Abstract
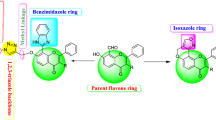
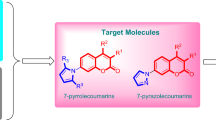
A series of flavonoid derivatives containing thiazole scaffold were designed, synthesized and evaluated for their inhibition effects against Rhizoctonia solani, Fusarium graminearum, Gaeumannomyces graminis, Alternaria solani, Botrytis cinerea, and Alternaria mali. The antifungal bioassays results in vitro indicated that some target compounds showed good antibacterial activity against the tested plant fungi, which were better than that of the flavonoid natural product quercetin. Strikingly, the title compounds 6a, 6b, and 6f demonstrated obvious antifungal activities against Rhizoctonia solani with EC50 value of 9.6, 35.5, and 21.5 μg/mL, respectively. The EC50 value of compound 6b against Gaeumannomyces graminis reached 2.7 μg/mL.
Graphical abstract




Similar content being viewed by others
References
Ah YC, Choi JK, Choi YK, Ki HM, Bae JH (2010) A novel transdermal patch incorporating meloxicam: in vitro and in vivo characterization. Int J Pharm 385:12–19. https://doi.org/10.1016/j.ijpharm.2009.10.013
Al Aboody MS, Mickymaray S (2020) Anti-fungal efficacy and mechanisms of flavonoids. Antibiotics 9:45–87. https://doi.org/10.3390/antibiotics9020045
Ballari MS, Cano NH, Lopez AG, Wunderlin DA, Feresin G, Santiago AN (2017) Green synthesis of potential antifungal agents: 2-benzyl substituted thiobenzoazoles. J Agric Food Chem 65:10325–10331. https://doi.org/10.1021/acs.jafc.7b04130
Benavente-García O, Castillo J (2008) Update on uses and properties of citrus flavonoids: new findings in anticancer, cardiovascular, and anti-inflammatory activity. J Agric Food Chem 56:6185–6205. https://doi.org/10.1021/jf8006568
Bhatt D, Soni R, Sharma GK, Dashora A (2016) Synthesis and pharmacological activities of flavones: a review. Indo Am J Pharm Res 6:4345–4363
Bhunia A, Patra A, Biju AT (2013) NHC-catalyzed reaction of enals with hydroxy chalcones: diastereoselective synthesis of functionalized coumarins. Org Lett 15:1756–1759. https://doi.org/10.1021/ol400562z
Guo X, Zhao B, Fan Z, Yang D, Zhang N, Wu Q, Yu B, Zhou S, Kalinina T, Belskaya NP (2019) Discovery of novel thiazole carboxamides as antifungal succinate dehydrogenase inhibitors. J Agric Food Chem 67:1647–1655. https://doi.org/10.1021/acs.jafc.8b06935
Jia LG, Sheng ZW, Xu WF, Li YX, Liu YG, Xia YJ, Zhang JH (2012) Modulation of anti-oxidation ability by proanthocyanidins during germination of arabidopsis thaliana seeds. Mol Plant 5:472–481. https://doi.org/10.1093/mp/ssr089
Jin YS (2019) Recent advances in natural antifungal flavonoids and their derivatives. Bioorg Med Chem Lett 29:126589–126601. https://doi.org/10.1016/j.bmcl.2019.07.048
Kaishap PP, Duarah G, Sarma B, Chetia D, Gogoi S (2018) Ruthenium(II)-catalyzed synthesis of spirobenzofuranones by a decarbonylative annulation reaction. Angew Chem Int Ed 57:456–460. https://doi.org/10.1002/anie.201710049
Kayser H, Wellmann H, Lee C, Decock A, Gomes M, Cheek B, Lind R, Baur M, Hattenschwiler J, Maienfisch P (2007) Thiamethoxam: high-affinity binding and unusual mode of interference with other neonicotinoids at aphid membranes. Synth Chem Agrochem VII 948:67–81. https://doi.org/10.1021/bk-2007-0948.ch006
Kowalski K, Koceva-Chyła A, Szczupak Ł, Hikisz P, Bernasińska J, Rajnisz A, Solecka J, Therrien B (2013) Ferrocenylvinyl-flavones: synthesis, structure, anticancer and antibacterial activity studies. J Organomet Chem 741–742:153–161. https://doi.org/10.1016/j.jorganchem.2013.05.009
