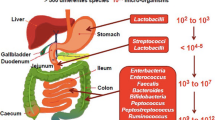
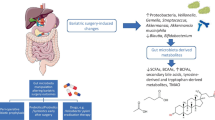
Abstract
We aim to review the available literature on obese patients treated with bariatric procedures, in order to assess their effect on the metabolic and gut microbiota profiles. A systematic literature search was performed in PubMed, Cochrane library, and Scopus databases, in accordance with the PRISMA guidelines. Twenty-two studies (562 patients) met the inclusion criteria. This study points to significant amelioration of postoperative levels of glucose, insulin, triglycerides, total cholesterol, LDL, HDL, HOMA-IR, food intake, and diabetes remission. Branched-chain amino acids (BCAAs) decreased, while trimethylamine-n-oxide (TMAO); glucagon-like peptide 1, 2 (GLP-1, GLP-2); and peptide YY (PYY) increased postoperatively. Postoperative gut microbiota was similar to that of lean and less obese objects. Well-designed randomized trials are necessary to further assess the host metabolic-microbial cross-talk after bariatric procedures.


Similar content being viewed by others
References
Flegal KM, Carroll MD, Kit BK, et al. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. J Am Med Assoc. 2012;307(5):491–7.
Sjφstrφm L, Peltonen M, Jacobson P, et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA. American Medical Association. 2014;311:2297–304. doi:10.1001/jama.2014.5988.
Sjφstrφm L, Peltonen M, Jacobson P, et al. Bariatric surgery and long-term cardiovascular events. JAMA American Medical Association. 2012;307:56–65.
Stewart B, WCP editors. IA for R on CW. World cancer report 2014. 2014; Available: http://www.thehealthwell.info/node/725845
Colquit JL, Picot J, Loveman E, et al. Surgery for obesity. Cochrane Database Syst Rev. 2009;(2):CD003641.
Kaska L, Sledzinski T, Chomiczewska A, et al. Improved glucose metabolism following bariatric surgery is associated with increased circulating bile acid concentrations and remodeling of the gut microbiome. World J Gastroenterol. 2016;22(39):8698–719.
Quercia I, Dutia R, Kotler DP, et al. Gastrointestinal changes after bariatric surgery. Diabetes Metab. 2014;40:87–94. doi:10.1016/j.diabet.2013.11.003.
Ley RE, Turnbaugh PJ, Klein S, et al. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444:1022–3.
Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480–4.
Flint HJ. Obesity and the gut microbiota. J Clin Gastroenterol. 2011;45(Suppl):S128–32.
Li JV, Ashrafian H, Bueter M, et al. Metabolic surgery profoundly influences gut microbial-host metabolic cross-talk. Gut. 2011;60(9):1214–23.
Ashrafian H, Li JV, Spagou K, et al. Bariatric surgery modulates circulating and cardiac metabolites. J Proteome Res. 2014;13:570–80. doi:10.1021/pr400748f.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100.
Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration. 2011 Available from www.cochrane-handbook.org
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–5.
H.J. GSE. (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]
Egger M, Smith DG, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Alamuddin N, Vetter ML, Ahima RS, et al. Changes in fasting and prandial gut and adiposity hormones following vertical sleeve gastrectomy or Roux-en-Y-gastric bypass: an 18-month prospective study. Obes Surg. 2016; doi:10.1007/s11695-016-2505-5.
Arora T, Velagapudi V, Pournaras DJ, et al. Roux-en-Y gastric bypass surgery induces early plasma metabolomic and lipidomic alterations in humans associated with diabetes remission. PLoS One. 2015;10(5):e0126401. doi:10.1371/journal.pone.0126401.
Federico A, Dallio M, Tolone S, et al. Gastrointestinal hormones, intestinal microbiota and metabolic homeostasis in obese patients: effect of bariatric surgery. in vivo. 2016;30:321–30.
Furet JP, Kong LC, Tap J, et al. Differential adaptation of human gut microbiota to bariatric surgery-induced weight loss: links with metabolic and low-grade inflammation markers. Diabetes. 2010;59(12):3049–57. doi:10.2337/db10-0253.
Graessler J, Qin Y, Zhong H, et al. Metagenomic sequencing of the human gut microbiome before and after bariatric surgery in obese patients with type 2 diabetes: correlation with inflammatory and metabolic parameters. The Pharmacogenomics Journal. 2013;13:514–22. doi:10.1038/tpj.2012.43.
Gralka E, Luchinat C, Tenori L, et al. Metabolomic fingerprint of severe obesity is dynamically affected by bariatric surgery in a procedure-dependent manner. Am J Clin Nutr. 2015; doi:10.3945/ajcn.115.110536.
Ishida RK, Faintuch J, Ribeiro AS, et al. Asymptomatic gastric bacterial overgrowth after bariatric surgery: are long-term metabolic consequences possible? Obes Surg. 2014;24:1856–61. doi:10.1007/s11695-014-1277-z.
Jüllig M, Yip S, Xu A, et al. Lower fetuin-A, retinol binding protein 4 and several metabolites after gastric bypass compared to sleeve gastrectomy in patients with type 2 diabetes. PLoS One. 2014;9(5):e96489. doi:10.1371/journal.pone.0096489.
Karamanakos SN, Vagenas K, Kalfarentzos F, et al. Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: a prospective, double blind study. Ann Surg. 2008;247:401–7.
Kong LC, Tap J, Aron-Wisnewsky J, et al. Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. Am J Clin Nutr. 2013;98:16–24.
Lips MA, Van Klinken JB, van Harmelen V, et al. Roux-en-Y gastric bypass surgery, but not calorie restriction, reduces plasma branched-chain amino acids in obese women independent of weight loss or the presence of type 2 diabetes. Diabetes Care. 2014;37:3150–6. doi:10.2337/dc14-0195.
