2019) Synthesis, properties, and applications of graphene oxide/reduced graphene oxide and their nanocomposites. Nano Mater Sci 1:31–47. https://doi.org/10.1016/j.nanoms.2019.02.004
Song D, Song B, Hu H, Du X, Zhou F (2015) Selectively splitting a droplet using superhydrophobic stripes on hydrophilic surfaces. Phys Chem Chem Phys 17:13800–13803. https://doi.org/10.1039/C5CP01530H
Stuart B (2004) Infrared spectroscopy: fundamental and applications. Wiley, New York, pp 1–248
Su Y, Kravets VG, Wong SL, Waters J, Geim AK, Nair RR (2014) Impermeable barrier films and protective coatings based on reduced graphene oxide. Nat Commun. https://doi.org/10.1038/ncomms5843
Tang Z, Zhang L, Zeng C, Lin T, Guo B (2012) General route to graphene with liquidlike behaviour by non-covalent modification. Soft Matter 8:9214–9220. https://doi.org/10.1039/C2SM26307F
Tomˇsiˇc B, Simoncic B, Orel B, Cerne L, Tavcer PF, Zorko M, Jerman I, Vilcnik A, Kovac J (2008) sol-gel coating of cellulose fibres with anti-microbials and repellent properties. J Sol-Gel Sci 47:44–57. https://doi.org/10.1007/s10971-008-1732-1
Van Engers CD, Cousens NEA, Babenko V, Britton J, Zappone B, Grobert N, Perkin S (2017) Direct measurement of the surface energy of graphene. Nano Lett 17:3815–3821. https://doi.org/10.1021/acs.nanolett.7b01181
Vijayan PP, Puglia D (2019) Biomimetic multifunctional materials: a review. Emerg Mater 2:391–415. https://doi.org/10.1007/s42247-019-00051-
Vinayan BP, Nagar R, Raman BN, Rajalakshami KS, Dhathathreyan BS, Ramaprabhu A (2012) Synthesis of graphene-multiwalled carbon nanotubes hybrid nanostructure by strengthened electrostatic interaction and its lithium-ion battery application. J Mater Chem 22:9949–9956. https://doi.org/10.1039/C2JM16294F
Wang S, Zhang Y, Abidi N, Cabrales L (2009) Wettability and surface free energy of graphene films. Langmuir 25:11078–11081. https://doi.org/10.1021/la901402f
Wang Z, Elimelech M, Lin S (2016) Environmental applications of interfacial materials with special wettability. Environ Sci Technol 50:2132–2150. https://doi.org/10.1021/acs.est.5b04351
Wu W, Wang X, Liu X, Zhou F (2009) Spray-coated fluorine-free superhydrophobic coatings with easy repairability and applicability. ACS Appl Mater Interfaces 8:1656–1661. https://doi.org/10.1021/am900136k
Yaragalla S, Meera AP, Kalarikkal N, Thomas S (2015) Chemistry associated with natural rubber-graphene nanocomposites and its effect on physical and structural properties. Ind Crops Prod 74:792–802. https://doi.org/10.1016/j.indcrop.2015.05.079
Ye H, Zhu L, Li W, Liu H, Chen H (2016) Constructing fluorine-free and cost-effective superhydrophobic surface with normal-alcohol-modified hydrophobic SiO2 nanoparticles. ACS Appl Mater Interfaces 9:858–867. https://doi.org/10.1021/acsami.6b12820
Yu H, Zhang B, Bulin C, Li R, Xing R (2016) High-efficient synthesis of graphene oxide based on improved hummers method. Sci Rep 6:36143. https://doi.org/10.1038/srep36143
Zaaba NI, Foo KL, Hashim U, Tan SJ, Liu W-W, Voon CH (2017) Synthesis of graphene oxide using modified hummers method: solvent influence. Procedia Eng 184:469–477. https://doi.org/10.1016/j.proeng.2017.04.118
Zhang X, Shi F, Niu J, Jiang YG, Wang ZQ (2008) Superhydrophobic surfaces: from structural control to functional application. J Mater Chem 18:621–633. https://doi.org/10.1039/B711226B
Zhang H, Zheng W, Yan Q, Yang Y, Wang J, Lu Z, Ji G, Yu Z (2010) Electrically conductive polyethylene terephthalate/graphene nanocomposites prepared by melt compounding. Polymer 51:1191–1196. https://doi.org/10.1016/j.polymer.2010.01.027
Zhang Y-L, Xia H, Kim E, Sun H-B (2012) Recent developments in superhydrophobic surfaces with unique structural and functional properties. Soft Matter 8:11217–11231. https://doi.org/10.1039/C2SM26517F
Zhang H, Liu Y, Zhang Z, Hua M, Dong G (2021) A superhydrophobic surface patterned with hydrophilic channels for directional sliding control and manipulation of droplets. Surf Coat Technol 409:126836–126845. https://doi.org/10.1016/j.surfcoat.2021.126836
Funding
Authors are highly thankful to Science and Engineering Research Board (SERB), India for the financial support (SRG/2019/000846).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (AVI 66971 KB)
Supplementary file2 (MP4 939 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bala, M., Singh, V. Facile fabrication of robust self-cleaning fluorine-free reduced graphene oxide based superhydrophobic surfaces. Chem. Pap. 77, 3373–3384 (2023). https://doi.org/10.1007/s11696-023-02710-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-02710-w