Lamberth C, Jeanmart S, Luksch T, Plant A (2013) Current challenges and trends in the siscovery of agrochemicals. Science 341:742–746. https://doi.org/10.1126/science.1237227
Lee MH, Pan SM, Ng TW, Chen PS, Wang LY, Chung KR (2011) Mutations of beta-tubulin codon 198 or 200 indicate thiabendazole resistance among isolates of Penicillium digitatum collected from citrus in Taiwan. Int J Food Microbiol 150:157–163. https://doi.org/10.1016/j.ijfoodmicro.2011.07.031
Lu A, Wang J, Liu T, Han J, Li Y, Su M, Chen J, Zhang H, Wang L, Wang Q (2014) Small changes result in large differences: discovery of (−)-incrustoporin derivatives as novel antiviral and antifungal agents. J Agric Food Chem 62:8799–8807. https://doi.org/10.1021/jf503060k
Meng F, Yan Z, Yang W, Yuan S, Dong S, Feng J, Deng W (2022) Preparation of thiazole containing flavonoid derivatives as agricultural fungicides. China, CN113999223 A 2022-02-01
Ray M, Ray A, Dash S, Mishra A, Achary KG, Nayak S, Singh S (2017) Fungal disease detection in plants: traditional assays, novel diagnostic techniques and biosensors. Biosens Bioelectron 87:708–723. https://doi.org/10.1016/j.bios.2016.09.032
Razzaghi-Abyaneh M, Rai M (eds) (2013) Antifungal metabolites from plant. 1–469. Berlin, Heidelberg: Springer. https://doi.org/10.1007/978-3-642-38076-1_1
Teoh D, Ayeni TA, Rubatt JM, Adams DJ, Grace L, Starr MD, Barry WT, Berchuck A, Murphy SK, Secord AA (2011) Dasatinib (BMS-35482) has synergistic activity with paclitaxel and carboplatin in ovarian cancer cells. Gynecol Oncol 121:187–192. https://doi.org/10.1016/j.ygyno.2010.11.017
Wang L, Li C, Zhang Y, Qiao C, Ye Y (2013) Synthesis and biological evaluation of benzofuroxan derivatives as fungicides against phytopathogenic fungi. J Agric Food Chem 61:8632–8640. https://doi.org/10.1021/jf402388x
Wang W, Zhang S, Wang J, Wu F, Wang T, Xu G (2021) Bioactivity-guided synthesis accelerates the discovery of 3-(iso)quinolinyl-4-chromenones as potent fungicide candidates. J Agric Food Chem 69:491–500. https://doi.org/10.1021/acs.jafc.0c06700
Wei DG, Yang GF, Wan J, Zhan CG (2005) Binding model construction of antifungal 2-aryl-4-chromanones using CoMFA, CoMSIA, and QSAR analyses. J Agric Food Chem 53:1604–1611. https://doi.org/10.1021/jf048313r
Yan Z, Liu A, Ou Y, Li J, Yi H, Zhang N, Liu M, Huang L, Ren J, Liu W, Hu A (2019) Design, synthesis and fungicidal activity evaluation of novel pyrimidinamine derivatives containing phenyl-thiazole/oxazole moiety. Bioorg Med Chem 27:3218–3228. https://doi.org/10.1016/j.bmc.2019.05.029
Zhao PL, Li J, Yang GF (2007) Synthesis and insecticidal activity of chromanone and chromone analogues of diacylhydrazines. Bioorg Med Chem 15:1888–1895. https://doi.org/10.1016/j.bmc.2007.01.008
Acknowledgements
The authors gratefully acknowledge the National Nature Science Foundation of China (No. 32202334) and High Level Introduction of Talent Research Start-up Foundation of Yangzhou University (No. 137012166).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Structures presented in this publication are claimed as agricultural fungicides in a patent (Meng et al. 2022).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, F., Yan, Z., Lu, Y. et al. Design, synthesis, and antifungal activity of flavonoid derivatives containing thiazole moiety. Chem. Pap. 77, 877–885 (2023). https://doi.org/10.1007/s11696-022-02522-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02522-4