Lopes TIB, Geloneze B, Pareja JC, et al. “Omics” prospective monitoring of bariatric surgery: Roux-en-Y gastric bypass outcomes using mixed-meal tolerance test and time-resolved 1H NMR-based metabolomics. OMICS A J Integ Biol 2016;20(7). doi:10.1089/omi.2016.0061
Magkos F, Bradley D, Schweitzer GG, et al. Effect of Roux-en-Y gastric bypass and laparoscopic adjustable gastric banding on branched-chain amino acid metabolism. Diabetes 2013;62. DOI: 10.2337/db13-0185.
Murphy R, Tsai P, Jüllig M, et al. Differential changes in gut microbiota after gastric bypass and sleeve gastrectomy bariatric surgery vary according to diabetes remission. Obes Surg. 2016; doi:10.1007/s11695-016-2399-2.
Narath SH, Mautner SI, Svehlikova E, et al. An untargeted metabolomics approach to characterize short-term and long-term metabolic changes after bariatric surgery. PLoS One. 2016;11(9):e0161425. doi:10.1371/journal.pone.0161425.
Palleja A, Kashani A, Allin KH, et al. Roux-en-Y gastric bypass surgery of morbidly obese patients induces swift and persistent changes of the individual gut microbiota. Genome Medicine. 2016;8:67. doi:10.1186/s13073-016-0312-1.
Papamargaritis D, le Roux CW, Sioka E, et al. Changes in gut hormone profile and glucose homeostasis after laparoscopic sleeve gastrectomy. Surg Obes Relat Dis. 2013;9:192–201.
Patrone V, Vajana E, Minuti A, et al. Postoperative changes in fecal bacterial communities and fermentation products in obese patients undergoing bilio-intestinal bypass. Front Microbiol. 2016;7:200. doi:10.3389/fmicb.2016.00200.
Peterli R, Wφlnerhanssen B, Peters T, et al. Improvement in glucose metabolism after bariatric surgery: comparison of laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy: a prospective randomized trial. Ann Surg. 2009 Aug;250(2):234–41. doi:10.1097/SLA.0b013e3181ae32e3.
Tremaroli V, Karlsson F, Werling M, et al. Roux-en-Y gastric bypass and vertical banded gastroplasty induce long-term changes on the human gut microbiome contributing to fat mass regulation. Cell Metab. 2015;22:228–38. doi:10.1016/j.cmet.2015.07.009.
Trøseid M, Hov JR, Nestvold TK, et al. Major increase in microbiota-dependent proatherogenic metabolite TMAO one year after bariatric surgery. METABOLIC SYNDROME AND RELATED DISORDERS Volume 14, Number 4, 2016 DOI: 10.1089/met.2015.0120
Valderas JP, Irribarra V, Rubio L, et al. Effects of sleeve gastrectomy and medical treatment for obesity on glucagon-like peptide 1 levels and glucose homeostasis in non-diabetic subjects. Obes Surg. 2011;21:902–9. doi:10.1007/s11695-011-0375-4.
Ley RE, Bäckhed F, Turnbaugh P, et al. Obesity alters gut microbial ecology. Proc Natl Acad Sci U S A. 2005;102:11070–5. doi:10.1073/pnas.0504978102.
Turnbaugh PJ, Bδckhed F, Fulton L, et al. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008;3:213–23. doi:10.1016/j.chom.2008.02.015.
Buchwald H, Oien DM. Metabolic/bariatric surgery worldwide 2011. Obes Surg. 2013;23:427–36.
Horowitz M, Cook DJ, Collins PJ, et al. Measurement of gastric emptying after gastric bypass surgery using radionuclides. Br J Surg. 1982;69:655–7.
Melissas J, Koukouraki S, Askoxylakis J, et al. Sleeve gastrectomy: a restrictive procedure? Obes Surg. 2007;17:57–62.
Thaler JP, Cummings DE. Minireview: Hormonal and metabolic mechanisms of diabetes remission after gastrointestinal surgery. Endocrinology. 2009;150:2518–25.
Rubino F, Forgione A, Cummings DE, et al. The mechanism of diabetes control after gastrointestinal bypass surgery reveals a role of the proximal small intestine in the pathophysiology of type 2 diabetes. Ann Surg. 2006;244:741–9.
Newgard CB, An J, Bain JR, et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009;9:311–26.
Adeva MM, Calviρo J, Souto G, et al. Insulin resistance and the metabolism of branched-chain amino acids in humans. Amino Acids. 2012;43:171–81.
Saha AK, Xu XJ, Balon TW, et al. Insulin resistance due to nutrient excess: is it a consequence of AMPK downregulation? Cell Cycle. 2011;10:3447–51.
Saha AK, Xu XJ, Lawson E, et al. Downregulation of AMPK accompanies leucine- and glucose-induced increases in protein synthesis and insulin resistance in rat skeletal muscle. Diabetes. 2010;59:2426–34.
Newgard CB. Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metab. 2012;15:606–14.
Magouliotis DE, Tasiopoulou VS, Sioka E, et al. Robotic versus laparoscopic sleeve gastrectomy for morbid obesity: a systematic review and meta-analysis. Obes Surg. 2017;27:245. doi:10.1007/s11695-016-2444-1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent does not apply.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Rights and permissions
About this article
Cite this article
Magouliotis, D.E., Tasiopoulou, V.S., Sioka, E. et al. Impact of Bariatric Surgery on Metabolic and Gut Microbiota Profile: a Systematic Review and Meta-analysis. OBES SURG 27, 1345–1357 (2017). https://doi.org/10.1007/s11695-017-2595-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2595-8